10-K: Annual report [Section 13 and 15(d), not S-K Item 405]
Published on March 10, 2026
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
_____________________________________
FORM 10-K
_____________________________________
(Mark One)
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the fiscal year ended December 31 , 2025
OR
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the transition period from _____ to _____
Commission file number 001-42771
_____________________________________
_____________________________________
(Exact name of registrant as specified in its charter)
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | ||||
(Address of Principal Executive Offices) | (Zip Code) | ||||
(616 ) 294-1026
Registrant’s telephone number, including area code
Securities registered pursuant to Section 12(b) of the Act:
Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||
Securities registered pursuant to section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Yes o No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act.
Yes o No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer | o | Accelerated filer | o | |||||||||||
x | Smaller reporting company | |||||||||||||
| Emerging growth company | ||||||||||||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report.
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements.
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b).
o
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act).
Yes o No x
As of June 30, 2025, the last business day of the Registrant’s most recently completed second fiscal quarter, there was no established public market for the Registrant’s common stock. Therefore, the aggregate market value of the Registrant’s common stock held by non-affiliates as of such date cannot be calculated. The Registrant’s common stock began trading on the New York Stock Exchange on July 31, 2025.
As of March 3, 2026, there were 20,647,526 shares of the Registrant’s common stock outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s definitive proxy statement for use in connection with its 2026 Annual Meeting of Stockholders, which is to be filed no later than 120 days after December 31, 2025, are incorporated by reference into Part III of this Annual Report on Form 10-K.
TABLE OF CONTENTS
| Page | |||||||||||
Special Note Regarding Forward-Looking Statements | |||||||||||
Risk Factors Summary | |||||||||||
2
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K for the year ended December 31, 2025 (“Annual Report”) contains forward-looking statements. We intend such forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). All statements other than statements of historical facts contained in this Annual Report, including but not limited to statements regarding our future results of operations and financial condition, our business plans and strategy, the potential market size, market trends and growth opportunities for our products, macroeconomic and geopolitical conditions, and the sufficiency of our cash balances, working capital and cash generated from our operations, investing and financing activities for future liquidity and capital resource needs, may be forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “would,” “should,” “expects,” “plans,” “anticipates,” “could,” “intends,” “targets,” “projects,” “contemplates,” “believes,” “estimates,” “forecasts,” “predicts,” “potential” or “continue” or the negative of these terms or other similar expressions that convey uncertainty of future events or outcomes, although not all forward-looking statements contain these identifying words.
The forward-looking statements in this Annual Report are only predictions. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition and results of operations. Forward-looking statements involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements, including, but not limited to, the important factors discussed in Part I, Item 1A “Risk Factors” in this Annual Report. The forward-looking statements in this Annual Report are based upon information available to us as of the date of this Annual Report, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain and investors are cautioned not to unduly rely upon these statements. Moreover, we operate in a very competitive and rapidly changing environment, and new risks emerge from time to time. It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. In light of these risks, uncertainties and assumptions, the forward-looking events and circumstances discussed in this Annual Report may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements.
You should read this Annual Report and the documents that we reference in this Annual Report and have filed as exhibits to this Annual Report with the understanding that our actual future results, performance and achievements may be materially different from what we expect. We qualify all of our forward-looking statements by these cautionary statements. These forward-looking statements speak only as of the date of this Annual Report. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained in this Annual Report, whether as a result of any new information, future events or otherwise.
As used in this Annual Report, unless otherwise stated or the context requires otherwise, the terms “Shoulder Innovations,” the “Company,” “we,” “us,” and “our” refer to Shoulder Innovations, Inc. “Shoulder Innovations,” the Shoulder Innovations logos, and other trade names, trademarks, or service marks of Shoulder Innovations appearing in this Annual Report are the property of Shoulder Innovations. Other trade names, trademarks, or service marks appearing in this Annual Report are the property of their respective holders. We do not intend our use or display of other companies’ trade names, trademarks, or service marks to imply a relationship with, or endorsement or sponsorship of us, by these other companies. Solely for convenience, trade names, trademarks, and service marks referred to in this Annual Report appear without the ®, ™, and SM symbols, but those references are not intended to indicate, in any way, that we will not assert, to the fullest extent under applicable law, our rights or that the applicable owner will not assert its rights, to these trade names, trademarks, and service marks.
3
RISK FACTORS SUMMARY
Our business is subject to numerous risks and uncertainties, including those described in Part I, Item 1A “Risk Factors” in this Annual Report. You should carefully consider these risks and uncertainties when investing in our common stock. If any of these risks actually occur, it could have a material adverse effect on our business, financial condition, and results of operations. In such case, the trading price of our common stock would likely decline, and you could lose all or part of your investment. The principal risks affecting our business include, but are not limited to:
•We are an early-stage company with a history of significant net losses, we expect to incur operating losses in the future and we may not be able to achieve or sustain profitability.
•If we fail to manage our growth effectively, our business could be materially and adversely affected.
•We have a significant amount of debt, which may affect our ability to operate our business and secure additional financing in the future.
•We may require substantial additional funding, which may not be available to us on acceptable terms, or at all, and, if not available, may require us to delay, scale back, or cease our innovation efforts or operations.
•We operate in a very competitive business environment, and if we are unable to compete successfully against our existing or potential competitors, our business, financial condition and results of operations may be adversely affected.
•If we fail to develop and retain an effective commercial organization, or if we are unable to successfully expand our organization, it could negatively impact our sales, and we may not generate sufficient net revenue to sustain profitability.
•Our business plan relies on certain assumptions about the market for our implant systems, however, the size and expected growth of our addressable market has not been established with precision and may be smaller than we estimate, and even if the addressable market is as large as we have estimated, we may not be able to capture additional market share.
•Our business is dependent upon the adoption of our implant systems by hospitals, ambulatory surgery centers (“ASCs”), surgeons and patients.
•Our long-term growth depends on our ability to enhance our implant systems, expand our indications and develop and commercialize additional products in a timely manner. If we cannot innovate, we may not be able to develop or exploit new products in time to remain competitive.
•We face the risk of product liability claims that could be expensive, divert management’s attention and harm our reputation and business. We may not be able to maintain adequate product liability insurance.
•Industry trends have resulted in increased downward pricing pressure on medical services and products, which may affect our ability to sell our products at prices necessary to support our current business strategy.
•If hospitals, ASCs, and other health care facilities do not approve the use of our implant systems, our sales may not increase.
•We depend on third-party contract manufacturers and suppliers, some of which are single source, to produce and package all elements comprising our shoulder implant systems, and if these suppliers and manufacturers fail to supply us, our products or their components or subcomponents in sufficient quantities or at all, or in accordance with applicable regulatory requirements and our specifications, it will have a material adverse effect on our business, financial condition, and results of operations.
•Our results of operations will be materially harmed if we are unable to accurately forecast demand for our implant systems and manage our inventory.
•We may be unable to continue to successfully demonstrate to shoulder specialists or key opinion leaders the merits of our implant systems and technologies compared to those of our competitors, which may make it difficult to achieve market acceptance.
•The loss of any member of our executive management team or our inability to attract and retain highly skilled members of our dedicated commercial leadership and marketing teams, as well as certain third-party engineers, could have a material adverse effect on our business, financial condition and results of operations.
•If we are unable to obtain and maintain significant patent or other intellectual property protection for our products, or if the scope of our patents and other intellectual property rights do not adequately protect our products, our competitors could develop and commercialize products similar or identical to ours and we may be unable to gain significant market share and be unable to operate our business profitably.
4
•Our devices and operations are subject to extensive government regulation and oversight in the United States, and our failure to comply with applicable requirements could harm our business.
•Our relationships with customers, physicians and third-party payors are subject to federal and state health care fraud and abuse laws, false claims laws, physician payment transparency laws and other health care laws and regulations. If we or our employees, independent contractors, consultants, commercial partners, or vendors violate these laws we could face substantial penalties.
5
Part I
Item 1. Business
Overview
We are a commercial-stage medical technology company exclusively focused on transforming the shoulder surgical care market. We currently offer advanced implant systems for shoulder arthroplasty. These systems are a core element of our ecosystem, which we designed to improve core components of shoulder surgical care – preoperative planning, implant design and procedural efficiency – to benefit each stakeholder in the care chain. Our ecosystem is also comprised of enabling technologies, efficient instrument systems, specialized support and surgeon-to-surgeon collaboration. Together, these elements seek to address the long-standing clinical and operational challenges in the shoulder surgical care market by delivering predictable outcomes, procedural simplicity, and efficiency across all sites of care. We believe our exclusive focus on shoulder surgical care, combined with a highly specialized commercial organization and strong clinical data, positions us well to capture significant share in this large, growing market.
Shoulder pain is highly prevalent, often chronic, and can significantly reduce quality of life. The primary conditions that can result in shoulder pain and reduced functionality include osteoarthritis, rheumatoid arthritis, rotator cuff tears and shoulder fractures. These shoulder conditions are widespread, often debilitating, and are commonly experienced concurrently as interrelated musculoskeletal disorders. According to data from the National Institutes of Health, we estimate that these conditions result in more than eight million physician visits annually in the United States. Despite this prevalence, we believe there has been a historical underutilization of surgical treatments for shoulder care due to several factors including patient hesitation to pursue surgical intervention, insufficient technology to appropriately treat shoulder conditions, complex shoulder anatomy, perceived difficulty of surgical intervention and barriers to patient access of care.
We believe the shoulder surgical care market today presents a significant market opportunity. Our initial focus within this broader market is on shoulder arthroplasty. Shoulder arthroplasty is an established surgical procedure involving the reconstruction of the shoulder joint with prosthetic implants through one of two main approaches: anatomic total shoulder arthroplasty (“aTSA”) and reverse total shoulder arthroplasty (“rTSA”). Both approaches can be performed in inpatient hospital settings and in outpatient settings, including ASCs. A key competitive advantage of ours has been the emergence of ASCs as a cost-efficient site of care with positive outcomes relative to hospital-based care. We expect that future growth in the shoulder surgical care market will be significantly driven by ASCs as hospitals face capacity constraints and are more limited in their ability to meet increasing demand.
We estimate that approximately 250,000 shoulder arthroplasty procedures were performed in the United States in 2025, which we believe represents an approximately $1.7 billion market opportunity. Based on our internal estimates, knowledge of our industry and third-party data regarding the number of shoulder arthroplasty procedures performed, we expect this market to grow by approximately 11% annually through 2029. We also believe a significant opportunity exists outside of the United States and, based on third-party industry research reports and our internal estimates, we estimate that the total annual international shoulder arthroplasty market is approximately $1.0 billion in 2025. Together, this represents a global annual shoulder arthroplasty market of approximately $2.7 billion. We believe we have an opportunity to expand to address adjacent areas in the shoulder surgical care market over time.
We believe traditional implants used in shoulder arthroplasty procedures are hindered by several limitations, including poor biomechanical fit, suboptimal kinematics, difficult replacement and conversion procedures (aTSA to rTSA or stemless to stem), imprecise implant positioning due to limited surgical planning, inefficient and burdensome workflow designs and non-specialized case support. These limitations can result in continued pain, lack of mobility, postoperative complications, low rates of implant survivorship, necessity of revision surgeries and costly and inefficient procedures for healthcare providers.
We developed our ecosystem with an approach to innovation that prioritizes ease of use, flexibility, predictability of outcomes and site of care efficiency, attributes we believe are critical to win in this market. Our ecosystem is comprised of the following elements:
•Advanced Implant Systems: Our advanced implants include a diverse range of interchangeable InSet anatomic and reverse total shoulder arthroplasty systems that leverage our novel, InSet Glenoid and InSet humeral stem technologies.
•ProVoyance Preoperative Planning Technology: This preoperative planning technology integrates artificial intelligence (“AI”) and machine learning (“ML”) to transform planar CT imaging into 3D renderings, allowing surgeons to create bespoke surgical plans considering patient-specific anatomy ahead of surgery.
6
•Efficient Instrument System: Our efficient instrument system supports both aTSA and rTSA procedures from start to finish with just two convenient trays, considerably less than the six to nine trays typically required by other offerings.
•Specialized Support: Our team of dedicated shoulder specialists enables us to deliver a highly tailored experience to surgeons operating in a complex and technically demanding procedure category.
•Surgeon-to-Surgeon Collaboration: Our Customer Experience and Medical Education (“CEME”) team fosters a collaborative network of expert surgeon educators and promotes surgeon-to-surgeon training and peer education.
Our ecosystem offers notable benefits that differentiate it within the shoulder surgical care market. These benefits include:
•Improved Fixation and Stability: Our foundational InSet Glenoid technology is biomechanically designed to specifically address what we believe is the primary problem in legacy aTSA implants: glenoid loosening.
•Restores Full Functionality: We engineered a range of humeral stem options to provide consistent, optimized biomechanics in both aTSA and rTSA.
•Exceptional Longevity: Our advanced implant systems are designed for longevity. In one published study, our InSet Glenoid demonstrated no surgical complications, cases of glenoid loosening or revision surgeries at a mean follow-up time of 8.7 years in a particularly challenging patient population.
•Improved Implant Selection and Positioning: ProVoyance preoperative planning technology empowers surgeons to create bespoke surgical plans that facilitate consistent, effective positioning of our implants.
•Optimized Procedural Workflow and Efficiency: Our efficient instrument system supports our implant portfolio across both aTSA and rTSA. This can provide significant workflow advantages for our customers by minimizing the operating room footprint, reducing procedural setup time, lowering sterilization requirements and costs and reducing the risk of errors – key advantages for the ASC setting.
•Access to Expert Advice: Our commercial organization creates deep relationships with surgeons and allows us to support their practice with specialized customer service and case support before, during and after surgery. In addition to direct support, our commercial team connects surgeons to the broader shoulder surgical community in various settings with a goal to improve connectivity across the shoulder surgical community and contribute to better patient outcomes.
We developed our comprehensive implant portfolio to address the unique needs of patients and surgeons. Our advanced implants are comprised of our aTSA and rTSA systems, which include various, specifically designed components capable of a wide array of system configurations to facilitate different modes of operation (anatomic or reverse) that are optimized for patient-specific needs. Our InSet Glenoid technology serves as the foundation for our advanced implant systems and includes a novel “InSet” design that aims to reduce mechanical stress at the bone implant interface, improve fixation mechanics, enhance stability and reduce micromotion. Our implant systems leverage consistent surgical techniques and the same efficient, two tray instrumentation system.
In addition to our advanced implant systems, we offer a leading preoperative surgical planning technology: ProVoyance. We believe that surgeon-level engagement in preoperative planning provides for better care for patients, and that bespoke surgical plans can help facilitate consistent positioning of implants. ProVoyance integrates AI and ML to transform planar CT imaging into 3D renderings of patient-specific anatomy ahead of surgery, and is cleared by the United States Food and Drug Administration (“FDA”) for preoperative shoulder planning. ProVoyance received 510(k) clearance in 2021 and is classified by the FDA as a Class II device. ProVoyance is listed on the FDA’s AI/ML-enabled medical devices list, which is a resource maintained, published, and periodically updated by the FDA to identify AI/ML-enabled devices that have been authorized for marketing in the United States through any of the standard paths to market for medical devices, although it is not intended to be a comprehensive list of all such devices that incorporate AI/ML. We believe the differentiation and value proposition of ProVoyance is validated by high utilization rates across procedures using our advanced implant systems. For the year ended December 31, 2025, to the extent we are able to measure, virtually all surgeries using our implants use ProVoyance, making it a routine part of surgeon workflow.
A key component of shoulder arthroplasty procedures are instrument trays, equipped with the specific instruments, supplies, and equipment needed for the surgery. We have developed a proprietary two tray instrument system designed to
7
enable interoperability between our aTSA and rTSA systems and a range of humeral stem options. We believe our efficient, two tray instrument system can enable surgeons and staff to reduce operating room footprint, procedural setup time, sterilization time and expense, and procedural complexity.
To best support our surgeon customers, we have built our product ecosystem around their unique needs. Our commercial organization is comprised of a dedicated commercial leadership team that drives our internal commercial efforts with an exclusive focus on shoulder care, a CEME team that enhances surgeon engagement and training, and a network of independent distributors. These three key components of our commercial organization work in tandem to form a commercial flywheel that is designed to build and provide key product support to surgeons and other stakeholders in the shoulder surgical care market, accelerate adoption, and enhance long-term retention.
We leverage our team’s decades of experience developing and launching novel shoulder surgical care technologies to identify the unmet needs of patients and surgeons and develop solutions to address those unmet needs. With respect to our advanced implant systems, we commenced development efforts with our InSet Glenoid in 2009 and received 510(k) clearance in 2011. We commercially launched an initial aTSA system with our InSet Glenoid in 2016. Since this initial launch in 2016, we have successfully launched a wide range of new technologies to enhance our ecosystem and provide surgeons with the tools and support needed to deliver quality outcomes for patients requiring shoulder surgical care. For example, we commenced development efforts for our InSet PLUS Augmented Glenoid in 2019 and received 510(k) clearance in 2020. We commenced development efforts for our rTSA system in 2019 and received 510(k) clearance in 2021. We commenced development efforts for our short stem, stemless and I-Series humeral stem system options for our aTSA and rTSA systems in 2017, 2019 and 2021, respectively, and received our primary 510(k) clearances in 2018 and 2022, with an additional 510(k) clearance in 2024 for use of our primary I-Series humeral stem for use with anatomic fractures. Each of these devices is classified by the FDA as a Class II device. We have a robust pipeline of new technologies in various stages of development and evaluation, including the anticipated expansion of our humeral stem line, indication expansions into fracture and revision, and implants tailored for metal-sensitive patients. For example, we commenced development of InSet 70, InSet 135 and InSet 185 stems to expand our I-Series humeral stem line in 2024. We have received FDA 510(k) clearance expanding I-Series humeral stem product line to include certain fracture indications and commercially launched the InSet 70 in September of 2025. In January 2026, we received FDA 510(k) clearance for products designed for patients with metal sensitivity. In December 2025, the Company announced the development of a robotics platform to design a transformative shoulder-specific micro-robotic solution designed to further enhance shoulder surgical precision, workflow efficiency, and enable exciting, new clinical approaches in the aTSA and rTSA markets. The robotic solution is designed to be integrated with the Company’s ProVoyance platform to deliver a comprehensive technology solution. We are also evaluating expansion into adjacent areas in shoulder surgical care, which may include sports medicine and shoulder trauma markets.
We have contributed to numerous publications that we believe evidence and strengthen our position as a leader in shoulder surgical care. There is a significant body of clinical evidence that supports the safety, efficacy, and durability of our implants in shoulder arthroplasty, including our InSet Glenoid technology. For example, a retrospective long-term follow-up analysis of patients who received our InSet Glenoid was published in the Journal of Shoulder and Elbow Surgery in 2019, which demonstrated a 72-point improvement in the mean American Shoulder and Elbow Surgeons (“ASES”) outcome score, statistically significant improvements in pain scores and range of motion, with no surgical complications, no cases of glenoid loosening and no revision surgeries performed at a mean follow-up time of 8.7 years. We are committed to continued investment in obtaining further clinical evidence with the support of surgeons who are recognized as thought leaders in shoulder surgical care. We believe these efforts will continue to generate a substantial body of clinical evidence that will drive increased awareness and adoption of our products.
Our Success Factors
We attribute our success to a combination of the following factors. We believe these attributes are central to our business outcomes and will be significant factors in our continued success and growth.
Disruptive Ecosystem to Address Existing Limitations Within Shoulder Surgical Care
Our purpose-built ecosystem is designed to directly address several of the limitations associated with shoulder surgical care, starting with shoulder arthroplasty. This ecosystem is comprised of our advanced implant systems for aTSA and rTSA, ProVoyance, efficient instrument system, specialized support and surgeon-to-surgeon collaboration. Our advanced implant systems are designed to address existing challenges, such as glenoid loosening, subscapularis tendon failure, limited range of motion, stability and eventual implant failure. Glenoid loosening is a leading cause of revision shoulder arthroplasty procedures, with studies showing that approximately 30% of implants exhibit moderate to severe loosening
8
within 6.6 years and up to 40% require revision within ten years. Unlike traditional glenoid designs, our InSet Glenoid sits within a rim of the native bone, which we believe enhances stability and reduces micromotion that contributes to loosening and potential failure. Complementing our InSet Glenoid technology, our humeral stem technology is fully compatible across both aTSA and rTSA procedures using consistent surgical techniques and instrumentation. This design facilitates interoperability and is intended to enable simplified revision and conversion procedures, which we believe provides surgeons flexibility, preserves bone mass and reduces the procedural burden associated with switching implant systems during a procedure. Surgeons leverage ProVoyance to create bespoke surgical plans that can help ensure consistent, effective positioning of our implants. Additionally, our design has allowed us to consolidate all of the instruments necessary to complete either an aTSA or a rTSA into two convenient trays, which can reduce operational complexity. We complement our offerings with specialist support and by facilitating surgeon-to-surgeon collaboration. We believe the elements of our ecosystem work together to help surgeons reduce operational complexity and surgical time, while supporting improved patient outcomes, lower revision risk and improved long-term satisfaction. We believe our differentiated design and approach positions us as a leader in addressing the critical clinical issues facing shoulder arthroplasty, as evidenced by our commercial traction to date. Since the initial launch of our InSet Glenoid in 2016, our implants have been utilized in more than 17,500 procedures.
Strong Clinical Results and Positive Outcomes for Patients and Surgeons
Our InSet Glenoid technology is supported by a compelling body of clinical evidence, with approximately nine years of published data and four clinical studies and articles published in the Journal of Shoulder and Elbow Surgery demonstrating its significant impact on patient outcomes. These studies demonstrate that, following surgery using our InSet Glenoid, patients have experienced greater functionality, lower pain and higher overall satisfaction than before surgery. Clinical data has also demonstrated strong performance in key indicators, such as forward flexion, rotational movement, range of motion, pain reduction and implant durability. Notably, a retrospective long-term follow-up analysis of patients who received our InSet Glenoid was published in the Journal of Shoulder and Elbow Surgery in 2019, which demonstrated a 72-point improvement in the mean ASES outcome score as compared to before surgery, reflecting a meaningful improvement in function (range and strength) and patient well-being following surgery. In addition, no surgical complications, cases of glenoid loosening or revision surgeries were reported at a mean follow-up time of 8.7 years. In a finite element analysis published in the Journal of Shoulder and Elbow Surgery in 2012, our InSet Glenoid demonstrated an 87% reduction in “rocking horse” motion, which is a widely known key contributor to implant loosening and failure. We have also focused on optimizing rTSA biomechanics by aiming for a more anatomic feel and aesthetic, preserving native bone and replicating the anatomic humeral positioning, which can contribute to improved postoperative range of motion and function. We believe the biomechanics of our reverse offerings are an important differentiator of our technology. Our improved biomechanics are driven by our proprietary InSet design, which was engineered to create a more anatomic relationship between the glenoid and humeral stem to enhance range of motion. We believe the existing published data and patient outcomes, coupled with growing clinical validation and positive physician feedback, support the long-term adoption and utilization of our products within the shoulder surgical care market.
Well Positioned as Shoulder Surgical Care Market Grows in Outpatient Settings
Shoulder procedures can be performed in multiple care settings, including at hospitals, outpatient care centers and ASCs. Over the past three decades, there has been significant growth in outpatient care, driven by lower overall costs and increased procedural efficiency. We believe we are well positioned to succeed in these outpatient settings, particularly ASCs, because our purpose-built ecosystem can facilitate reproducible procedures with low complication rates, intraoperative flexibility, simplified workflows and efficient use of operating room space. We believe these attributes are particularly relevant in the ASC setting, where surgeons commonly face more limited resources (cost constraints and limited space for instrumentation) and financial risk if a surgery requires escalation. In addition to procedural and clinical reliability, ASCs generally prioritize or require cost-efficient, streamlined solutions. Our efficient instrument system supports both aTSA and rTSA procedures from start to finish with just two convenient trays, considerably less than the six to nine trays typically required by other offerings. This two tray instrument system can reduce operational complexity and lower costs by, for example, increasing room turnover and surgical throughput and minimizing storage and handling, ultimately helping ASCs to treat more patients with fewer resources.
Proven and Experienced Management Team
Our proven and experienced management team collectively has decades of experience in orthopedic product development and commercialization. Members of our leadership team have held senior and executive roles at some of the most recognized companies in the medical technology industry, including publicly traded companies, and have track records of delivering strong growth and results. We are an innovation-centric team, having played instrumental roles in the
9
development and commercialization of foundational technologies in the shoulder space, including rTSA, stemless implants and advanced preoperative planning technologies. Our commitment to innovation is further evidenced by our combined 250+ orthopedic patents developed over the course of our management team’s careers, reflecting deep clinical insight, technical expertise and a nuanced understanding of the orthopedic market. As a result, we are able to leverage deep clinical insight, technical expertise and an informed understanding as to the limitations of existing technologies in the development of next-generation solutions. In addition, through decades of thought leadership in the space, our team has established strong relationships with key opinion leaders and surgeons. We believe the history and experience of our management team position us to effectively execute on our strategic objectives.
Unique Commercial Organization Dedicated to the Shoulder Surgical Care Market
Shoulder surgeries are some of the most complex procedures in the orthopedic space due to the joint’s complex range of motion, surrounding soft tissue structures and difficult surgical exposure. This complexity requires a high degree of surgical precision, clinical support, depth of expertise and specialization. We view ourselves as specialists serving specialists and are exclusively focused on shoulder care, which we believe represents a level of expertise that is essential for addressing the unique challenges of these procedures. Our commercial organization is comprised of a dedicated commercial leadership team that drives our internal commercial efforts, our CEME team, and our network of independent distributors. As of December 31, 2025, we had 45 shoulder specialists across our dedicated commercial leadership team and CEME team, representing more than 750 years of combined direct selling, physician engagement and clinical education experience. These specialists collaborate closely with our distributors to direct our sales activities and specialized support services, including hands-on support for surgeons—from procedure planning to execution—and help to facilitate surgeon training and performance management. In addition, our network of independent distributors allows for prospecting, relationship management and case coverage. These three key components of our commercial organization work in tandem to form a commercial flywheel that is designed to build and provide key product support to surgeons and other stakeholders in the shoulder surgical care market. We estimate that there are approximately 15,000 surgeons in the United States who perform at least one shoulder arthroplasty procedure per year, of which we target the approximately 1,800 high-volume surgeons who perform the vast majority of procedures. We believe these high-volume surgeons require dedicated shoulder expertise and support. Our entrenched relationships with many high-volume surgeons, together with engagement through highly tailored support, ongoing medical education and third-party symposiums, lab events, and surgeon-to-surgeon activities, all contribute to fuel our strategy and reinforce our position as a leader in the shoulder surgical care market.
AI-Enabled Business and Clinical Intelligence Technologies
We leverage AI-enabled technology solutions to support our business and surgeons. At the center of our commercial strategy is our internal business intelligence platform, which draws insights from our proprietary database to help us target the right stakeholders in the shoulder surgical care market. We also offer similar tools to physician practices to help them identify and acquire new patients through proprietary search engine and funnel optimization strategies. By engaging with surgeons early in the care process and supporting patient acquisition, we become deeply integrated across the surgical workflow. In addition, our proprietary AI powered preoperative planning technology, ProVoyance, is designed to enable consistent, effective positioning of our advanced implant systems. ProVoyance is cleared by the FDA for preoperative shoulder planning, and is listed on the FDA’s AI/ML-enabled medical devices list. We believe the benefits of our preoperative planning technology are evidenced by the fact that, for the year ended December 31, 2025, to the extent we are able to measure, virtually all surgeries using our implants use ProVoyance, making it a routine part of surgeon workflow. We also maintain a patient registry that allows practices to monitor outcomes and engage with patients postoperatively. Together, these tools are designed to provide information to enable surgeons to improve care delivery, optimize practice performance and achieve better outcomes. We believe these technologies play an important role in solidifying us as a differentiated, data-enabled leader in shoulder surgical care.
Our Growth Strategies
Our goal is to leverage our purpose-built ecosystem to become the leader for shoulder surgical care. The key elements of our growth strategy include:
Increase Awareness of our Purpose-Built, Innovative Shoulder Surgical Care Ecosystem to Continue Taking Share Across Care Settings
We plan to center our marketing efforts around educating surgeons about our ecosystem’s advantages across all care settings, including hospitals, outpatient care centers and ASCs. Our ecosystem has been thoughtfully developed from the ground up to address the limitations associated with current offerings and help surgeons and patients access consistent
10
quality care regardless of where it is delivered. We believe that by focusing on increasing awareness of our holistic, innovation-driven approach, we can drive broader adoption of our ecosystem. We believe our ecosystem provides key advantages that surgeons seek in shoulder arthroplasty solutions, including improved fixation and stability, restoration of full functionality, exceptional longevity, improved implant selection and positioning, optimized procedural workflow and efficiency and access to expert advice. The output of our innovation-first and patient-centric approach has resonated with surgeons, as demonstrated by our 50% increase in procedure volume from 2024 to 2025. As the shoulder surgical care market continues to grow, we believe increased awareness of our ecosystem’s advantages will position us to continue capturing market share from less specialized offerings.
Expand our Commercial Flywheel of Shoulder Specialists, Surgeon-to-Surgeon Collaboration, and Network of Independent Distributors
Our commercial flywheel includes our dedicated commercial leadership team, CEME team and network of independent distributors. We believe our dedication to shoulder surgical care is a strategic differentiator that enables us to deliver a highly tailored experience to surgeons operating in what is a complex and technically demanding procedure category. We intend to continue scaling our commercial organization with a continued specialized focus on shoulder care. By increasing the size of our dedicated commercial leadership team, we believe we can enhance targeting and prospecting efficiency, in an effort to ensure we engage the highest-value surgeon customers. Likewise, we believe growing our CEME team will allow us to increase the frequency and reach of our educational initiatives, such as symposiums, lab events, and surgeon-to-surgeon training programs. This increased presence will aim to continue to foster and grow our collaborative network of expert surgeon educators who can serve as powerful voices regarding the safe and effective use of our products and appropriate disease states and surgical techniques, which in turn allows broader adoption of our products through surgeon-to-surgeon training and peer education. Finally, by expanding our network of independent distributors, we will strive to ensure the quality of our customer service and execution remains up to our current standard as we scale. We believe our commercial flywheel and exclusive focus on shoulder care allow us to convert high-potential accounts and nurture new surgeons into loyal users of our ecosystem. Through expansion of this integrated and specialized commercial approach, we aim to accelerate growth, drive surgeon engagement, and expand adoption of our differentiated shoulder surgical care solutions in a cost-efficient manner.
Capitalize on Our Unique Advantages to Capture Outsized ASC Growth
We intend to expand our presence in the ASC setting through a multi-pronged approach that aims to capitalize on the outsized growth of ASCs as key sites for shoulder surgical care. We will drive targeted sales initiatives to increase awareness of our unique advantages in the ASC setting, including the potential efficiency and economic benefits of our ecosystem. We will also leverage surgeon-to-surgeon collaboration to reinforce these advantages with real-world experiences and will continue to generate clinical and economic validation of our ecosystem’s benefits in the ASC. In addition, we intend to continue innovating our systems with the ASC setting in mind. As ASC procedure volume grows, we believe we will be able to drive meaningful growth and establish ourselves as the leader for shoulder arthroplasty in the ASC setting.
Increase our Addressable Market Through our Commitment to Continuous Innovation and Advancing Shoulder Surgical Care
We have a strong track record of innovation and product commercialization, having successfully launched a wide range of new technologies to address unmet needs in the shoulder surgical care market. We believe this track record demonstrates our ability to identify clinical needs and bring differentiated solutions to market. We plan to continue investing in our robust product pipeline to expand our capabilities and service additional segments of the shoulder surgical care market. Our near-term development efforts in shoulder arthroplasty include a fracture-specific system, a revision solution, and implants tailored for metal-sensitive patients. Additionally, we intend to build on our existing aTSA and rTSA systems by introducing product line extensions that support continuous improvement. We are also evaluating expansion into adjacent areas in shoulder surgical care, which may include sports medicine and shoulder trauma markets. By leveraging our innovation capabilities and deep customer relationships, we believe we can expand our addressable market and generate incremental sources of revenue through targeted product development. We may also evaluate entering other markets through business development opportunities.
Continue Building and Driving Marketing of our Technology Solutions
We view digital enablement as an essential component of modern shoulder surgical care and believe that our technologies can drive greater clinical value and deepen physician engagement. We see four key aspects to the growth opportunity for our technologies. First, we can continue to leverage our internal business intelligence platform to help us
11
efficiently target high-volume surgeon prospects. Second, we can strategically deploy our enabling technologies to connect patients to those physicians. Third, ProVoyance preoperative planning technology can increase surgeon loyalty by helping to improve workflows and drive positive clinical outcomes. Fourth, we can offer surgeons a platform for outcomes measurement to assess performance. We expect our underlying algorithms and predictive capabilities to improve as we drive increased adoption of our technologies and can leverage additional data inputs, which we believe will reinforce a virtuous cycle of increased accuracy, better outcomes and greater reliance on our tools. Additionally, we intend to continue investing in and expanding our technology capabilities and we are actively developing other complementary enabling technologies to support the full continuum of care in shoulder surgery. We believe these innovations will further establish our position as a comprehensive, technology-driven solution provider within the shoulder surgical care market.
Pursue Expansion in International Markets
While our primary commercial focus to date has been within the United States, we believe the international shoulder surgical care market represents a compelling opportunity for long-term growth. Based on third-party industry reports and our internal estimates, we estimate the annual international shoulder arthroplasty market opportunity outside the United States to be approximately $1.0 billion, driven by many of the same factors we see domestically. We plan to strategically pursue entry into certain international markets over time.
Market Overview
Overview of Shoulder Anatomy
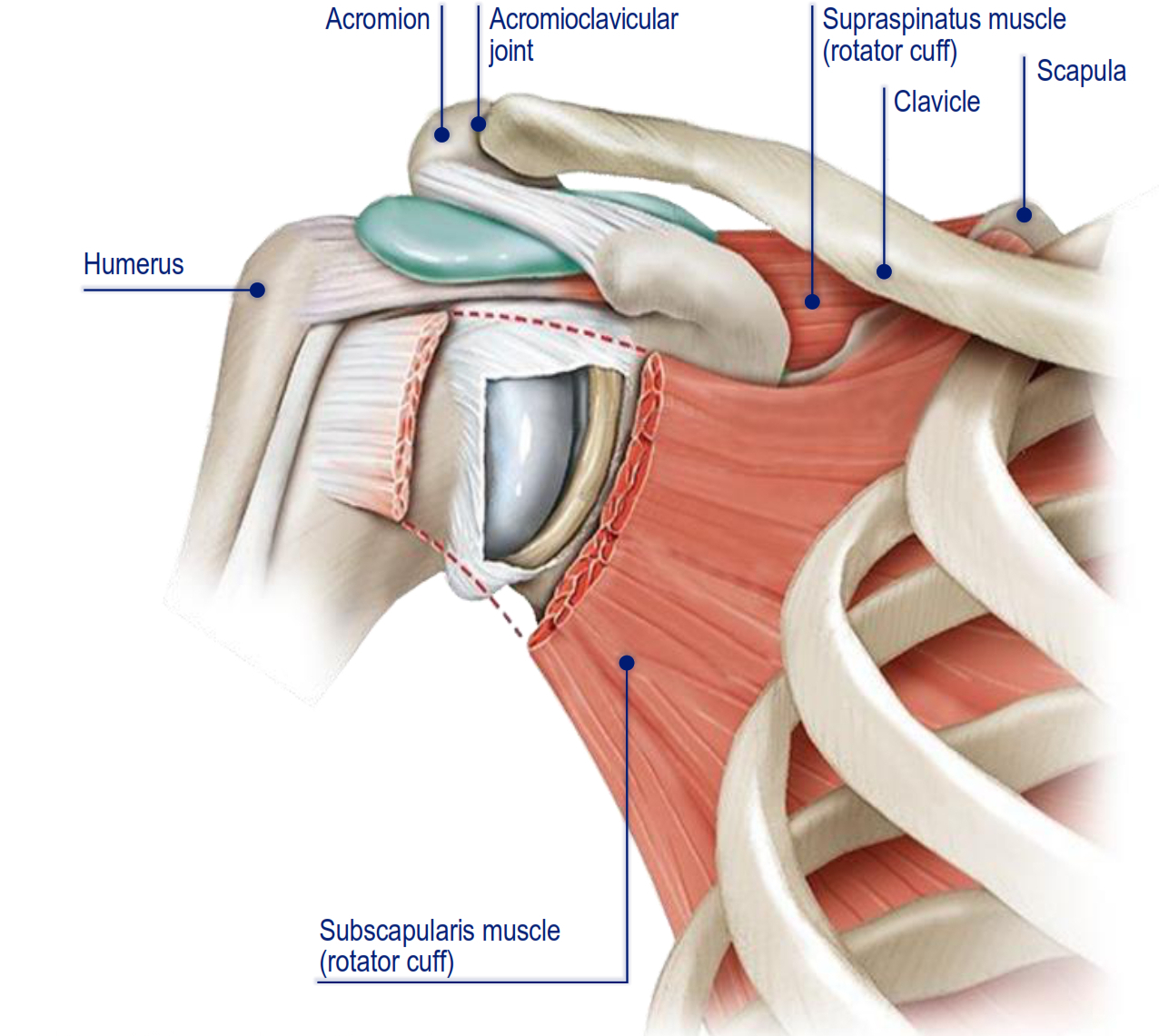
The shoulder is a distinct anatomical structure comprised of bones, muscles, tendons, and ligaments that work in tandem to allow for a complex range of motion for the arm. The key bones of the shoulder include the humerus (upper arm bone), scapula (shoulder blade), and clavicle (collarbone). The humerus articulates with the glenoid fossa of the scapula to form the glenohumeral joint, while the acromion (scapular projection) and clavicle form the acromioclavicular joint. The rotator cuff includes four muscles and their tendons (infraspinatus, subscapularis, supraspinatus, and teres minor) that serve to stabilize the head of the humerus in the glenoid.
The illustration below depicts the key anatomy of the shoulder (anterior view):
12
Overview of Conditions Impacting the Shoulder
Shoulder pain is highly prevalent and can significantly impact and reduce quality of life as well as result in chronic pain. According to a study published in the Journal of Shoulder and Elbow Surgery, an estimated 38% of patients with shoulder pain reported an inability to perform activities of daily life. Furthermore, a study published in the American Journal of Sports Medicine estimated 89% of patients with rotator cuff tears had difficulty sleeping. Today, approximately one in five adults in the United States over the age of 65 experience shoulder pain.
The primary conditions that can result in shoulder pain and reduced functionality include:
•Osteoarthritis (“OA”): OA is a degenerative joint disease characterized by the breakdown of cartilage and underlying bone, leading to pain, stiffness, and reduced mobility in the shoulder’s glenohumeral joint. This condition results from wear-and-tear, aging, or prior injury, progressively worsening over time. OA is highly prevalent, affecting approximately one in three adults in the United States over the age of 60.
•Rheumatoid Arthritis (“RA”): RA is a chronic autoimmune disease characterized by synovial inflammation as well as bone and cartilage destruction and deformity. RA typically impacts the large joints more than peripheral joints in later stages of the disease leading to progressive damage in the shoulder. According to the Centers for Disease Control and Prevention, RA impacts over 1.5 million adults in the United States, with various studies estimating incidence in the shoulder as high as 90%.
•Shoulder Fractures: Acute and traumatic injuries may result in severe fractures of the humerus or other adjacent bones in the shoulder. Such incidents are most prevalent among elderly patients and those with chronic conditions such as OA.
•Rotator Cuff Tears: Rotator cuff tears refer to partial or full-thickness disruptions of the tendons stabilizing the shoulder joint. Rotator cuff tears can be caused by acute trauma, repetitive stress, or degeneration. Rotator cuff tears are highly prevalent and impact nearly 25% of adults over the age of 40 in the United States.
These shoulder conditions are widespread, often debilitating, and are commonly experienced concurrently as interrelated musculoskeletal disorders. Based on publicly available industry data, including from the National Institutes of Health, we estimate that these conditions result in more than eight million physician visits annually in the United States. Despite this prevalence, we believe there has been a historical underutilization of surgical treatments for shoulder care due to several factors including patient hesitation to pursue surgical intervention, insufficient technology to appropriately treat shoulder conditions, perceived difficulty of surgical intervention, and barriers to patient access of care.
Our Addressable Market Opportunity in Shoulder Surgical Care
We believe the shoulder surgical care market today presents a significant market opportunity. Our initial focus within this broader market is on shoulder arthroplasty, and we believe we have an opportunity to expand to address adjacent areas in the shoulder surgical care market over time.
We currently serve the large, immediately addressable and rapidly growing market for shoulder arthroplasty procedures. We estimate that approximately 250,000 shoulder arthroplasty procedures were performed in the United States in 2025, which we believe represents an approximately $1.7 billion market opportunity based on our average sales price. Based on our internal estimates, knowledge of our industry and third-party data regarding the number of shoulder arthroplasty procedures performed, we expect this market to grow by approximately 11% annually through 2029. While our current commercial focus is on the United States, we plan to pursue market access initiatives in other attractive, high-growth international markets. We believe a significant opportunity exists outside of the United States and, based on third-party industry reports and our internal estimates, we estimate that the total international shoulder arthroplasty market is approximately $1.0 billion in 2025. Together, we believe these markets represent a global annual addressable market of approximately $2.7 billion.
The number of shoulder arthroplasty procedures in the United States grew at approximately 10% per year from 2019 to 2024, reflecting one of the fastest-growing segments within orthopedic reconstruction since other sectors are growing in low single digits. This growth was primarily driven by multiple, ongoing demographic factors and industry tailwinds. For example, the population in the United States is both aging and remaining more active later in life, which has contributed to a higher incidence of degenerative shoulder conditions. Additionally, greater awareness of treatment options, earlier diagnosis and broader acceptance of shoulder arthroplasty as an interventional solution have expanded the pool of potential
13
patients. In parallel, there has been continued growth of outpatient and ASC settings, which has further increased patient access to care and care settings where surgeons can perform these procedures.
We believe the growth in shoulder arthroplasty procedures will continue to be driven by existing tailwinds. Further, we believe the number of patients who may potentially benefit from shoulder arthroplasty is significantly higher than the number of patients who currently seek treatment today. Over time, as access to care increases and clinical outcomes improve as a result of better technology, we believe the proportion of patients seeking shoulder arthroplasty will eventually approach that of the knee arthroplasty market.
Overview of Shoulder Arthroplasty
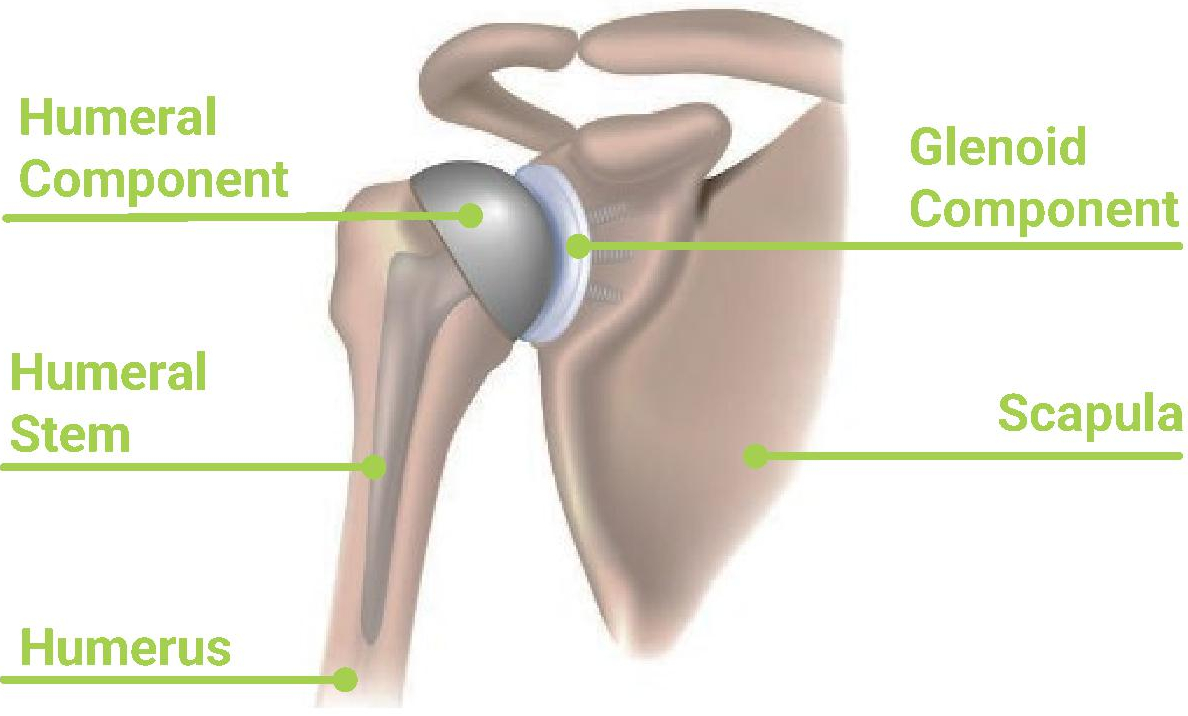
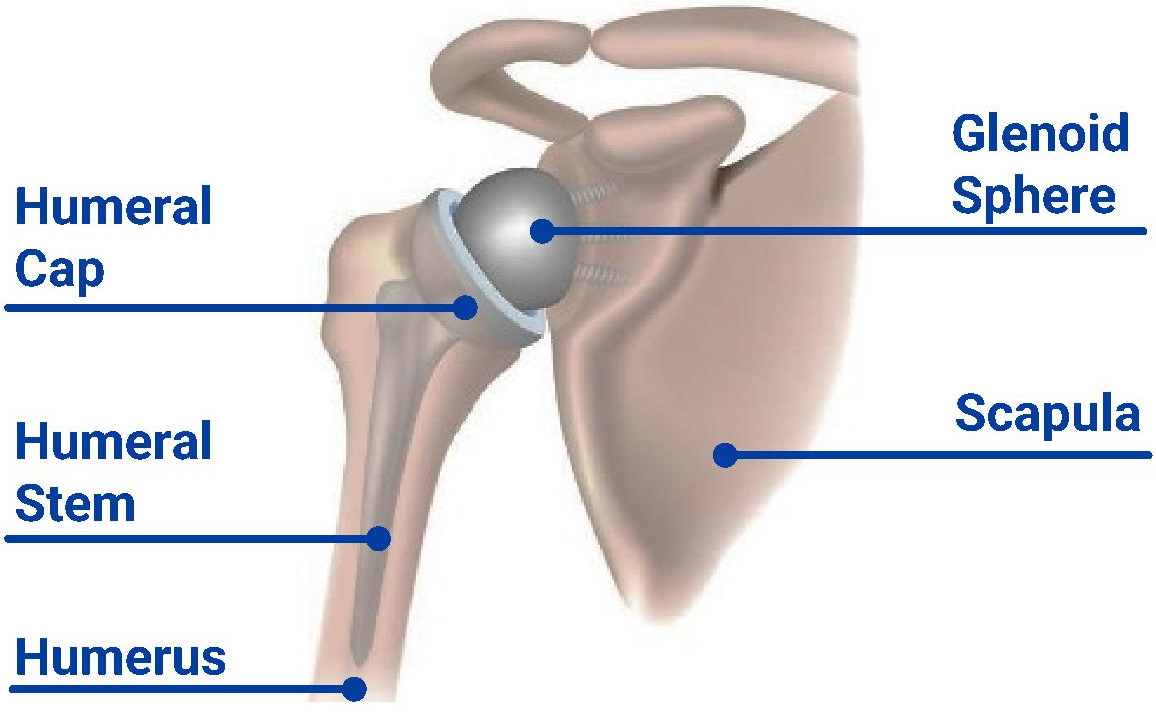
Shoulder arthroplasty is an established surgical procedure involving a reconstruction of the shoulder joint with prosthetic implants. Arthroplasty involves the resection of surfaces around the glenoid fossa and humeral head followed by the implantation of prostheses that articulate with each other. Typically, an arthroplasty procedure includes the implantation of two devices, one “glenoid component” which attaches to the scapula and one “humeral component” which attaches to the humerus. Overall success of these procedures is measured by reduced pain, improved function, fewer post-operative complications, and increased survivorship of the implant.
In all shoulder arthroplasty procedures, appropriate biomechanical fit of the protheses is critical to ensure precise alignment with the patient’s natural shoulder anatomy to optimize stability, range of motion, and long-term functionality. Appropriate device type, size, positioning and fixation are all essential to ensuring biomechanical fit and procedure success. ASES scores, which are derived from a postoperative questionnaire used by surgeons to assess shoulder function and patient well-being following surgeries, are commonly used to measure operative success. Today, there are two approaches to shoulder arthroplasty, aTSA and rTSA:
Anatomic Total Shoulder Arthroplasty (“aTSA”) — aTSA imitates the natural joint anatomy by using prosthetic implant components to replace the humeral head with a prosthetic ball and stem and the glenoid with a prosthetic cup. aTSA is most often used in OA and RA patients with arthritic shoulder joints and intact rotator cuffs, using the rotator cuff to stabilize the joint and dictate motion. While aTSA procedures may reduce pain and restore function, complications such as subscapularis tendon failure and glenoid loosening have been relatively common with aTSA procedures.
Reverse Total Shoulder Arthroplasty (“rTSA”) — rTSA inverts the shoulder anatomy and joint configuration, reversing the ball and socket positions. rTSA transforms the humeral head into a socket and the scapula into a ball, shifting the reliance from the rotator cuff to the deltoid muscle for stability and motion. rTSA addresses a broader range of conditions, including rotator cuff arthropathy, irreparable rotator cuff tears, glenoid bone loss, acute fractures, and post-traumatic reconstruction. The development of the rTSA procedure has allowed shoulder arthroplasty to be used to address additional shoulder issues, including rotator cuff damage. While rTSA procedures have corrected some of the limitations of aTSA, historically there has been a tradeoff with patients often sacrificing shoulder motion and movement patterns in rTSA.
Anatomical Replicates the normal mechanics and anatomy of the shoulder, replacing the ball and socket as they are normally found in the body with prosthetic implants. | Reverse “Reverses” the ball and socket placement, transforming the humeral head into a socket and the scapula into a ball. | ||||
 |  | ||||
14
Market Dynamics in Shoulder Arthroplasty
Shoulder arthroplasty can be performed both as an inpatient hospital procedure and in outpatient settings, including ASCs. In recent years, as the volume of shoulder arthroplasty procedures has grown, the majority of the incremental procedures have been performed at ASCs. This trend is similar to what has occurred in the hip and knee joint replacement markets and tracks the broader long-term growth of surgical procedures shifting to outpatient settings. ASCs have emerged as a low-cost site of care and positive post-care outcomes. We expect that future growth in the market will be significantly driven by ASCs as hospitals face capacity constraints and are more limited in their ability to meet increasing demand. In 2024, CMS added total shoulder arthroplasty to the list of covered procedures that can be performed in ASCs, which facilitated reimbursement and further supported the growth of ASCs as a key site of care in the shoulder surgical care market. This growth within ASCs also presents other benefits for key stakeholders, including streamlined workflows, scheduling flexibility and operational autonomy. Further, surgeons often have economic ownership of the ASCs where they operate, with as many as 80% of ASCs owned at least in part by operating surgeons according to a study published in Global Spine Journal, which we believe further incentivizes surgeons to utilize ASCs for shoulder arthroplasty procedures.
Shoulder arthroplasty volumes in the United States experienced a significant increase driven by clear and meaningful growth of the rTSA procedure. First approved in the United States in 2004, rTSA has expanded the market and now accounts for a significant majority of shoulder arthroplasty procedures due to their broad treatment applications, surgeon preference relative to aTSA, and advantages in clinical outcomes, such as reduced likelihood of revision surgeries. In 2020, approximately 70% of all shoulder arthroplasty procedures were rTSA procedures.
Limitations of Existing Product Offerings in Shoulder Arthroplasty
Despite their frequent use and wide adoption, we believe traditional implants used in shoulder arthroplasty procedures are hindered by several limitations, including poor biomechanical fit, suboptimal kinematics, difficult replacement and conversion procedures (aTSA to rTSA or stemless to stem), imprecise implant positioning due to limited surgical planning, inefficient and burdensome workflow designs and non-specialized case support. These shortcomings can impact both patients and surgeons and result in continued pain or discomfort, lack of mobility, postoperative complications, low rates of implant survivorship, necessity of revision surgeries and costly and inefficient procedures for healthcare providers.
We believe traditional shoulder arthroplasty products present several limitations, including:
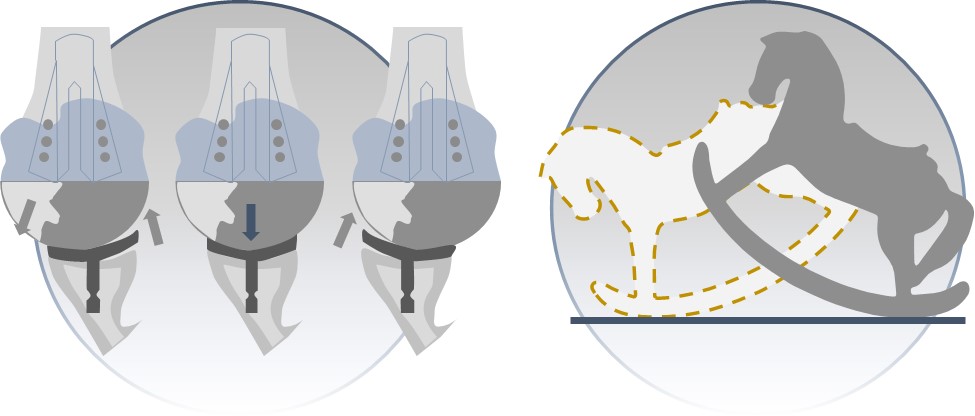
•Fixation of the Glenoid Component in aTSA: Due to poor fixation of the glenoid component in aTSA, normal movement from the humerus can also rock the glenoid loose, commonly referred to as the “rocking horse” effect, creating instability within the implant. The illustration below depicts the “rocking horse” effect resulting from an implant with poor fixation:
•Poor Product Design Resulting in Suboptimal Kinematics in rTSA: Traditional implants in rTSA utilize an onlay design, placed above the anatomic neck of the humerus on top of a bony surface, without directly penetrating the bone. This design often results in overstuffing whereby the implant excessively fills the joint space and alters the shoulder’s natural anatomy. This can increase pressure on surrounding tissues, restrict motion, and result in pain and discomfort. Furthermore, competitor implants that utilize an inlay design inserted directly into the bone often result in poor kinematics, restricting shoulder rotation and movement. These traditional implants may also modify the shoulder anatomy and move the humerus further away from the scapula than where it would naturally sit, resulting in poor aesthetic results where the shoulder does not regain its natural form post-procedure.
•Difficult to Replace and Convert: Traditional implant designs place the implant stem deep into the humerus, requiring the removal of the stem from the humerus for replacement and revision surgeries and for conversions to rTSAs. This design results in a high degree of bone removal and potential damage to the surrounding soft tissue region during such procedures, and significantly limits and complicates intraoperative flexibility and the ability of surgeons to switch from aTSA to rTSA. aTSA procedures are increasingly using a stemless humeral implant as a
15
bone-preserving alternative. These implants, however, are often not easily convertible to stemmed implants or in rTSA procedures, which restricts their utility.
•Imprecise Implant Positioning Due to Limited Surgical Planning: Shoulder arthroplasty procedures require precision and accuracy and clinical evidence demonstrates that positioning as little as five degrees off angle can lead to inferior patient outcomes. Surgeons have historically relied on basic imagery to inform their surgical approach, using tools that often lack 3D bone rendering or biomechanical simulation that do not fully capture patient-specific anatomy and properly simulate postoperative range of motion and joint stability. These existing market offerings have resulted in limited surgeon engagement as well as the outsourcing of imaging interpretation components and planning that surgeons might otherwise seek to perform themselves.
•Inefficient and Burdensome Surgical Workflow Design: Traditional implant systems have typically been developed and expanded product by product, indication by indication, over long periods of time. The complexity of these systems and the related equipment, including multiple trays of instruments, generally requires a wide range of surgical techniques and can considerably complicate surgical workflows across care settings. This legacy approach has led to an outsized amount of equipment in the operating room, leading to logistical challenges including an increased capital footprint, and extended procedure and operating room turnover time, thereby creating unnecessary costs to healthcare providers.
•Non-Specialized Case Support: Shoulder arthroplasty is a highly complex and technical procedure. Operating room staff and surgeons routinely rely on the product knowledge and know-how of orthopedic salespeople. Legacy shoulder implants are predominantly sold by non-specialized salespeople who also sell devices in other segments of orthopedics, such as hip and knee implants. The lack of specific product and technical knowledge in shoulder arthroplasty can result in suboptimal case support and patient outcomes.
These limitations of legacy shoulder arthroplasty products can result in the following shortcomings:
•Failure to Reduce Pain and Improve Shoulder Function: The key objectives of undertaking shoulder arthroplasty surgery – reduction in pain and increased shoulder functionality – are often not achieved post-surgery. For example, studies have shown that approximately 20% of shoulder arthroplasty patients remain in pain or discomfort beyond one to two years following surgery.
•Frequent Post-Operative Complications: As a result of poor fixation and suboptimal kinematics, traditional implants can lead to several complications including overstuffing, glenoid loosening, shoulder subluxation (dislocation), humeral fractures, and subscapularis tendon failure (a condition where the tendon connecting the subscapularis muscle to the humeral bone is damaged, which in one study published in the Journal of Shoulder and Elbow Surgery was demonstrated to occur in approximately 15% of aTSA procedures). These complications may result in further pain, reduced function for patients, and require additional intervention.
•Low Rates of Implant Survivorship: Glenoid implant loosening is frequent and common. According to a study published in the Journal of Shoulder and Elbow Surgery, approximately 30% of aTSA procedures demonstrated moderate to severe loosening at mean follow-up time of 6.6 years. The resulting pain and inflammation from excessive and continued loosening over time may require additional intervention. In a separate study of aTSA revision surgeries published in the same journal, approximately 70% of glenoid failures that resulted in revision surgery demonstrated glenoid loosening. In this study, approximately 20% demonstrated glenoid loosening within 5 years post procedure and approximately 25% demonstrated precursors to loosening within 5 years post procedure.
•Necessity of Revision Surgeries: Poor clinical outcomes and complications significantly increase the likelihood for a second surgery, often requiring a revision surgery to correct or replace a failed implant. Revision surgeries are common with studies showing up to 40% of glenoid implants were subject to revision surgery at 10-year follow-up. These repeat surgeries compromise patients’ health and quality of life and are an unnecessary burden and avoidable cost to the healthcare system, with clinical evidence demonstrating that complication rates in revision surgeries are significantly higher. According to a study published in the Journal of Shoulder and Elbow Surgery, overall complication rates were 69% in revision rTSA, significantly higher than the 25% complication rate for initial rTSA procedures.
•Costly and Inefficient for Healthcare Providers: Current instrumentation processes require up to nine trays for a single shoulder arthroplasty procedure. Each tray requires significant storage space and must be sterilized before and after every use resulting in higher costs and burdening staff. This inefficient workflow has an outsized impact
16
on outpatient care centers and ASCs given the inherent resource and space constraints in these settings. We believe that requiring more time and space in the operating room for maintaining surgery trays and using large quantities of unfamiliar equipment increase the risk of infection and impact patient safety.
Additional Opportunities
We plan to continue investing in our robust product pipeline to expand our capabilities in shoulder arthroplasty and serve additional, adjacent segments of the shoulder surgical care market that we do not currently address. Our near-term development efforts include a revision solution to address the $85 million annual shoulder revision market in the United States (which, based on our estimates, represents approximately 5% of the estimated $1.7 billion annual shoulder arthroplasty market in the United States). In December 2025, the Company announced the development of a robotics platform to design a transformative shoulder-specific micro-robotic solution designed to further enhance shoulder surgical precision, workflow efficiency, and enable exciting, new clinical approaches in the aTSA and rTSA markets. The robotic solution is designed to be integrated with the Company’s ProVoyance platform to deliver a comprehensive technology solution. In addition to these near-term development efforts, we are also evaluating expansion into adjacent areas in shoulder surgical care, which may include sports medicine and shoulder trauma markets.
Our Solutions
Our Approach to Innovation
We are building Shoulder Innovations with the goal of addressing some of the most pressing challenges to patient outcomes in shoulder surgical care: poor biomechanical fit, suboptimal kinematics, difficult replacement and conversion procedures (aTSA to rTSA or stemless to stem), imprecise implant positioning, inefficient and burdensome workflow designs, and limitations of a generalist approach. Our innovation-first mentality centers on the patient, realizing that each stakeholder in the care chain is motivated by and benefits from focused and improved patient care. We seek to improve core components of shoulder surgical care — preoperative planning, implant design and procedural efficiency — in an effort to benefit each stakeholder in the care chain. We believe our approach has positioned us to drive long-term success in the shoulder surgical care market.
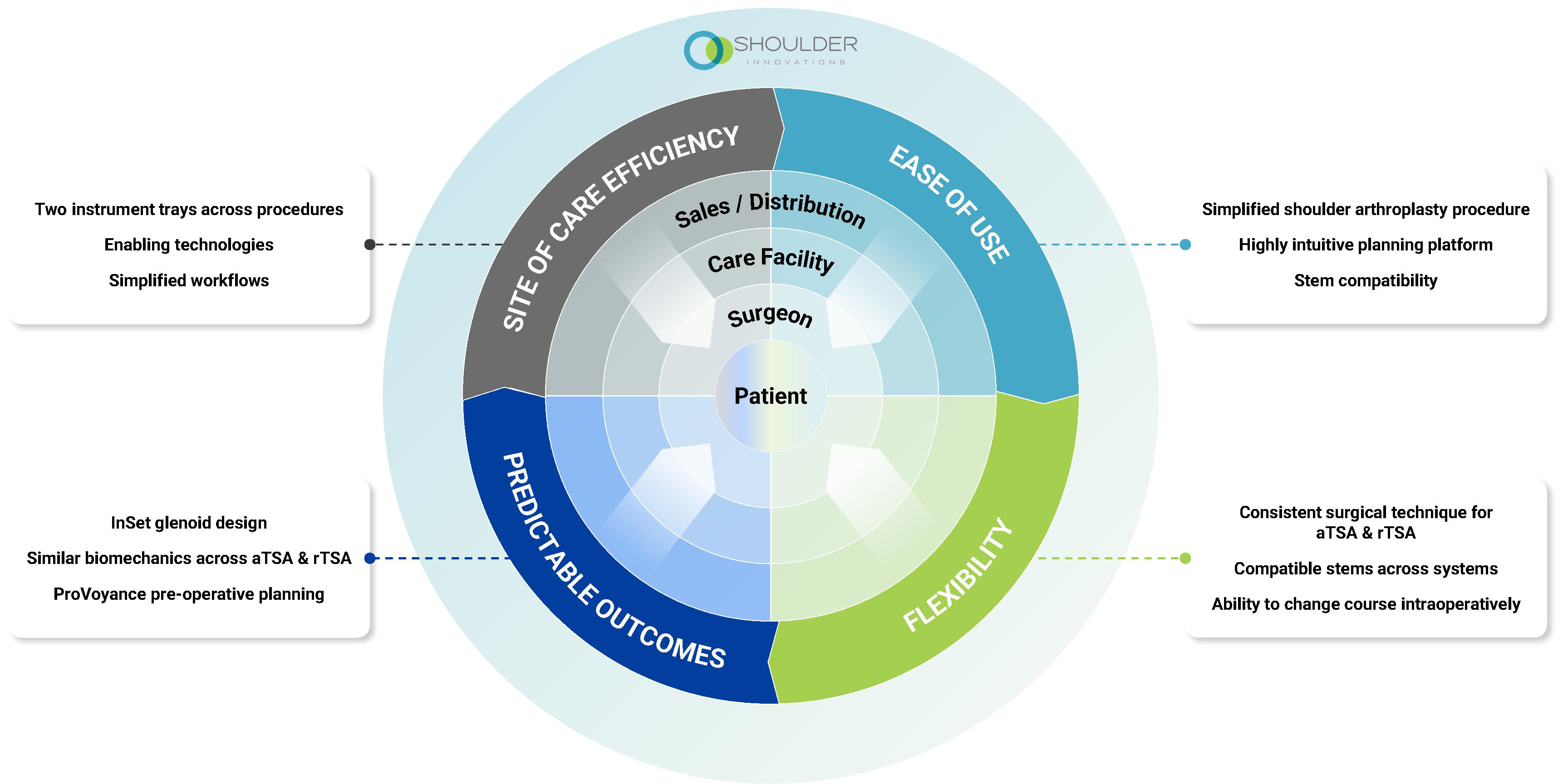
We believe that the following attributes are critical to win in this market:
•Ease of Use: We are simplifying the shoulder arthroplasty procedure to help surgeons achieve reproducible results with confidence and ease.
•Flexibility: We are developing implant systems to enable surgeons to perform both aTSA and rTSA procedures with consistent surgical techniques and instrumentation, with interchangeable systems.
•Predictable Outcomes: We are leveraging a leading AI-enabled technology to analyze patient anatomy and preoperatively plan surgeries that, together with thoughtful implant design, can lead to improved implant positioning and outcomes.
•Site of Care Efficiency: We are engineering our offerings to optimize workflow efficiency across care settings.
The image below depicts our approach to innovation and key attributes for success in the shoulder surgical care market:
17
Key Elements of Our Ecosystem
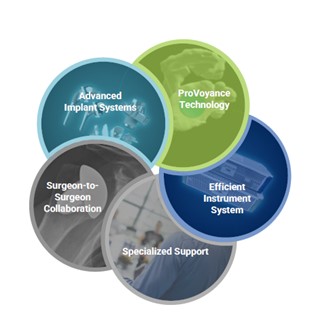
Our ecosystem is comprised of the following key elements:
•Advanced Implant Systems: Our advanced implants include a diverse range of interchangeable InSet aTSA and rTSA systems that leverage our novel, InSet Glenoid and InSet humeral stem technologies.
•ProVoyance Preoperative Planning Technology: This preoperative planning technology integrates AI and ML to transform planar CT imaging into 3D renderings, allowing surgeons to create bespoke surgical plans considering patient-specific anatomy ahead of surgery.
•Efficient Instrument System: Our efficient instrument system supports both aTSA and rTSA procedures from start to finish with just two convenient trays.
•Specialized Support: Our team of dedicated shoulder specialists enable us to deliver a highly tailored experience to surgeons operating in a complex and technically demanding procedure category.
•Surgeon-to-Surgeon Collaboration: Our CEME team fosters a collaborative network of expert surgeon educators and promotes surgeon-to-surgeon training and peer education.
The image below depicts the key elements of our ecosystem:
18
Key Benefits of Our Ecosystem
Our ecosystem offers notable benefits that differentiate it within the shoulder surgical care market. These key benefits include:
•Improved Fixation and Stability: Our foundational InSet Glenoid technology is biomechanically designed to specifically address what we believe is the primary problem in legacy aTSA implants: glenoid loosening. Our design aims to reduce mechanical stress at the bone-implant interface, improve fixation mechanics, enhance stability and reduce micromotion. These biomechanical advantages are evidenced by a published finite element analysis in which our InSet Glenoid technology demonstrated an 87% reduction in “rocking horse” motion, which is a widely known key contributor to implant loosening and failure. Notably, a separate published retrospective long-term follow-up analysis of patients who received our InSet Glenoid demonstrated a 72-point improvement in the mean ASES outcome score as compared to before surgery, reflecting a meaningful improvement in function (range and strength) and patient well-being following surgery.
•Restores Full Functionality: We engineered a range of humeral stem options to provide consistent, optimized biomechanics in both aTSA and rTSA. Using our InSet approach for rTSA, our system is designed with the goal of enabling patients to regain full range of motion, including the ability to raise their arm and reach behind (i.e., forward elevation and internal rotation), and avoid lengthening and overstuffing. We believe our design supports improved postoperative functionality and results in a postoperative feel and aesthetic that more closely tracks the shoulder’s natural form.
•Exceptional Longevity: Our advanced implant systems are designed for longevity, with minimal need for replacement. A published retrospective long-term follow-up analysis of patients who received our InSet Glenoid demonstrated no surgical complications, cases of glenoid loosening or revision surgeries at a mean follow-up time of 8.7 years in a particularly challenging patient population. We believe this durability provides surgeons and patients with confidence in our long-term implant performance and clinical outcomes.
•Improved Implant Selection and Positioning: ProVoyance preoperative planning technology empowers surgeons to create bespoke surgical plans that facilitate consistent, effective positioning of our implants. The highly intuitive interface enables surgeon planning without the need for third-party intervention and we believe that surgeon-level engagement in preoperative planning provides for better care for patients.
•Optimized Procedural Workflow and Efficiency: Our efficient instrument system supports our implant portfolio across both aTSA and rTSA. This can provide significant workflow advantages for our customers by minimizing the operating room footprint, reducing procedural setup time, lowering sterilization requirements and costs and reducing the risk of errors. This streamlined approach is enabled by leveraging our InSet Glenoid and
19
humeral stem technologies across our aTSA and rTSA systems, minimizing the number of sets and instruments required. Our advanced implant systems leverage consistent surgical techniques, which we believe can improve procedural outcomes while allowing for intraoperative flexibility and seamless interchangeability between aTSA and rTSA procedures.
•Access to Expert Advice: Our commercial organization creates deep relationships with surgeons and allows us to support their practice with specialized customer service and case support before, during and after surgery. In addition to direct support, our commercial team connects surgeons to the broader shoulder surgical community in various settings with a goal to improve connectivity across the shoulder surgical community and contribute to better patient outcomes.
Our Implants Systems
We developed a comprehensive implant portfolio to address the unique needs of patients and surgeons. Our advanced implants are comprised of our aTSA and rTSA systems, which include various, specifically designed components capable of a wide array of system configurations to facilitate different modes of operation (anatomic or reverse) that are optimized for patient-specific needs. These systems leverage consistent surgical techniques and the same efficient, two tray instrumentation system. The design of our advanced implant systems helps to facilitate shoulder surgical care with significant operational flexibility and reduced equipment and operating room footprint requirements.
InSet aTSA System
Our aTSA configuration consists of two main components, a glenoid scapular implant and a humeral fixation device, or stem. Each of these components was developed with a view to the overall implant system in an effort to provide maximum benefits to patients and flexibility for surgeons. A retrospective long-term follow-up analysis of patients who underwent aTSA procedures with our InSet Glenoid was published in the Journal of Shoulder and Elbow Surgery in 2019, which demonstrated a 72-point improvement in the mean ASES outcome score, statistically significant improvements in pain scores and range of motion, with no surgical complications, no cases of glenoid loosening and no revision surgeries performed at a mean follow-up time of 8.7 years.
InSet Glenoid
Unlike traditional glenoid implants that utilize an onlay design, our InSet Glenoid technology features a design that positions the implant within a pocket of sclerotic or cortical bone and aims to reduce mechanical stress at the bone-implant interface, improve fixation mechanics, enhance stability and reduce micromotion, all of which are key contributors to glenoid loosening. This design also enables a less invasive surgical approach that reduces surgical steps and difficulty of tissue exposure. Our InSet Glenoid implants were developed with a patented complex articular surface that contributes to reduction of the rocking horse motion. The InSet Glenoid design also enables a significantly reduced central fixation peg to provide further surgical flexibility.
The InSet PLUS Augmented Glenoid was designed to treat advanced shoulder conditions in patients with severe bone loss. The design of the InSet PLUS Augmented Glenoid corrects defects on the articular surface (in contrast to the fixation side of the implant) with five or ten-degree options. This method allows surgeons to easily adjust the implant’s position for optimal patient fit and stability through this dialable and articular side augmentation. The image below depicts the implant characteristics of our InSet Glenoid:
20
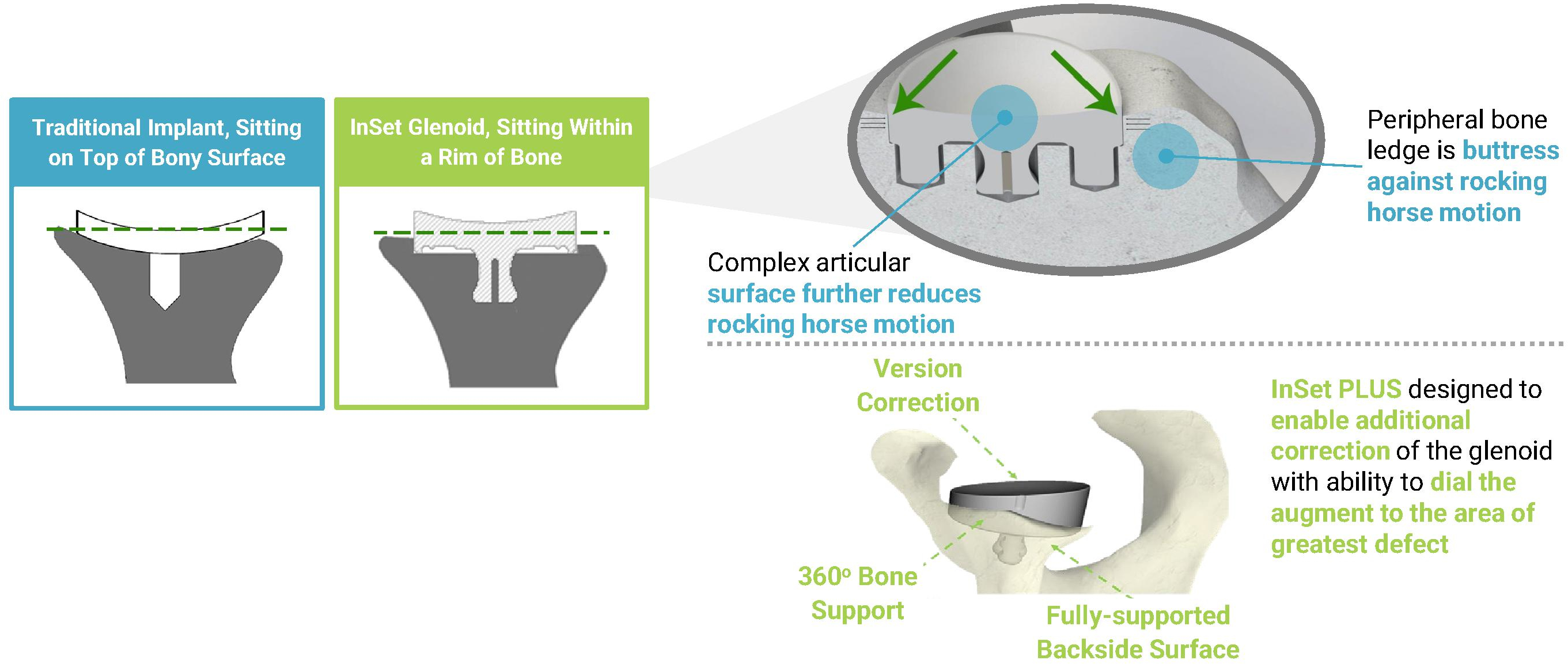
Supported by a growing body of peer-reviewed studies and approximately nine years of published data, our InSet Glenoid technology has demonstrated effectiveness across a variety of clinical indications, including cases involving advanced arthritis and severe bone loss. In addition to preserving bone in patients, our InSet Glenoid technology has been shown to facilitate quick recovery times, decreased pain, and improved range of motion for patients after surgery.
We commenced development efforts with our InSet Glenoid in 2009 and received 510(k) clearance in 2011. We commercially launched an initial aTSA system with our InSet Glenoid in 2016. We commenced development efforts for our InSet PLUS Augmented Glenoid in 2019 and received 510(k) clearance in 2020. Each of these devices is classified by the FDA as a Class II device.
InSet Humeral Stems
We offer three unique InSet humeral stem system options for our aTSA and rTSA systems—Humeral Stemless, Humeral Short Stem, and I-Series Humeral Stem—each purpose-built to address the specific needs of patients and physicians. Our humeral stems feature a proprietary porous coating designed to promote bone ingrowth by allowing bone tissue to grow into the porous structure (osseointegration), which creates a strong natural biological fixation and improves implant stability. Our stems also feature what we believe is an industry-only 132.5 degree neck shaft angle, a feature that enables our simplified surgical technique focused on proximal geometry—the shape, size, and position of the proximal humerus (upper arm bone) and its relationship to the glenoid (shoulder blade). The image below depicts our primary current humeral stems:

We commenced development efforts for our short stem, stemless and I-Series humeral stem system options for our aTSA and rTSA systems in 2017, 2019 and 2021, respectively, and received our primary 510(k) clearances in 2018 and 2022, with an additional 510(k) clearance in 2024 for use of the InSet 95 stem for use with anatomic fractures. Each of these devices is classified by the FDA as a Class II device.
21
Our novel humeral stem solutions are differentiated and designed to be compatible across the full range of our implant systems. Surgery with each humeral stem involves a consistent surgical technique and instruments, which enables seamless interchangeability between our aTSA and rTSA systems. This interchangeability is important in cases where the surgeon makes an intraoperative decision to switch from a stemless to a stemmed implant, or in revision or conversion surgeries where the determination is made to switch to a different implant modality (anatomic or reverse) or alternative humeral stem design. The image below depicts the designed compatibility of our systems:
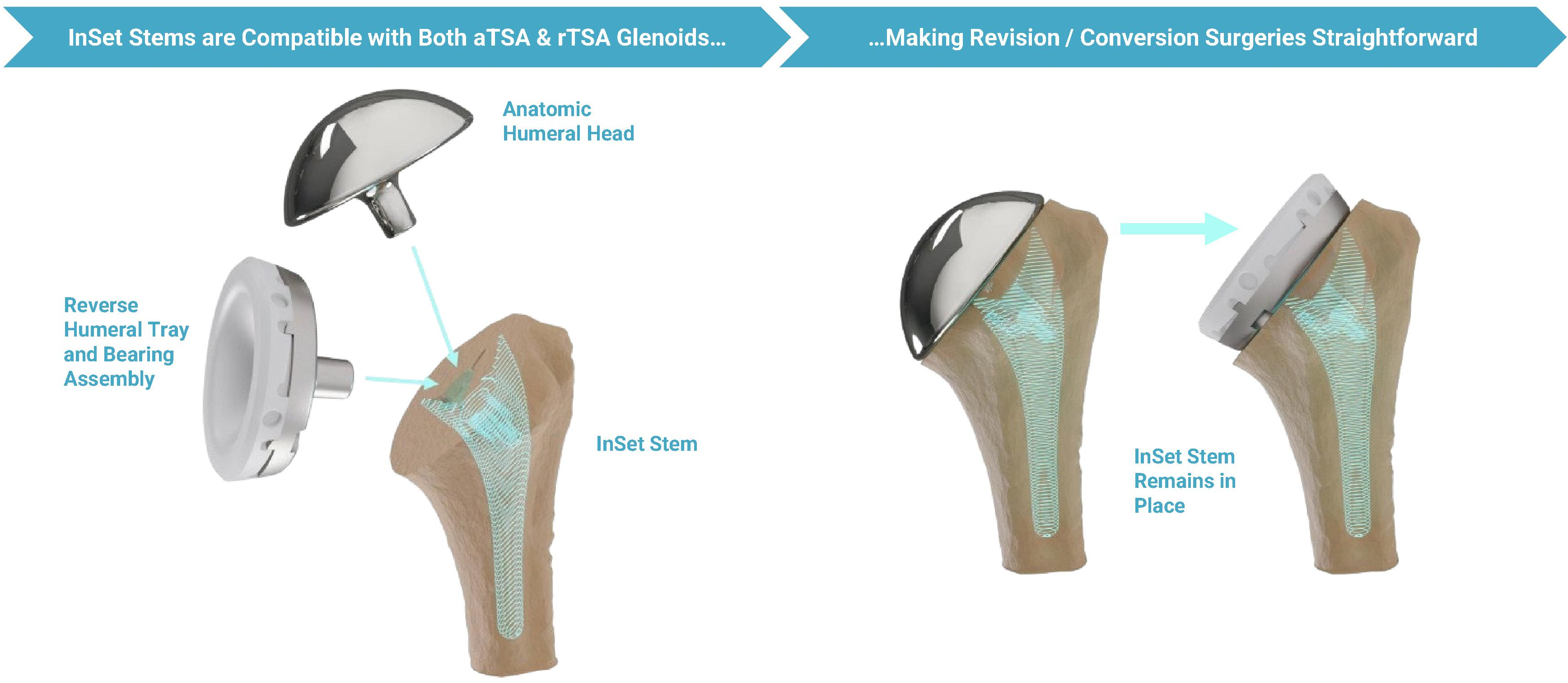
InSet rTSA System
Our rTSA configuration leverages the same humeral fixation platform as our aTSA configuration but utilizes alternative humeral and glenoid articular components designed for the reverse modality. The humeral component is configured to allow attachment of a tray and bearing assembly, designed to articulate with the glenoid sphere (“glenosphere”). The glenosphere is attached to a baseplate configured to provide reliable fixation to the glenoid on the scapular bone.
Our rTSA system was designed to optimize biomechanics and provide patients and physicians with a novel reverse implant solution that functions more like an anatomic implant in terms of improved range of motion. The InSet design of our rTSA system allows for retention of the patient’s native anatomy and aesthetic and helps to achieve desirable, impingement free range of motion by avoiding arm lengthening, overstuffing, and other common challenges.
Humeral Tray and Bearing Assembly
Our humeral tray and bearing assembly are comprised of two components—a titanium humeral tray, and a polyethylene bearing—designed for low-profile assembly using our proprietary “Twistlock” locking mechanism. The Twistlock mechanism improves bearing-to-tray pullout strength, reducing the potential for bearing disassociation, a serious complication in rTSA where the polyethylene liner (“socket”) separates from the humeral tray (“ball”).
Our humeral tray and bearing assembly feature a unique bowl-shaped design that, together with the low profile nature of the Twistlock design, allows for inlay biomechanics with a rTSA modality, in which the apex of the glenosphere is positioned on the articular surface. This tray and bearing assembly, a key component of our rTSA system, connects to our humeral stems in the same manner as our InSet humeral head solutions, a key component of our aTSA system. The identical connection mechanism between these key components of our aTSA and rTSA systems is a critical design feature that enables the interchangeability of our systems and the potential for the humeral stem to remain in place during a revision or conversion surgery.
Scapular Reverse Assembly
The scapular assembly for rTSA is comprised of two main components—a baseplate and a glenosphere—assembled by attaching the baseplate to the scapula using peripheral and central screws, then connecting the glenosphere to the baseplate using our proprietary locking cap technology. We have developed a wide range of baseplate and glenosphere options, which allows for multiple configurations to support a broad range of patient anatomy. With the use of
22
ProVoyance, the assembly is configured to a patient’s unique needs. This assembly enables a lateralized center of rotation that, together with our humeral assembly, results in a reverse implant that we believe feels and functions more like an anatomic shoulder replacement, with optimized range of motion and joint stability.
A key advantage of our rTSA system is the ability to treat complex glenoid deformities with minimal bone removal, enabled by our augmented baseplates developed with a concave backside design. The augmented baseplate is positioned within the subchondral bone which allows for correction of complex glenoid defects, which we believe improves implant stability and can preserve bone. Unlike typical augmentation options, our InSet reverse scapular components enable complete, flush contact between the implant and the native bone, without the need for a bone graft and eliminating multiple bone preparation steps.
We commenced development efforts for our rTSA system in 2019 and received 510(k) clearance in 2021. Our rTSA system is classified by the FDA as a Class II device.
ProVoyance Preoperative Planning Technology
We believe that surgeon-level engagement in preoperative planning—that is, detail-by-detail procedural planning performed by surgeons themselves—is a key element in providing effective surgical care for the patient. Hands-on preoperative planning by the surgeon provides for intimate familiarization with the patient’s unique anatomy and surgical needs and reveals key learnings which guide preoperative and intraoperative decision making. For these reasons, we offer a highly approachable, easy to use preoperative planning technology with an intuitive interface designed to engage surgeons at levels we believe are industry leading.
ProVoyance integrates AI and ML to transform planar CT imaging into 3D renderings of patient-specific anatomy ahead of surgery. Its AI and ML algorithms empower surgeons to independently create bespoke surgical plans that can help ensure consistent, effective positioning of our implants.
ProVoyance received 510(k) clearance from the FDA in 2021 and is classified by the FDA as a Class II device. ProVoyance is listed on the FDA’s AI/ML-enabled medical devices list, which is a resource maintained, published, and periodically updated by the FDA to identify AI/ML-enabled devices that have been authorized for marketing in the United States through any of the standard paths to market for medical devices, although it is not intended to be a comprehensive list of all such devices that incorporate AI/ML. This is highly differentiated from other preoperative planning solutions, where the implant manufacturer or another third-party creates a surgical plan that is sent to the surgeon for their consideration.
We believe the differentiation and value proposition of ProVoyance is validated by high utilization rates of ProVoyance across procedures using our advanced implant systems. For the year ended December 31, 2025, to the extent we are able to measure, virtually all surgeries using our implants use ProVoyance, making it a routine part of surgeon workflow. We believe this high surgeon utilization rate of ProVoyance is due to its ease of use and technical feature set. Ease of use is facilitated by the platform’s highly intuitive, Unity-based interface with full user control and high-quality graphics capabilities. Technically, ProVoyance delivers true full-depth 3D rendering, 2D/3D bone density analysis, and biplanar glenoid deformity correction. ProVoyance provides an enhanced preoperative planning experience for surgeons and allows them to approach procedures with high confidence in their surgical plan.
23
The image below depicts the ProVoyance interface:
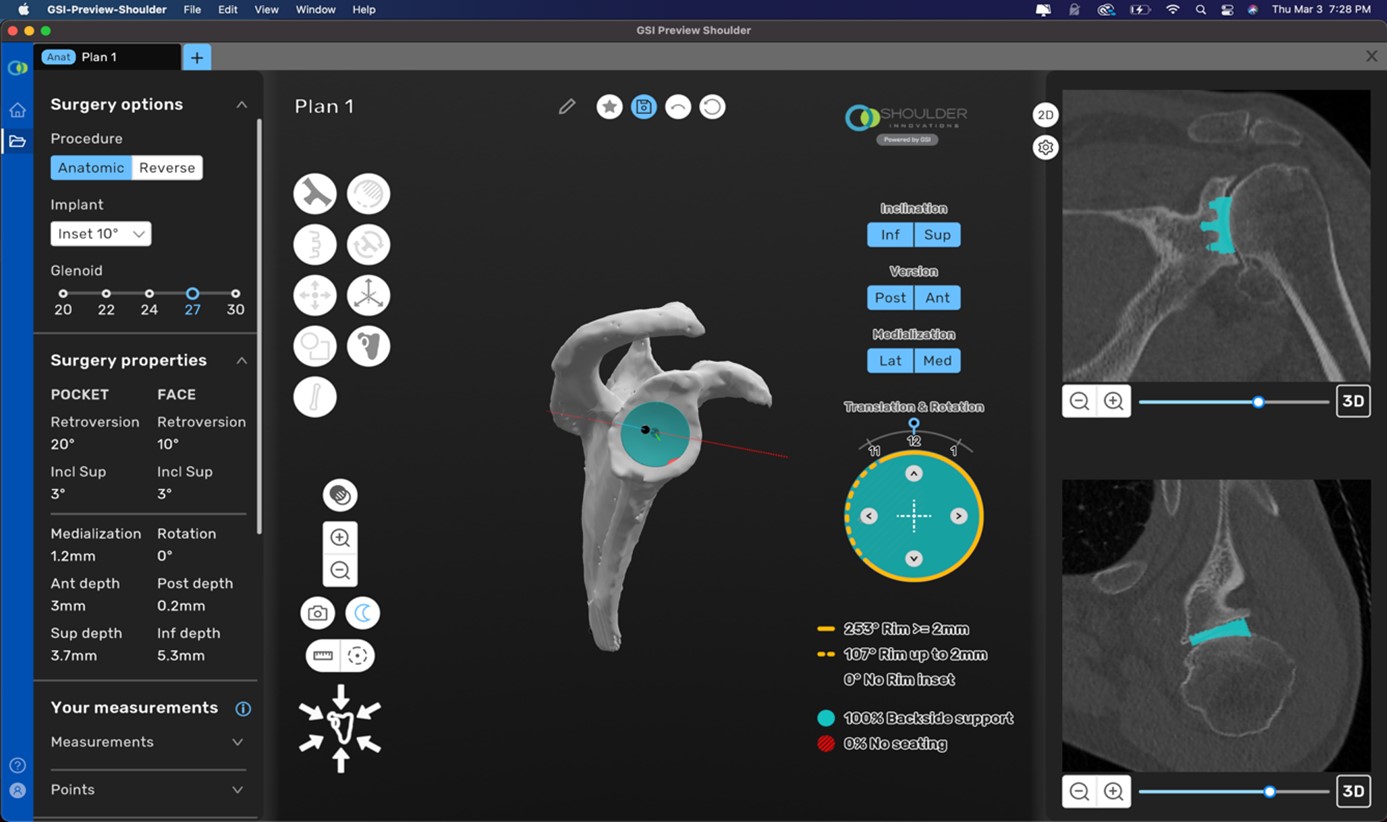
Efficient Instrument System
We re-engineered the components and technique for shoulder arthroplasty in order to create our proprietary InSet instrument system. This system was designed to enable interoperability between our novel aTSA and rTSA systems and a range of humeral stem options, while minimizing complexity and streamlining workflows.
Our instrument system is comprised of many components that fit within two trays, which we have specifically designed to facilitate the surgical technique used in connection with our implant systems. The components of each tray were designed for our implant characteristics and function. For example, the shape of the fixation fins on the humeral components were specifically designed to facilitate a surgical technique that can be used across procedure types and systems, and each instrument was designed with a view to maximize utility and reduce the overall quantity of instruments used in procedures involving our systems.
We believe our efficient, two tray instrument system can enable surgeons and staff to reduce operating room footprint, procedural setup time, sterilization time and expense and procedural complexity. We believe these benefits make it an optimal solution for aTSA and rTSA procedures performed across care settings and meaningfully reduce the per procedure capital outlay in shoulder surgical care. This advantage is particularly evident in ASCs, which generally have fewer resources and smaller footprints to handle or coordinate significant operative tools and complex instrument systems. The image below depicts our efficient instrumentation system for aTSA and rTSA:
24

Our Commercial Approach
We view ourselves as specialists serving specialists, having purposefully built our commercial organization around the unique needs of shoulder surgeons. Our commercial organization is comprised of three key components: (a) a dedicated commercial leadership team, (b) a CEME team and (c) a network of independent distributors. These key components work in tandem to form a commercial flywheel that is designed to build and provide key product support to surgeons and other stakeholders in the shoulder surgical care market, accelerate adoption, and enhance long-term retention.
The image below depicts the three key components of our commercial organization:
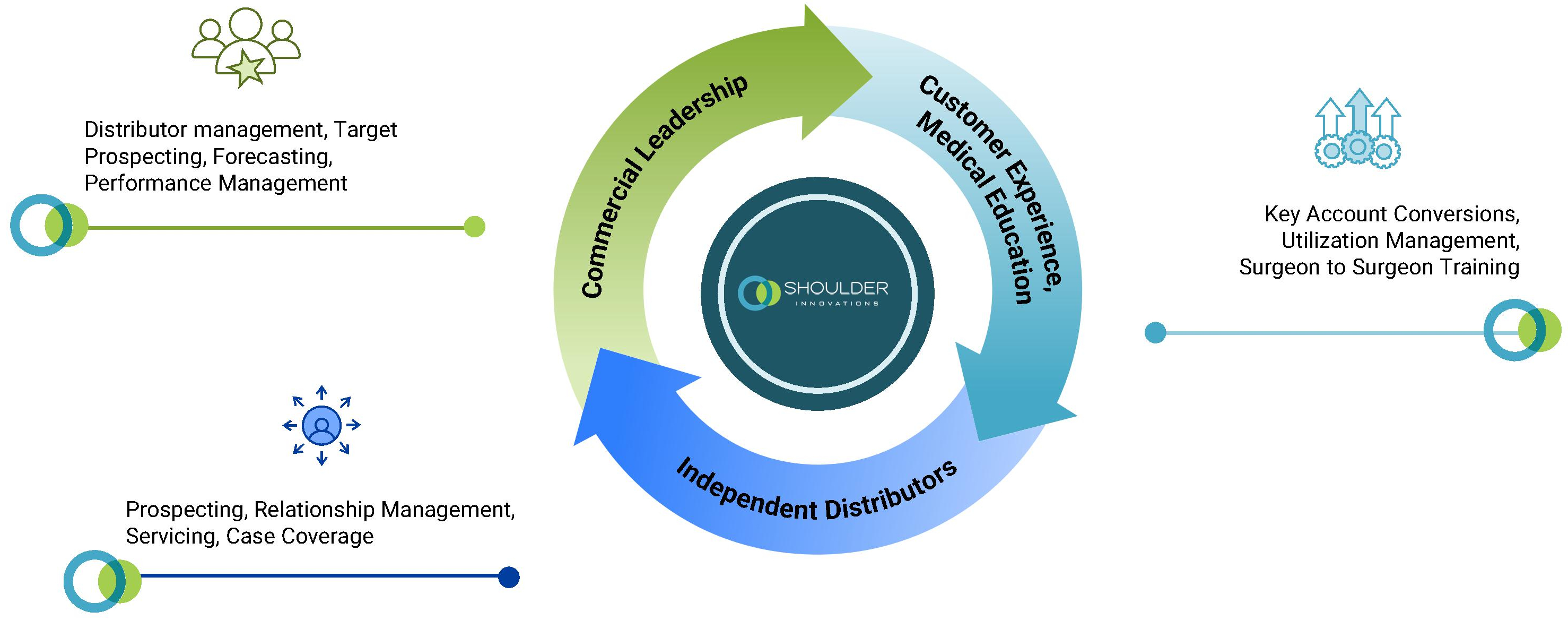
We estimate that approximately 15,000 surgeons in the United States perform at least one shoulder arthroplasty procedure per year, with approximately 1,800 high-volume surgeons performing the vast majority of procedures. We believe these high-volume surgeons require dedicated expertise and support. To optimize our commercial strategy, we have developed proprietary business intelligence tools that enable us to identify and engage with the high-volume surgeons most likely to adopt our solutions. These tools analyze key data points such as surgeon location, procedure mix and care settings, enabling us to prioritize outreach and allocate commercial resources efficiently. By understanding where and how these high-volume surgeons practice, we can tailor our engagement strategies to align with their clinical and operational needs.
Once a surgeon is integrated into our ecosystem, we focus on expanding utilization by increasing the percentage of procedures performed with our solutions. To support our targeting and commercial efforts, we have classified surgeon customers into three categories: prospect surgeons, who perform between one and two shoulder arthroplasty procedures using our implant systems per quarter (“Prospect Surgeons”); contender surgeons, who perform between three and eight shoulder arthroplasty procedures using our implant systems per quarter (“Contender Surgeons”); and core surgeons, who
25
perform at least nine shoulder arthroplasty procedures using our implant systems per quarter (“Core Surgeons”). A key driver of increasing adoption within our existing surgeon base over time (helping Prospect Surgeons become Contender Surgeons, and Contender Surgeons to become Core Surgeons) is our surgeon-to-surgeon education program facilitated by our CEME team. We are committed to fostering a collaborative community among shoulder surgeons where expertise is shared, new ideas are exchanged and best practices are disseminated to enhance clinical outcomes. By facilitating these peer connections, we strengthen engagement within our ecosystem and reinforce its real-world success. As surgeons share their positive experiences with our ecosystem and the impact on patient outcomes, we believe this organic advocacy further accelerates adoption, positioning us to benefit from the natural network effects within the shoulder surgical care community.
Using this commercial approach, we aim to drive adoption and momentum with both existing and new surgeons and are continuing to increase our number of high-volume surgeons over time. For example, our number of Core Surgeons and Contender Surgeons has increased over time. For the year ended December 31, 2023, 2024 and 2025, we had 60, 83 and 134 Core Surgeons and Contender Surgeons, respectively.
Dedicated Commercial Leadership Team
As of December 31, 2025, our dedicated commercial leadership team was comprised of 33 specialized representatives, organized into two commercial territories in the United States. Our team is focused on identifying high-volume surgeons specializing in shoulder surgical care. Their main responsibilities include: (a) target prospecting, by leveraging our business intelligence tools to identify and engage high-volume surgeons; (b) manage our distributor relationships, which helps to ensure alignment across our commercial organization and network of third-party distributors; and (c) facilitate case coverage for each shoulder surgery performed with our systems, either directly or through our network of distributors.
Dedicated Customer Experience and Medical Education Team
As of December 31, 2025, our CEME team was comprised of seven expert surgeon educators who facilitate rewarding and meaningful experiences for surgeons focused on clinical value. A key responsibility of our CEME team is facilitating quality surgeon-to-surgeon educational opportunities, which naturally results in expanding surgeon utilization of our solutions. This team hosts medical education events and activities such as symposiums and lab events where like-minded surgeons can exchange ideas and nurture a culture of innovation in shoulder care. These in-person engagements are designed to build long-term relationships that extend well beyond the events themselves. For that purpose, we have developed online forums where high-volume users of our products can collaborate, share case experiences, and consult one another in real time. This community of surgeons often becomes the most passionate advocates for our solutions, often educating, training, and engaging other surgeons in the benefits of joining our ecosystem.
Independent Distributor Network
Unlike typical stocking distributors, this network plays a critical role in expanding our market reach and driving adoption of our solutions in a cost-effective and scalable manner. In most cases, these distributors are focused on the orthopedic marketplace and sign exclusive agreements to carry our systems as their dedicated shoulder solution. They provide valuable feedback from the marketplace and are responsible for prospecting new surgeon customers, managing relationships, servicing existing accounts, and providing case coverage to support surgical execution. By maintaining strong, localized relationships with surgeons and healthcare facilities, our distributors help facilitate initial engagement, ongoing support, and long-term retention. Our dedicated commercial leadership team actively manages these relationships, ensuring distributors are meeting performance expectations and driving sales growth. We continuously evaluate distributor performance, maintaining relationships with those who generate the most sales while upgrading distribution partners where commercial execution can be improved.
Product Development and Pipeline
We leverage our team’s decades of collective experience developing and launching novel shoulder surgical care technologies to identify solutions addressing the unmet needs of patients and surgeons. Since the initial launch of our InSet Glenoid in 2016, we have successfully commercialized a wide range of new technologies to enhance our ecosystem and provide surgeons with the tools and support needed to deliver quality outcomes for patients requiring shoulder surgical care. We believe our commitment to continuous innovation has been demonstrated by our track record of developing leading new technologies.
We continue to see clinical outcomes in the shoulder surgical care space that are inferior to those seen in more mature orthopedic markets and intend to continue working with our surgeon advisors to further improve clinical outcomes in
26
shoulder surgical care. We believe this continued commitment to innovation will further expand our addressable market opportunity and improve our competitive position in shoulder surgical care.
We have a robust pipeline of new technologies in various stages of development and evaluation, including the following select projects.
I-Series Expansion and New Indications
We engineered the I-Series humeral stem line to provide a novel stem option for patients suffering from arthritis and a wide spectrum of proximal humeral bone loss or density. Our I-Series humeral stems feature a 2-fin design, specifically engineered for superior rotational control and stability. We launched our initial I-Series system in 2023, the InSet 95 Humeral Stem, which has been well received by the marketplace. The InSet 70 was launched in September 2025, and certain InSet 135 indications have received approval. We are also developing InSet 185 stems to further expand our I-Series humeral stem line. We anticipate pursuing FDA clearance of the InSet 185 stems, as needed, over the next twelve months. These additional clearances, if received from the FDA, will extend the range of available stem sizes and include expanded indications into fracture and revision surgeries.
Technologies for Metal Hypersensitive Patients
Market awareness of the risks and prevalence of metal hypersensitivity has risen in recent years. We have developed a line of humeral head and glenoid technologies for the approximately 10% to 15% of the general population who test positively for a metal hypersensitivity and may experience persistent pain, or other symptoms associated with allergic reactions from metal implants. These development technologies include our Humeral Head and Glenosphere, each constructed from alternative materials which do not incorporate elements that typically represent higher allergic risk for patients who test positively for a metal hypersensitivity. We have received FDA clearance for these solutions in January of 2026.
Adjacent Market Expansion
We are also evaluating expansion into adjacent areas in shoulder surgical care, which may include sports medicine and shoulder trauma markets.
Robotics Platform
In December 2025, the Company announced the development of a robotics platform to design a transformative shoulder-specific micro-robotic solution designed to further enhance shoulder surgical precision, workflow efficiency, and enable exciting, new clinical approaches in the aTSA and rTSA markets. The robotic solution is designed to be integrated with the Company’s ProVoyance platform to deliver a comprehensive technology solution.
Clinical Overview
There is a significant body of clinical evidence that supports the safety, efficacy, and durability of our implants in shoulder arthroplasty, including our InSet Glenoid technology. We are committed to continued investment in obtaining further clinical evidence with the support of surgeons who are recognized as thought leaders in shoulder surgical care. We believe these efforts will continue to generate a substantial body of additional clinical evidence that will drive increased awareness and adoption of our products.
Since our founding, we have contributed to numerous publications that we believe evidence and strengthen our position as a leader in our industry. These studies focus on various patient outcome measures related to pain relief, range of motion, and complication rates, such as ASES outcome scores, Visual Analog Scale (“VAS”) scores and Single-Assessment Numeric Evaluation (“SANE”) scores. An ASES score is a metric derived from a postoperative questionnaire used by surgeons to assess shoulder function and patient well-being. ASES scores are based on a 100-point scale with higher scores indicating better function and less pain. A VAS score is a metric used to measure patient-perceived pain intensity with 10 indicating highest level of pain and 0 indicating no pain. A SANE score is a metric used to measure patient improvement where patients rate their shoulder on a scale from 0 to 100, with 100 being the patient’s normal function. In addition, these studies focus on the most frequent complication associated with shoulder arthroplasty, failure of the glenoid component and published research demonstrates superior implant survivorship (i.e., lack of revision).
27
As described in greater detail in the table below, the following is a summary of certain of these supportive, peer-reviewed publications:
•A study published in The Journal of Shoulder and Elbow Surgery in 2011 evaluated clinical outcomes of our InSet Glenoid implants at a mean follow-up time of 4.3 years. The results demonstrated statistically significant improvements in the mean ASES outcome score (68-point improvement), VAS scores (6.8-point improvement) and range of motion, with no surgical complications.
•A study published in The Journal of Shoulder and Elbow Surgery in 2012 evaluated the fixation strength and stress distribution of our InSet Glenoid fixation technique. In this study, our InSet Glenoid implants demonstrated no signs of loosening and up to an 87% reduction in displacement.
•A study published in The Journal of Shoulder and Elbow Surgery in 2019 evaluated clinical outcomes of aTSA procedures with our InSet Glenoid at a mean follow-up time of 8.7 years. The results indicated statistically significant improvements in the mean ASES outcome score (72-point improvement), VAS scores (7.6-point improvement) and range of motion, with no surgical complications, no cases of glenoid loosening and no revision surgeries performed at follow-up.
•A study published in The Journal of Shoulder and Elbow Surgery in 2023 evaluated clinical outcomes of aTSA procedures with our InSet Glenoid at a mean follow-up time of 28.7 months. There results indicated significant improvements in ASES scores (43-point improvement), VAS scores (4.2-point improvement) and SANE scores (51.6-point improvement, and statistically significant improvement in range of motion). Additionally, there were low rates of central peg radiographic lucency, asymptomatic loosening of the InSet Glenoid implant, and no patients were revised for implant-related complications.
The table below sets forth additional information regarding these peer-reviewed publications. Some of these studies were conducted with small sample sizes, did not control for clinical variables or have other design limitations (e.g., the studies may be retrospective and are not randomized controlled trials). Except as otherwise noted in the table below, we did not fund or sponsor any of these peer-reviewed publications nor were our employees involved in the studies or publications.
28
| Reference | Source | Study Summary | |||||||||||||||
| Gunther et al. (2011) | The Journal of Shoulder and Elbow Surgery | Title: Total shoulder replacement surgery with custom glenoid implants for severe bone deficiency Authors: Stephen B. Gunther, MD; Tennyson L. Lynch, BS Description: A retrospective review of seven consecutive patients (three men, four women) treated with shoulder arthroplasty surgery with custom inset glenoid implants for severe bone deficiency at a minimum of three years. Conclusions: This study documented for the first time the use of custom-made inset glenoid implants in the treatment of severely deficient bone defects for which standard implants are contraindicated. The surgical technique has allowed glenoid implantation to be performed safely in this small group of patients, and the inset technique has been an effective short-term solution for these patients. At a mean follow-up of 4.3 years, there were statistically significant improvements (P < 0.02) in VAS scores (6.9 to 0.1), ASES outcome scores (26 to 94), and range of motion. No surgical complications occurred. Independent radiographic analysis determined all implants were classified as “low risk” for glenoid loosening. | |||||||||||||||
| Gunther et al. (2012) | The Journal of Shoulder and Elbow Surgery | Title: Finite element analysis and physiologic testing of a novel, inset glenoid fixation technique Authors: Stephen B. Gunther, MD; Tennyson L. Lynch, BS; Desmond O’Farrell, B.Eng (Hons) MM; Christian Calyore; Andrew Rodenhouse, BS Description: A comparative scientific analysis of glenoid loosening using an inset glenoid fixation technique vs. the standard onlay technique used with a keel or pegged implant. The analysis consists of two separate methods: First, physiologic in vivo cyclic loading of glenoid implants was simulated using the dynamic model described by Anglin et al. and American Society for Testing and Materials (ASTM) F2028-08 Standard Test Methods for Dynamic Evaluation of Glenoid Loosening or Disassociation. Second, finite element analysis was performed to estimate the glenohumeral joint stress and displacement for both standard onlay implants and an inset glenoid implant. Conclusions: The mechanical testing in this study of the inset glenoid design and fixation technique showed a significant reduction in post-testing distraction after physiologic loading compared with standard onlay design and fixation. The results of the finite element analysis support the concept of inset glenoid fixation based on the significant reduction in stresses on the backside surface on the inset implant compared with the standard onlay implant. Also, the nodal displacement at the edges of the implants under rim loading conditions was significantly reduced for the inset implants, and there was more uniformity of stress distribution along the inset polyethylene surface. This testing shows increased glenoid fixation strength using an inset technique, which may be beneficial in minimizing clinical glenoid loosening. | |||||||||||||||
29
| Gunther et al. (2019)* | The Journal of Shoulder and Elbow Surgery | Title: Long-term follow-up of total shoulder replacement surgery with inset glenoid implants for arthritis with deficient bone Authors: Stephen B. Gunther, MD; Sterling K. Tran, BS Description: A retrospective analysis was performed on 21 of 24 consecutive patients treated with inset glenoid implants for severe glenohumeral joint arthritis with bone deficiency with prospectively collected data. Inclusion criteria were patients with shoulder arthritis and severe glenoid bone deficiency, defined by perpendicular glenoid vault depth less than 15 mm. No bone grafts were used. All patients were evaluated preoperatively and after surgery with physical examination, radiographic studies and outcome measures. There were 10 males and 11 females, 17 cases with osteoarthritis and 4 with inflammatory arthritis, and 5 patients with rotator cuff tears (3 full thickness and 2 partial tears). Mean age was 68 years. Conclusions: This inset glenoid fixation technique offers an innovative approach to a difficult clinical conundrum of shoulder arthritis with deficient glenoid bone. In this series, there were no complications, no cases of glenoid implant loosening and no revision surgeries performed at a mean 8.7-year follow-up. This technique is also safe, because there is only minimal penetration of the glenoid surface bone. Finally, the technique is simple and easily reproducible. It is a reasonable alternative to other current techniques available for patients with shoulder joint arthritis, severe bone deficiency, and an intact rotator cuff. At a mean follow-up of 8.7 years, there were statistically significant improvements (P < 0.001) in VAS scores (7.7 to 0.1), ASES outcome scores (23 to 95) and range of motion. | |||||||||||||||
| Johnston et al. (2023) | The Journal of Shoulder and Elbow Surgery | Title: Clinical and radiographic outcomes following anatomic total shoulder arthroplasty utilizing an inset glenoid component at 2-year minimum follow-up: a dual center study Authors: Peter S. Johnston, MD; John T. Strony, MD; Jessica L. Churchill, MD; Roma Kankaria, BS; Benjamin W. Sears, MD; Grant E. Garrigues, MD; Robert J. Gillespie, MD Description: A retrospective review of 75 patients undergoing aTSA using an inset glenoid component by two fellowship-trained shoulder surgeons at two separate institutions from August 2016 to August 2019 with minimum follow-up of two years. Range of motion and ASES, VAS and SANE scores were obtained. Radiographic outcomes, including central peg lucency and glenoid loosening, were assessed by three independent reviewers on the postoperative Grashey and axillary radiographs obtained at the final follow-up. Conclusions: At 2-year minimum follow-up, there were significant improvements in range of motion, VAS, SANE and ASES scores with low rates of central peg radiographic lucency and glenoid loosening. Additional long-term follow-up is needed to further clarify the advantages of InSet glenoid components when used in anatomic shoulder arthroplasty. | |||||||||||||||
*As part of this publication, Mr. Gunther received 6,551 warrants to purchase our Series B convertible preferred stock in January 2020.
In addition, we are investing in a registry to collect observational non-randomized standard-of-care data to document survival of our implant and evaluate the short- and long-term clinical and radiographic outcomes associated with real world use of our implant systems. The primary purpose of the registry will be to assess survival of our implant with or without
30
revision at 2 years, 5 years and 10 years post-operatively. We believe this registry will provide further clinical support and additional clinical evidence that will drive increased awareness and adoption of our products.
Coverage and Reimbursement
We sell our systems directly to hospitals, outpatient care centers and ASCs. These customers in turn bill various third-party payors, such as Medicare, Medicaid and private health insurance plans, for the total healthcare services required to treat the patient. Government agencies, private insurers and other payors determine whether to provide coverage for a particular procedure and to reimburse hospitals, outpatient care centers and ASCs at rates based on a prospective payment system. For procedures performed during a hospital inpatient stay, Medicare generally reimburse hospitals a single bundled payment that is based on the patient’s principal diagnosis, up to 24 additional diagnoses and up to 25 procedures performed during the stay. Cases are classified into Medicare Severity-Diagnosis Related Groups, for payment under the Medicare Inpatient Prospective Payment System, for all items and services provided to the patient during a single hospitalization, regardless of whether procedures utilizing our products are performed during such hospitalization. Medicare rates for the same or similar procedures vary due to geographic location, nature of facility in which the procedure is performed and other factors. With respect to procedures performed in a hospital outpatient setting, all items and services paid under the Medicare outpatient prospective payment system, are assigned to payment groups called Ambulatory Payment Classifications, which group together items and services that are similar clinically and in terms of resource use. With respect to ASCs, covered procedures are assigned to ASC payment groups, which then determines the amount that Medicare pays for facility services furnished in connection with a covered procedure. Effective January 1, 2024, CMS added total shoulder arthroplasty to the list of covered procedures that can be performed in ASCs, which facilitated reimbursement and further supported the growth of ASCs as a key site of care in the shoulder surgical care market. While private payors vary in their coverage and payment policies, most use coverage and payment by Medicare as a benchmark by which to make their own decisions.
Reimbursement for professional services performed by physicians are also reported using CPT codes and based on a different payment methodology. Procedures using our systems are reported under CPT code 23470 for partial shoulder arthroplasty and CPT Code 23472 for total shoulder arthroplasty:
•CPT 23470 (Arthroplasty, glenohumeral joint; hemiarthroplasty) – Customers may categorize a procedure using our system as a partial shoulder arthroplasty when the reconstruction procedure is limited to the humeral articulation, and no replacement of the scapular articular surface (glenoid) is made.
•CPT 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement (e.g., total shoulder))) – Customers may categorize a procedure using our system as a total shoulder arthroplasty when either aTSA or rTSA reconstruction is performed and the articular surfaces of both the humerus and the scapula are replaced by artificial components.
Intellectual Property
Intellectual property, including patents, trade secrets, trademarks and copyrights, is important to our business. Our commercial success depends in part on our ability to obtain and maintain proprietary intellectual property protection for our current products as well as for future product candidates and novel discoveries, product development technologies, and know-how. Our commercial success also depends in part on our ability to operate without infringing the proprietary rights of others and to prevent others from infringing our proprietary rights. We seek to maintain our proprietary position by, among other means, filing United States and foreign patent applications to obtain issued patents with claims directed to our products, product candidates, technology, inventions, and improvements that are important to the development and implementation of our business. We may also license from third parties certain patent rights and proprietary know-how that we believe to be necessary or useful to our business. Additionally, we protect our proprietary know-how that may not be patentable, and other confidential information, by maintaining and implementing appropriate policies and procedures with respect to secrecy and confidentiality.
We are currently seeking and maintaining patent protection in the United States and key foreign jurisdictions where we intend to market our products, and plan to do so with respect to any of our future product candidates. Our patent portfolio includes a combination of patents and pending patent applications solely held by us.
As of December 31, 2025, our owned patent estate contains 11 patent families comprising 13 issued U.S. patents, nine issued foreign patents, 13 pending U.S. non-provisional patent applications and 20 pending foreign patent applications. The patent families cover different aspects of glenoid implants, humeral implants, instrumentation, and reverse shoulder replacement systems, devices and methods.The 13 issued U.S. patents are expected to expire between August 2027 and
31
October 2041, after accounting for potentially available patent term adjustments, and assuming payment of appropriate maintenance, renewal, annuity and other governmental fees. Any patents that may issue from the 13 pending U.S. patent applications are expected to expire between September 2035 and August 2043, without accounting for potentially available patent term adjustments, term-limiting effects of terminal disclaimers and assuming payment of appropriate maintenance, renewal, annuity and other governmental fees. The nine issued foreign patents include one or more issued patents in jurisdictions such as Australia, Canada, Europe (validated in one or more of Germany, France, Switzerland and the U.K. and/or registered as having unitary effect) and Japan, and are expected to expire between February 2026 and March 2040, without accounting for potentially available patent term extensions and assuming payment of appropriate maintenance, renewal, annuity and other governmental fees. The 20 pending foreign patent applications include one or more pending applications in jurisdictions such as Australia, China, Europe and Japan, and are expected to expire between April 2038 and January 2045, without accounting for potentially available patent term extensions and assuming payment of appropriate maintenance, renewal, annuity and other governmental fees. Calculation of the expiration of issued patents is complex, varies by country and is based upon many factors. Accordingly, the expiration dates are estimates.
Our owned U.S. and foreign patents and patent applications generally relate to aTSA, rTSA, and instrumentation systems for aTSA and rTSA. Our owned issued U.S. and foreign patents are set forth in the table below, which are directed to our relevant technologies, including glenoid implants, humeral implants, reverse shoulder replacement systems and instrumentation for shoulder replacement.
The term of individual patents in our portfolio depends upon the legal term of patents in the countries in which they are obtained. In most countries in which we file, including the United States, the patent term is 20 years from the earliest date of filing a non-provisional patent application. In the United States, the term of a patent may be reduced due to terminal disclaimer made to overcome a double patenting rejection, or may be lengthened by patent term adjustment, which permits patent term restoration as compensation for delays incurred at the USPTO during the patent prosecution process.
The patent positions of companies like ours are generally uncertain and involve complex legal and factual questions. The relevant patent laws and their interpretation outside of the United States is also uncertain. Changes in either the patent laws or their interpretation in the United States and other countries may diminish our ability to protect our technology or product candidates and could affect the value of such intellectual property. In particular, our ability to stop third parties from making, using, selling, offering to sell or importing products that infringe our intellectual property will depend in part on our success in obtaining and enforcing patent claims that cover our technology, inventions and improvements. We cannot guarantee that patents will be granted with respect to any of our pending patent applications or with respect to any patent applications we may file in the future, nor can we be sure that any patents that may be granted to us in the future will be commercially useful in protecting our products, the methods of use or manufacture of those products. Moreover, issued patents do not guarantee the right to practice our technology in relation to the commercialization of our products. Issued patents only allow us to block potential competitors from practicing the claimed inventions of the issued patents in the countries in which such patents are issued.
Further, patents and other intellectual property rights in the medical device space are evolving and involve many risks and uncertainties. For example, third parties may have blocking patents that could be used to prevent us from commercializing our product candidates and practicing our proprietary technology. Our issued patents may be challenged, invalidated or circumvented, which could limit our ability to stop competitors from marketing related products or could limit the term of patent protection that otherwise may exist for our product candidates. In addition, the scope of the rights granted under any issued patents may not provide us with protection or competitive advantages against competitors with similar technology. Furthermore, our competitors may independently develop similar technologies that are outside the scope of the rights granted under any issued patents. For these reasons, we may face competition with respect to our product candidates. Moreover, because of the extensive time required for development, testing and regulatory review of a potential product, it is possible that, before any particular product candidate can be commercialized, any patent protection for such product may expire or remain in force for only a short period following commercialization, thereby reducing the commercial advantage the patent provides.
We also rely on trade secrets relating to our discovery programs and product candidates, and seek to protect and maintain the confidentiality of proprietary information to protect aspects of our business that are not amenable to, or that we do not consider appropriate for, patent protection. It is our policy and practice to require our employees, consultants, outside scientific collaborators, sponsored researchers and other advisors to execute confidentiality agreements upon the commencement of employment or consulting relationships with us, and for employees and consultants to enter into invention assignment agreements with us. These agreements provide that all confidential information developed or made known to the individual during the course of the individual’s relationship with us is to be kept confidential and not disclosed to third parties except in specific circumstances. Where applicable, the agreements provide that all inventions to
32
which the individual contributed as an inventor shall be assigned to us, and as such, will become our property. There can be no assurance, however, that these agreements will provide meaningful protection or adequate remedies for our trade secrets in the event of unauthorized use or disclosure of such information.
Software License Agreement with Genesis Software
On October 22, 2020, we entered into a software license agreement with Genesis Software, which was amended and restated on January 1, 2023 (as amended and restated, the “License Agreement”). Pursuant to the License Agreement, Genesis Software granted us an exclusive, worldwide, transferable, sublicensable and royalty-bearing license under Genesis Software’s SaaS surgery planning platform software, including all intellectual property rights therein, updates, upgrades and other modifications thereto, and object code, source code and other surgery planning software developed or controlled by Genesis Software during the term of the License Agreement (collectively, the “Licensed Software”), to store, use copy, make derivative works from, modify, extend, enhance and improve the Licensed Software in the field of shoulder surgical procedures primarily related to replacement of the shoulder joint articulation (i.e., anatomic shoulder arthroplasty, reverse shoulder arthroplasty, revision shoulder arthroplasty, and hemi-shoulder arthroplasty) (the “Field of Use”). The Licensed Software is utilized in connection with ProVoyance. We granted Genesis Software a limited, non-exclusive, non-transferable license to use data generated by us through our use of the Licensed Software solely in connection with customization of an implant other software products outside the Field of Use. Genesis Software granted us a limited, non-exclusive, non-transferable license under identified data collected by Genesis Software through its use of the Licensed Software outside of the Field of Use for our use within the Field of Use. We also agreed to compensate Genesis Software for certain development, consulting, and support services related to the Licensed Software that we may request from time to time.
Pursuant to the License Agreement, we paid Genesis Software an upfront license fee of $1.0 million and a milestone payment of $0.5 million upon receipt of FDA clearance of the Licensed Software. We are also required to pay Genesis Software royalties of a mid-single-digit percentage on net sales of shoulder replacement products that incorporate or use the Licensed Software until the earlier of (i) the tenth anniversary of the effective date of the License Agreement and (ii) such time as the aggregate amount of royalties, upfront license fee and milestone due upon receipt of FDA clearance paid to Genesis Software reaches $7.0 million. At any time prior to the tenth anniversary of the effective date of the License Agreement, we have the right to buy out our royalty obligations by paying Genesis Software the difference between the aggregate amounts already paid to Genesis Software and $7.0 million, upon which our rights under the License Agreement shall become irrevocable and perpetual and shall be royalty-free and fully-paid.
The term of the License Agreement shall continue until earlier terminated in accordance with the terms therein. The License Agreement may be terminated for cause by either party: (i) if the other party breaches a material provision of the License Agreement and fails to cure such breach within a specified notice and cure period or (ii) upon the bankruptcy, insolvency or certain dissolution or liquidation events of other party.
Manufacturing and Supply
We utilize third-party manufacturing and supply providers to manufacture our implants. We believe this outsourcing strategy provides the expertise and capacity required to effectively and efficiently scale production based on demand, and helps to ensure low-cost production and a capital efficient business model. The majority of our products are provided by single-source suppliers. For example, Avalign Technologies manufactures and supplies our humeral stems, Micropulse is our sole source for Inset Glenoid, Trifecta Medical Group manufactures and supplies our trays and RMD supplies surgical instruments used during procedures involving our systems. We also utilize a single supplier for significant majority of our sterilization needs.
We generally do not have long-term contracts with these third parties and our supply arrangements generally do not include minimum manufacturing or purchase obligations. We primarily order products through the use of purchase orders. As such, we generally do not have any obligation to purchase any given quantity of products, and our suppliers generally have no obligation to sell to us or to manufacture for us any given quantity of our products or components of our systems. We maintain strong working relationships with our suppliers and we believe our current network of third-party manufacturing and supply providers provides for sufficient capacity to meet projected market demand for our products for the foreseeable future.
While there are other suppliers that could make or provide any one of our products, we seek to manage single-source supplier risk by regularly assessing the quality and capacity of our suppliers, implementing supply and quality control protocols where appropriate and actively managing lead times and inventory levels. In addition, we are currently in the process of identifying and approving alternative suppliers to dual- or multi-source certain of our products and we are
33
working with key strategic partners on internal redundancy of certain products. We generally seek to maintain sufficient supply levels to help mitigate any supply interruptions and enable us to find and qualify another source of supply. For certain products, we estimate that it could take between six and twelve months to find and qualify a second source. Order quantities and lead times are based on internal forecasts, which are derived from historical demand and anticipated future demand. Lead times vary depending on the size of the order, time required to manufacture, specific supplier requirements and current market demand and dynamics for the raw materials, sub-assemblies and parts related to our products.
Our third-party manufacturing and supply providers are evaluated, qualified and approved through our supplier quality program, which includes verification and monitoring procedures to help ensure that our suppliers comply with FDA and ISO standards, as well as our own specifications and requirements. We inspect and verify products under strict processes supported by internal policies and procedures. We maintain a rigorous change control policy to assure that no product or process changes are implemented without our prior review and approval.
Competition
Our industry is competitive, subject to technological change and significantly affected by new product introductions and market activities of other industry participants. Our competition includes medical device manufacturers in the shoulder surgical care market and in the shoulder arthroplasty market in particular. For example, companies operating in our market include Arthrex, Enovis, Exectech, Johnson & Johnson, Smith & Nephew, Stryker, and Zimmer Biomet as well as a number of smaller companies. There are a significant number of approved devices for shoulder surgical care and shoulder arthroplasty, and we expect competition will intensify over time.
Many of our competitors have longer, more established operating histories, and in some cases significantly greater name recognition and financial, technical, marketing, sales, distribution and other resources. In addition, certain competitors have several competitive advantages, including international operations with significant scale and established relationships with hospitals, outpatient care centers and ASCs and surgeons who use their devices and are familiar with existing devices on the market. In addition to competing for market share, we also compete against these companies for personnel, including qualified personnel that are necessary to grow our business.
We believe the principal competitive factors in our market include:
•product features and design;
•patient experience, including recovery time, level of discomfort and post-operative results;
•acceptance by surgeons and other key stakeholders in the shoulder surgical care market;
•surgeon learning curves and willingness to adopt new techniques;
•ease of use and reliability, including preoperative planning tools;
•economic benefits and efficiencies for hospitals, outpatient care centers, ASCs and surgeons;
•effective distribution and marketing to surgeons and potential patients, including physician education and information sharing programs;
•product quality and standards, including our reputation with customers and surgeons;
•intellectual property; and
•customer service and support capabilities.
We believe we have established a compelling value proposition to compete favorably in this market. However, conditions in our market could change rapidly and significantly as a result of technological advancements, partnerships, or acquisitions by competitors or continuing market consolidation and we expect the competitive environment to remain intense. For example, we have seen and continue to see consolidation amongst our competitors and have seen and continue to see our competitors innovate and improve upon traditional implants and technologies. If our competitors have greater resources and access to funding than us, they may be able to finance the development of new technologies and products before we are able to do so, which may allow them to gain market share or enter new markets before us or provide lower-priced or better-quality offerings.
34
Government Regulation
Our products and our operations are subject to extensive regulation by the FDA, and other federal and state authorities in the United States. Our products are subject to regulation as medical devices in the United States under the Federal Food, Drug and Cosmetic Act (the “FDCA”), as implemented and enforced by the FDA.
United States Regulation of Medical Devices
The FDA regulates the development, design, non-clinical and clinical research, manufacturing, safety, efficacy, labeling, packaging, storage, installation, servicing, recordkeeping, premarket clearance or approval, adverse event reporting, advertising, promotion, marketing and distribution, and import and export of medical devices, including software such as AI/ML, to ensure that medical devices distributed domestically are safe and effective for their intended uses and otherwise meet the requirements of the FDCA. In addition, the FDA collects user fees for certain medical device submissions (including all submissions for marketing authorization) and annual fees for medical device establishments.
FDA Marketing Authorization Requirements
Unless an exemption applies, each medical device commercially distributed in the United States requires either FDA clearance of a premarket notification submitted under Section 510(k) of the FDCA, approval of a PMA, or classification under the de novo classification process. Under the FDCA, medical devices are classified into one of three classes—Class I, Class II or Class III—depending on the degree of risk associated with each medical device and the extent of manufacturer and regulatory control needed to ensure its safety and effectiveness. Class I includes devices with the lowest risk to the patient and are those for which safety and effectiveness can be assured by adherence to the FDA’s General Controls for medical devices, which include compliance with the applicable portions of the Quality Management System Regulation (the “QMSR”), which amended the QSR, facility registration and product listing, Medical Device Reporting, and truthful and non-misleading labeling, advertising, and promotional materials. Class II devices are subject to the FDA’s General Controls, and special controls as deemed necessary by the FDA to provide reasonable assurance of device safety and effectiveness. These special controls can include performance standards, post-market surveillance, patient registries, and FDA guidance documents.
While most Class I devices are exempt from the 510(k) premarket notification requirement, manufacturers of most Class II devices are required to submit to the FDA a premarket notification under Section 510(k) of the FDCA (or a “510(k) notice”) requesting permission to commercially distribute the device. The FDA’s permission to commercially distribute a device subject to a 510(k) notice is generally known as 510(k) clearance. Devices deemed by the FDA to pose the greatest risks, such as life sustaining, life supporting or some implantable devices, or devices that have a new intended use or use advanced technology that is not substantially equivalent to that of a legally marketed device, are placed in Class III, requiring approval of a PMA. Some pre-amendment devices are unclassified, but are subject to FDA’s premarket notification and clearance process in order to be commercially distributed. Where software, including AI/ML-based software, qualifies as a device, this same classification scheme and regulatory path to market applies.
510(k) Clearance Marketing Pathway
To obtain 510(k) clearance, a company must submit to the FDA a premarket notification demonstrating that the proposed device is “substantially equivalent” to a legally marketed predicate device. A predicate device is a legally marketed device that is not subject to premarket approval, i.e., a device that was legally marketed prior to May 28, 1976 (pre-amendments device) and for which a PMA is not required, a device that has been reclassified from Class III to Class II or I, a device that was found substantially equivalent through the 510(k) process, or a 501(k)-exempt device. To be “substantially equivalent,” the proposed device must have the same intended use as the predicate device, and either have the same technological characteristics as the predicate device or have different technological characteristics and not raise different questions of safety or effectiveness than the predicate device. The FDA may require additional information, including clinical data, to make a determination regarding substantial equivalence. In addition, FDA collects user fees for certain medical device submissions and annual fees and for medical device establishments.
If the FDA agrees that the device is substantially equivalent to a predicate device currently on the market, it will grant 510(k) clearance to commercially market the device. If the FDA determines that the device is “not substantially equivalent” to a previously cleared device, the device is automatically designated as a Class III device. The device sponsor must then fulfill more rigorous PMA requirements, or can request a risk-based classification determination for the device in accordance with the de novo classification process.
35
After a device receives 510(k) clearance, any modification that could significantly affect its safety or effectiveness, or that would constitute a major change or modification in its intended use, will require a new 510(k) clearance or, depending on the modification, PMA approval. The FDA requires each manufacturer to determine whether the proposed change requires a new marketing submission in the first instance, but the FDA can review any such decision and disagree with a manufacturer’s determination. If the FDA disagrees with a manufacturer’s determination, the FDA can require the manufacturer to cease marketing and/or request the recall of the modified device until such marketing authorization has been granted. Also, in these circumstances, the manufacturer may be subject to significant regulatory fines or penalties.
PMA Approval Pathway
The PMA process is typically required for devices that are deemed to pose the greatest risk, such as life-sustaining, life-supporting, or implantable devices. Most Class III devices require PMA approval before they can be marketed, although some pre-amendment Class III devices for which FDA has not yet required a PMA are cleared through the 510(k) process. The PMA process is more demanding than the 510(k) premarket notification process. In a PMA, the manufacturer must demonstrate that the device is safe and effective, and the PMA must be supported by extensive data, including data from preclinical studies and human clinical trials. The PMA must also contain a full description of the device and its components, a full description of the methods, facilities, and controls used for manufacturing, and proposed labeling. Following receipt of a PMA, the FDA determines whether the application is sufficiently complete to permit a substantive review. If FDA accepts the application for review, it has 180 days under the FDCA to complete its review of a PMA, although in practice, the FDA’s review often takes significantly longer, and can take up to several years. An advisory panel of experts from outside the FDA may be convened to review and evaluate the application and provide recommendations to the FDA as to the approvability of the device. The FDA may or may not accept the panel’s recommendation. In addition, the FDA will generally conduct a pre-approval inspection of the applicant and/or its third-party manufacturers’ or suppliers’ manufacturing facility or facilities to ensure compliance with the QMSR.
The FDA will approve the new device for commercial distribution if it determines that the data and information in the PMA constitute valid scientific evidence and that there is reasonable assurance that the device is safe and effective for its intended use(s). The FDA may approve a PMA with post-approval conditions intended to ensure the safety and effectiveness of the device, including, among other things, restrictions on labeling, promotion, sale and distribution, and collection of long-term follow-up data from patients in the clinical study that supported PMA approval or requirements to conduct additional clinical studies post-approval. The FDA may condition PMA approval on some form of post-market surveillance when deemed necessary to protect the public health or to provide additional safety and efficacy data for the device in a larger population or for a longer period of use. In such cases, the manufacturer might be required to follow certain patient groups for a number of years and to make periodic reports to the FDA on the clinical status of those patients. Failure to comply with the conditions of approval can result in material adverse enforcement action, including withdrawal of the approval.
Certain changes to an approved device, such as changes in manufacturing facilities, methods, or quality control procedures, or changes in the design performance specifications, which affect the safety or effectiveness of the device, require submission of a PMA supplement. PMA supplements often require submission of the same types of information as a PMA, but are limited to information needed to support any changes from the device covered by the original PMA and may not require as extensive clinical data or the convening of an advisory panel. Certain other changes to an approved device require the submission of a new PMA, such as when the design change causes a different intended use, mode of operation, and technical basis of operation, or when the design change is so significant that a new generation of the device will be developed, and the data that were submitted with the original PMA are not applicable to demonstrate a reasonable assurance of safety and effectiveness for the modified device.
De Novo Classification
Medical device types that the FDA has not previously classified as Class I, II, or III are automatically classified into Class III regardless of the level of risk they pose. The Food and Drug Administration Modernization Act of 1997 established a route to market for low-to-moderate risk medical devices that are automatically placed into Class III due to the absence of a suitable predicate device, called the “Request for Evaluation of Automatic Class III Designation,” or the de novo classification procedure. This procedure allows a manufacturer whose novel device is automatically classified into Class III to request down-classification of its medical device into Class I or Class II on the basis that the device presents low to moderate risk, rather than requiring the submission and approval of a PMA application. Pursuant to the Food and Drug Administration Safety and Innovation Act (“FDASIA”), manufacturers may request de novo classification directly without first submitting a 510(k) notice and receiving a not-substantially-equivalent determination.
36
Under FDASIA, FDA is required to classify the device within 120 days following receipt of the de novo request, although the process may take significantly longer. A de novo request seeking reclassification into Class II must include a draft proposal for special controls that are necessary to provide a reasonable assurance of the safety and effectiveness of the medical device. If FDA grants the de novo request, the device may be legally marketed in the United States, and the FDA will issue a new classification regulation defining the new device type. This device type may be used subsequently as a predicate device for future 510(k) submissions. However, the FDA may reject the request if the FDA identifies a legally marketed predicate device that would be appropriate for a 510(k) notice, determines that the device is not low-to-moderate risk, or determines that General Controls would be inadequate to mitigate the risks and/or adequate special controls cannot be developed. After a device receives de novo classification, any modification that could significantly affect its safety or effectiveness, or that would constitute a major change or modification in its intended use, will require a new 510(k) clearance or, depending on the modification, another de novo request or even PMA approval.
Clinical Trials
Clinical trials are almost always required to support a PMA and de novo classification, and are sometimes required to support a 510(k) submission. All clinical investigations of devices to determine safety and effectiveness must be conducted in accordance with the FDA’s investigational device exemption (“IDE”) regulations, which govern investigational device labeling, prohibit promotion of the investigational device, and specify an array of recordkeeping, reporting and monitoring responsibilities of study sponsors and study investigators. If the device presents a “significant risk” to human health, as defined by the FDA in 21 C.F.R. § 812.3(m), the device sponsor must submit an IDE application to the FDA, which must become effective prior to commencing human clinical trials. If the device under evaluation does not present a significant risk to human health as defined by the FDA, then the IDE application is not required before initiating human clinical trials, but the sponsor must still comply with abbreviated IDE requirements when conducting such trials. An IDE application must be supported by appropriate data, such as animal and laboratory test results, showing that it is safe to test the device in humans and that the testing protocol is scientifically sound. The IDE will automatically become effective 30 days after receipt by the FDA unless the FDA notifies the company that the investigation may not begin. If the FDA determines that there are deficiencies or other concerns with an IDE for which it requires modification, the FDA may permit a clinical trial to proceed under a conditional approval.
Regardless of the degree of risk presented by the medical device, clinical studies must be approved by, and conducted under the oversight of, an Institutional Review Board (“IRB”) for each clinical site. The IRB is responsible for the initial and continuing review of any IDE, and may impose additional requirements for the conduct of the study. If an IDE application is approved by the FDA and one or more IRBs, human clinical trials may begin at a specific number of investigational sites with a specific number of patients, as approved by the FDA. If the device presents a non-significant risk, a sponsor may begin the clinical trial after obtaining approval for the trial by one or more IRBs without separate approval from the FDA, but must still follow abbreviated IDE requirements, such as monitoring the investigation, ensuring that the investigators obtain informed consent, and complying with labeling and record-keeping requirements. In some cases, an IDE supplement must be submitted to, and approved by, the FDA before a sponsor or investigator may make a change to the investigational plan that may affect its scientific soundness or the rights, safety or welfare of human subjects.
During a study, the sponsor is required to comply with the applicable FDA requirements, including, for example, trial monitoring, selecting clinical investigators and providing them with the investigational plan, ensuring IRB review, adverse event reporting, record keeping and prohibitions on the promotion of investigational devices or on making safety or effectiveness claims for them. The clinical investigators in the clinical study are also subject to FDA’s regulations and must obtain patient informed consent, rigorously follow the investigational plan and study protocol, control the disposition of the investigational device, and comply with all reporting and recordkeeping requirements. Additionally, after a trial begins, the sponsor, the FDA or the IRB could suspend or terminate a clinical trial at any time for various reasons, including a belief that the risks to study subjects outweigh the anticipated benefits.
Post-Market Regulation of Medical Devices
After a medical device obtains premarket authorization and is placed on the market, numerous regulatory requirements continue to apply. These requirements include:
•device listing and establishment registration, which helps facilitate FDA inspections and other regulatory action;
•the QMSR, which requires manufacturers, including third-party manufacturers, to follow stringent design, validation, testing, control, documentation and other quality assurance procedures during all aspects of the design and manufacturing process;
37
•labeling regulations and FDA prohibitions against the promotion of products for uncleared or unapproved uses or indications;
•obtaining a new marketing authorization prior to implementing modifications that could significantly affect the device’s safety or effectiveness or that would constitute a major change in intended use;
•medical device reporting regulations, which require that a manufacturer report to the FDA if a device it markets may have caused or contributed to a death or serious injury, or has malfunctioned and the device or a similar device that it markets would be likely to cause or contribute to a death or serious injury, if the malfunction were to recur;
•correction, removal and recall reporting regulations, which require that manufacturers report to the FDA field corrections and product recalls or removals if undertaken to reduce a risk to health posed by the device or to remedy a violation of the FDCA that may present a risk to health;
•regulations pertaining to voluntary recalls;
•the FDA’s recall authority, whereby it can ask, or under certain conditions order, manufacturers to recall from the market a device that is in violation of governing laws and regulations;
•post-market restrictions or conditions, including post-market study commitments; and
•post-market surveillance regulations, which apply, when necessary, to protect the public health or to provide additional safety and effectiveness data for the medical product.
Manufacturing processes for medical devices are required to comply with the applicable portions of the QMSR, which cover the methods and the facilities and controls for the design, manufacture, testing, production, processes, controls, quality assurance, labeling, packaging, distribution, installation and servicing of finished devices intended for human use. The QMSR also requires, among other things, maintenance of a device master file, design history file, and complaint files. Manufacturers are subject to periodic scheduled and unscheduled inspections by the FDA. Failure to maintain compliance with the QMSR requirements could result in the shut-down of, or restrictions on, manufacturing operations and the recall or seizure of marketed products. The discovery of previously unknown problems with any marketed products, including unanticipated adverse events or adverse events of increasing severity or frequency, whether resulting from the use of the device within the scope of its clearance or approval, or off-label in the practice of medicine, could result in restrictions on the device, including the removal of the product from the market or voluntary or mandatory device recalls.
The FDA has broad regulatory compliance and enforcement powers. If the FDA determines that a manufacturer has failed to comply with applicable regulatory requirements, it can take a variety of compliance or enforcement actions, which may result in any of the following sanctions:
•warning letters, untitled letters, fines, injunctions, consent decrees and civil penalties;
•recalls, withdrawals, or administrative detention or seizure of our products;
•operating restrictions or partial suspension or total shutdown of production;
•refusing or delaying requests for 510(k) clearance, de novo classification, or PMA approval of new or modified products;
•withdrawing 510(k) clearances, de novo classifications, or PMA approvals that have already been granted;
•refusal to grant export approvals for our products; or
•criminal prosecution.
Foreign Government Regulation
In addition to U.S. regulations, medical devices are subject to a variety of foreign government regulations.
38
Foreign Regulation of Medical Devices
Regulation of Medical Devices in the European Union
The EU has adopted specific directives and regulations regulating the design, manufacture, clinical investigation, conformity assessment, labeling and adverse event reporting for medical devices.
Until May 25, 2021, medical devices were regulated by the Council Directive 93/42/EEC (“EU Medical Devices Directive”), which has been repealed and replaced by Regulation (EU) No 2017/745 (“EU Medical Devices Regulation”). Unlike directives, regulations are directly applicable in all EU member states without the need for member states to implement into national law.
In the EU, there is currently no premarket government review of medical devices. However, all medical devices placed on the EU market must meet general safety and performance requirements, including the requirement that a medical device must be designed and manufactured in such a way that, during normal conditions of use, it is suitable for its intended purpose. Medical devices must be safe and effective and must not compromise the clinical condition or safety of patients, or the safety and health of users and – where applicable – other persons, provided that any risks which may be associated with their use constitute acceptable risks when weighed against the benefits to the patient and are compatible with a high level of protection of health and safety, taking into account the generally acknowledged state of the art.
Compliance with the general safety and performance requirements is a prerequisite for European conformity marking (“CE mark”), without which medical devices cannot be marketed or sold in the EU. To demonstrate compliance with the general safety and performance requirements medical device manufacturers must undergo a conformity assessment procedure, which varies according to the type of medical device and its (risk) classification. Except for low-risk medical devices (Class I), where the manufacturer can self-assess the conformity of its products with the general safety and performance requirements (except for any parts which relate to sterility, metrology or reuse aspects), a conformity assessment procedure requires the intervention of a notified body. Notified bodies are independent organizations designated by EU member states to assess the conformity of devices before being placed on the market. A notified body would typically audit and examine a product’s technical dossiers and the manufacturer’s quality system. If satisfied that the relevant product conforms to the relevant general safety and performance requirements, the notified body issues a certificate of conformity, which the manufacturer uses as a basis for its own declaration of conformity. The manufacturer may then apply the CE mark to the device, which allows the device to be placed on the market throughout the EU.
Throughout the term of the certificate of conformity, the manufacturer will be subject to periodic surveillance audits to verify continued compliance with the applicable requirements. In particular, there will be a new audit by the notified body before it will renew the relevant certificate(s).
All manufacturers placing medical devices into the market in the EU must comply with the EU medical device vigilance system. Under this system, serious incidents and Field Safety Corrective Actions (“FSCAs”), must be reported to the relevant authorities of the EU member states. Manufacturers are required to take FSCAs defined as any corrective action for technical or medical reasons to prevent or reduce a risk of a serious incident associated with the use of a medical device that is made available on the market. An FSCA may include the recall, modification, exchange, destruction or retrofitting of the device.
The aforementioned EU rules are generally applicable in the EEA, which consists of the 27 EU Member States plus Norway, Liechtenstein and Iceland.
Regulation of Medical Devices in the United Kingdom
The EU Medical Devices Regulation is not applicable in Great Britain due to Brexit. Existing EU directives governing all medical devices have been given effect in domestic law through the Medical Devices Regulations 2002 (SI 2002 No 618, as amended) (“UK Medical Devices Regulations”). This means that since January 1, 2021, the Great Britain route to market is still based on the requirements derived from the pre-existing EU legislation. As a standalone regulatory body, the Medicines and Healthcare products Regulatory Agency (“MHRA”) is responsible for regulating medical devices in Great Britain (England, Scotland and Wales) and Northern Ireland, though Northern Ireland is aligned with EU legislation and regulations regarding medical devices as a result of the Northern Ireland Protocol that took effect in January 2021. The UK government has passed the Medicines and Medical Devices Act which came into force on 11 February 2021 and which allows the secretary of state or an ‘appropriate authority’ to amend or supplement existing regulations in the area of medical devices including the UK Medical Devices Regulations. In addition, the Trade Deal between the UK and the EU generally provides for cooperation and exchange of information between the parties in the areas of product safety and
39
compliance, including market surveillance, enforcement activities and measures, standardization-related activities, exchanges of officials, and coordinated product recalls. As such, processes for compliance and reporting should reflect requirements from regulatory authorities.
Under the UK Medical Devices Regulations, in order to be lawfully placed on the Great Britain market, certain medical devices need to be “UKCA” certified by a UK approved body. However, certain medical devices in compliance with: (1) the EU Medical Devices Directive can continue to be placed on the Great Britain market until the sooner of certificate expiration or June 30, 2028; or (2) the EU Medical Devices Regulation can continue to be placed on the Great Britain market until the sooner of certificate expiration or June 30, 2030. The EU no longer recognizes conformity assessment activities performed by UK notified bodies for medical devices placed on the market since January 1, 2021. Notified bodies must be located in an EU member state, or territory where there is a Mutual Recognition Agreement (“MRA”), that allows the marketing of medical devices that meet EU requirements, there is (currently) no such MRA between the UK and the EU.
Furthermore, on December 16, 2024, the UK government published an amendment to UK Medical Devices Regulations to clarify and strengthen the post-market surveillance requirements for medical devices in Great Britain. This amendment will come into force on June 16, 2025 and aims to facilitate greater traceability of incidents and trends enabling the MHRA to act swiftly when needed to address safety issues and support the entire health system in better protecting patients. In addition, the MHRA launched a consultation from November 14, 2024 to January 5, 2025 on proposals to update the pre-market requirements for medical devices in Great Britain, covering four topics, namely: (1) a new international reliance scheme to enable swifter market access for certain devices that have already been approved in a comparable regulator country; (2) the new UK Conformity Assessed (“UKCA”) mark and, in particular, proposals to remove the requirement to place such UKCA marking on devices; (3) conformity assessment procedures for in vitro diagnostic devices; and (4) maintaining in UK law certain pieces of “assimilated” EU law which are due to sunset in 2025. This consultation builds on the MHRA’s previous consultation between September and November 2021, and the UK government’s response to that consultation which was published on June 26, 2022. The MHRA has stated that it will incorporate feedback to its recent consultation into new legislation on pre-market requirements for medical devices in Great Britain. The new legislation is expected to be implemented in 2026 and aims to enable greater international collaboration and practices, with more patient-centered, proportionate requirements for medical devices which are responsive to technological advances.
Coverage and Reimbursement
In the United States, our currently authorized products are commonly treated as general supplies utilized in orthopedic surgery and if covered by third-party payors, are paid for as part of the surgical procedure. Our commercial success depends in part on the extent to which governmental authorities, private health insurers and other third-party payors provide coverage for and establish adequate reimbursement levels for the procedures during which our products are used. Failure by physicians, hospitals, ASCs and other users of our products to obtain sufficient coverage and reimbursement from third-party payors for procedures in which our products are used, or adverse changes in government and private third-party payors’ coverage and reimbursement policies, could directly impact the demand for our products.
Based on our experience to date, third-party payors generally reimburse for the surgical procedures in which our products are used only if the patient meets the established medical necessity criteria for surgery. Some payors are moving toward a managed care system and control their healthcare costs by limiting authorizations for surgical procedures, including elective procedures using our devices. Although no uniform policy of coverage and reimbursement among payors in the United States exists and coverage and reimbursement for procedures can differ significantly from payor to payor, reimbursement decisions by particular third-party payors may depend upon a number of factors, including the payor’s determination that use of a product is:
•a covered benefit under its health plan;
•appropriate and medically necessary for the specific indication;
•cost effective; and
•neither experimental nor investigational.
Third-party payors are increasingly auditing and challenging the prices charged for medical products and services with concern for upcoding, miscoding, using inappropriate modifiers, or billing for inappropriate care settings. Some third-party payors must approve coverage for new or innovative devices or procedures before they will reimburse healthcare providers
40
who use the products or therapies. Even though a new product may have been cleared for commercial distribution by the FDA, we may find limited demand for the product unless and until reimbursement approval has been obtained from governmental and private third-party payors.
We believe the overall escalating cost of medical products and services being paid for by the government and private health insurance has led to, and will continue to lead to, increased pressures on the healthcare and medical device industry to reduce the costs of products and services. All third-party reimbursement programs are developing increasingly sophisticated methods of controlling healthcare costs through prospective reimbursement and capitation programs, group purchasing, redesign of benefits, requiring second opinions prior to major surgery, careful review of bills, encouragement of healthier lifestyles and other preventative services and exploration of more cost-effective methods of delivering healthcare.
In addition to uncertainties surrounding coverage policies, there are periodic changes to reimbursement levels. Third-party payors regularly update reimbursement amounts and also from time to time revise the methodologies used to determine reimbursement amounts. This includes routine updates to payments to physicians, hospitals and ASCs for procedures during which our products are used. These updates could directly impact the demand for our products.
Other U.S. Healthcare Laws
Device manufacturers are subject to additional healthcare regulation and enforcement by the federal government and by authorities in the states and foreign jurisdictions in which they conduct their business. Such laws include, without limitation, U.S. federal and state anti-kickback, fraud and abuse, false claims, consumer fraud, and transparency laws and regulations.
For example, the federal Anti-Kickback Statute prohibits, among other things, individuals or entities from knowingly and willfully offering, paying, soliciting or receiving remuneration, directly or indirectly, overtly or covertly, in cash or in kind to induce or in return for purchasing, leasing, ordering or arranging for or recommending the purchase, lease or order of any item or service reimbursable under Medicare, Medicaid or other federal healthcare programs. A person or entity does not need to have actual knowledge of this statute or specific intent to violate it in order to have committed a violation.
The federal civil and criminal false claims laws, including the civil False Claims Act, prohibit, among other things, any individual or entity from knowingly presenting, or causing to be presented, a false claim for payment to the federal government or knowingly making, using or causing to be made or used a false record or statement material to a false or fraudulent claim to the federal government. In addition, the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act.
The Civil Monetary Penalty Laws impose penalties against any person or entity that, among other things, is determined to have presented or caused to be presented a claim to a federal healthcare program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent, or offering or transferring remuneration to a federal healthcare beneficiary that a person knows or should know is likely to influence the beneficiary’s decision to order or receive items or services reimbursable by the government from a particular provider or supplier.
HIPAA created additional federal civil and criminal statutes that prohibit, among other things, knowingly and willfully executing a scheme to defraud any healthcare benefit program. Similar to the federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the healthcare fraud statute implemented under HIPAA or specific intent to violate it in order to have committed a violation.
The federal Physician Payments Sunshine Act requires certain manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program, with specific exceptions, to report annually to the CMS information related to payments or other transfers of value made to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors), certain non-physician practitioners (defined to include physician assistants, nurse practitioners, clinical nurse specialists, certified nurse anesthetists, anesthesiology assistants and certified nurse midwives) and teaching hospitals, and further requires applicable manufacturers and applicable group purchasing organizations to report annually to CMS ownership and investment interests held by physicians and their immediate family members.
Similar state and local laws and regulations may also restrict business practices in the medical device industries, such as state anti-kickback and false claims laws, which may apply to business practices, including but not limited to, research, distribution, sales and marketing arrangements and claims involving healthcare items or services reimbursed by non-
41
government third-party payors, including private insurers, or by patients themselves; state laws that require device companies to comply with the industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government, or otherwise restrict payments that may be made to healthcare providers and other potential referral sources; state laws and regulations that require manufacturers to file reports relating to pricing and marketing information or which require tracking gifts and other remuneration and items of value provided to physicians, other healthcare providers and entities; state laws that prohibit fee-splitting arrangements between companies and physicians and other healthcare professionals; and state and local laws that require the registration of sales representatives.
Violation of any of such laws or any other government regulations that apply may result in penalties, including, without limitation, civil and criminal penalties, damages, fines, additional reporting obligations, the curtailment or restructuring of operations, exclusion from participation in government healthcare programs and individual imprisonment.
Healthcare Reform
The United States is considering or has enacted a number of legislative and regulatory proposals to change the healthcare system in ways that could affect our ability to sell our products profitably. Among policy makers and payors in the United States, there is significant interest in promoting changes in healthcare systems with the stated goals of containing healthcare costs, improving quality or expanding access. Current and future legislative proposals to further reform healthcare or reduce healthcare costs may limit coverage of or lower reimbursement for the procedures associated with the use of our products. The cost containment measures that payors and providers are instituting and the effect of any healthcare reform initiative implemented in the future could impact our revenue from the sale of our products.
The implementation of the ACA in the United States, for example, has changed healthcare financing and delivery by both governmental and private insurers substantially, and affected medical device manufacturers significantly. The ACA, among other things, provided incentives to programs that increase the federal government’s comparative effectiveness research, and implemented payment system reforms including a national pilot program on payment bundling to encourage hospitals, physicians and other providers to improve the coordination, quality and efficiency of certain healthcare services through bundled payment models. Additionally, the ACA has expanded eligibility criteria for Medicaid programs and created a new Patient-Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research.
In addition, other legislative changes have been proposed and adopted since the ACA was enacted that tighten margins and intensify burdens for healthcare providers that use our products. For example, the Budget Control Act of 2011, among other things, reduced Medicare payments to providers, effective on April 1, 2013 and, due to subsequent legislative amendments to the statute, will remain in effect through 2032 unless additional Congressional action is taken. Additionally, the American Taxpayer Relief Act of 2012, among other things, reduced Medicare payments to several providers, including hospitals, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years.
The Medicare Access and CHIP Reauthorization Act of 2015 repealed the formula by which Medicare made annual payment adjustments to physicians and replaced the former formula with fixed annual updates and a new system of incentive payments which began in 2019 that are based on various performance measures and physicians’ participation in alternative payment models, such as accountable care organizations. Each year, CMS updates Medicare payments for physician services through rulemaking, based on parameters established under law. In November 2024, CMS finalized a 2.83% decrease in the physician fee schedule conversion factor, a key aspect of physician payment rates under the Medicare program. This resulted in an average payment cut of 2.93% to physicians and other clinicians, which took effect on January 1, 2025 and remains in effect today, unless Congress takes additional action.
We expect additional state and federal healthcare reform measures to be adopted in the future, particularly given the recent change in administration, any of which could limit the amounts that federal and state governments will pay for healthcare products and services, which could result in reduced demand for our products or additional pricing pressure.
Data Privacy and Security Laws
Numerous state, federal and foreign laws, regulations and standards govern the collection, use, access to, confidentiality and security of health-related and other personal information, and could apply now or in the future to our operations or the operations of our partners. In the United States, numerous federal and state laws and regulations, including data breach notification laws, health information privacy and security laws and consumer protection laws and regulations govern the collection, use, disclosure, and protection of health-related and other personal information. In addition, certain foreign laws govern the privacy and security of personal data, including health-related data. Privacy and
42
security laws, regulations, and other obligations are constantly evolving, may conflict with each other to complicate compliance efforts, and can result in investigations, proceedings, or actions that lead to significant civil and/or criminal penalties and restrictions on data processing.
Employees and Human Capital Resources
As of December 31, 2025, we had 75 employees, 72 of which were full-time. None of our employees are represented by a labor union or party to a collective bargaining agreement. We consider our relationship with our employees to be good.
Our human capital resources objectives include, as applicable, identifying, recruiting, retaining, incentivizing, and integrating our existing and additional employees. The principal purposes of our equity incentive plans are to attract, retain, and motivate selected employees, consultants, and directors through the granting of stock-based compensation awards and cash-based performance bonus awards.
Corporate Information
Shoulder Innovations was originally incorporated under the laws of the State of Delaware in October 2014. Our principal executive offices are located at 1535 Steele Avenue SW, Suite B, Grand Rapids, Michigan 49507 and our telephone number is (616) 294-1026. Our website address is https://shoulderinnovations.com/. Our website also contains copies of our Corporate Governance Guidelines and Code of Ethics and Conduct, as well as the charters of our Audit Committee, Compensation Committee and Nominating and Corporate Governance Committee. Within the time period required by the SEC, we will post on our website any amendment to our Corporate Governance Guidelines and Code of Ethics and Conduct and any waiver applicable to our principal executive officer, principal financial officer, principal accounting officer or controller or persons performing similar functions. The information contained on, or that can be accessed through, our website is not incorporated by reference into, and is not a part of, this Annual Report. Investors should not rely on any such information in deciding whether to purchase our common stock.
Available Information
Our reports filed with or furnished to the Securities and Exchange Commission (the “SEC”) pursuant to Sections 13(a) and 15(d) of the Exchange Act, are available, free of charge, on our investor relations website at https://ir.shoulderinnovations.com/ as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC. The SEC maintains a website at http://www.sec.gov that contains reports, proxy and information statements, and other information regarding us and other companies that file materials with the SEC electronically. Copies of our reports on Form 10-K, Forms 10-Q, and Forms 8-K, and amendments to those reports may also be obtained, free of charge, electronically through our investor relations website located at the web address appearing above as soon as reasonably practicable after we file such material with, or furnish it to, the SEC. We use our investor relations website as a means of disclosing material information and for complying with our disclosure obligations under Regulation FD. Accordingly, investors should monitor our investor relations website, in addition to following our press releases, SEC filings, and public conference calls and webcasts.
The content of our websites and information that we may post on or provide to online and social media channels and information that can be accessed through our websites or these online and social media channels are not incorporated by reference into this Annual Report or in any other report or document we file with the SEC, and any references to our websites or these online and social media channels are intended to be inactive textual references only.
43
Item 1A. Risk Factors
Our business involves a high degree of risk. The risks and uncertainties described below are not the only ones we face. If any of the following risks and uncertainties develop into actual events or circumstances, they could have a material adverse effect on our business, financial condition and results of operations. In that event, the market price of our common stock could decline, and you could lose all your investments. You should carefully read the following risk factors, together with our audited financial statements and the related notes and all other information included in this Annual Report and our other filings with the SEC.
Risks Related to Our Financial Condition and Capital Requirements
We are an early-stage company with a history of significant net losses, we expect to incur operating losses in the future and we may not be able to achieve or sustain profitability.
We have incurred annual net losses since our inception, and we expect to incur additional substantial losses in the foreseeable future. For the year ended December 31, 2025 and 2024, we had a net loss of $40,359 thousand and $15,619 thousand, respectively. As of December 31, 2025, we had an accumulated deficit of $97,400 thousand. To date, we have financed our operations primarily through equity and debt financings and from sales of our shoulder replacement implants. The losses and accumulated deficit have primarily been due to the substantial investments we have made to develop our implant systems, costs related to our sales and marketing efforts and general research and development expenses, including costs related to regulatory initiatives to obtain marketing approval. These investments may be more costly than we expect, and if we do not achieve the benefits anticipated from these investments, or if the realization of these benefits is delayed or not enough to offset our higher operating expenses, such investments may not result in increased revenue or growth in our business, which will lead to increased net losses. In addition, as a public company we incur significant legal, accounting and other expenses that we did not incur as a private company. Accordingly, we expect to continue to incur operating losses for the foreseeable future. Our failure to achieve and sustain profitability in the future will make it more difficult to finance our business and accomplish our strategic objectives, which would have a material adverse effect on our business, financial condition and results of operations and cause the market price of our common stock to decline. In addition, failure of our solutions to significantly penetrate the target markets would negatively affect our business, financial condition and results of operations.
If we fail to manage our growth effectively, our business could be materially and adversely affected.
We have experienced recent rapid growth and anticipate further growth in the future. This growth has placed significant demands on our management, financial, operational, technological and other resources, and we expect that our future growth will continue to place significant demands on our management and other resources and will require us to continue developing and improving our operational, financial and other internal controls. In particular, continued growth increases the challenges involved in a number of areas, including managing our supplier relationships and ensuring adequate inventory is available, recruiting and retaining sufficient skilled personnel for our dedicated commercial leadership team, contracting with independent distributors to market our implant systems, providing adequate training and supervision to maintain our high-quality standards and preserving our culture and values. We may not be able to address these challenges in a cost-effective manner, or at all. To achieve our revenue goals, we must also successfully increase our supply of implants and other products, components and instruments from third-party manufacturers to meet expected customer demand. In the future, we may experience difficulties with quality control, component supply and shortages of qualified personnel, among other problems. These problems could result in delays in product availability and increases in expenses. Any such delay or increased expense could adversely affect our business, financial condition and results of operations. In addition, rapid and significant growth will place a strain on our administrative and operational infrastructure. In order to manage our operations and growth, we will need to continue to improve our operational and management controls, hiring process, reporting and information technology systems and financial internal control procedures. If we do not effectively manage our growth, we may not be able to execute on our business plan, respond to competitive pressures, take advantage of market opportunities, satisfy customer requirements or maintain high-quality product offerings, which could have a material adverse effect on our business, financial condition and results of operations.
We have a significant amount of debt, which may affect our ability to operate our business and secure additional financing in the future.
As of December 31, 2025, we had an aggregate of approximately $15,000 thousand in principal outstanding under Trinity Loan Agreement. The Trinity Loan Agreement, which we entered into in August 2023, as amended by the First Amendment to the Loan and Security Agreement on April 23, 2024 and the Second Amendment to the Loan and Security Agreement on July 21, 2025, consists of term loans of up to $45,000 thousand and bears interest at an annual rate equal to
44
the greater of the prime rate plus 3.50% and 11.00%. Under the Trinity Loan Agreement, the prime rate is equal to the greater of 8.0% per year and the prime rate as reported in The Wall Street Journal. We must make interest payments under the Trinity Loan Agreement, which has diverted and will continue to divert resources from other activities. For the year ended December 31, 2025, we incurred interest expense, net of $70 thousand, which included payments made under the Trinity Loan Agreement. Our obligations under the Trinity Loan Agreement are collateralized by substantially all of our assets, including intellectual property, and we are subject to customary affirmative and negative covenants limiting our ability to, among other things, relocate or dispose of assets, undergo a change in control, merge or consolidate, enter into certain transactions with affiliates, make acquisitions, incur debt, pay dividends, grant liens, store certain amounts of inventory or equipment with third parties and make investments, in each case subject to certain exceptions.
The covenants related to the Trinity Loan Agreement, as well as any future financing agreements into which we may enter, may restrict our ability to finance our operations and engage in, expand or otherwise pursue our business activities and strategies.
While we have not previously breached and are not currently in breach of these or any other covenants contained in our Trinity Loan Agreement or other debt arrangements, there can be no guarantee that we will not breach these covenants in the future. Our ability to comply with these covenants may be affected by events beyond our control, and future breaches of any of these covenants could result in a default under the Trinity Loan Agreement. If not waived, future defaults could cause all of the outstanding indebtedness under the Trinity Loan Agreement to become immediately due and payable and terminate commitments to extend further credit and foreclose on the collateral granted to it to collateralize such indebtedness. If we do not have or are unable to generate sufficient cash available to repay our debt obligations when they become due and payable, either upon maturity or in the event of a default, our assets could be foreclosed upon and we may not be able to obtain additional debt or equity financing on favorable terms, if at all, which may negatively impact our ability to operate and continue our business as a going concern.
If we default under the Trinity Loan Agreement, Trinity Capital on behalf of the lenders will be able to declare all obligations immediately due and payable and take control of our pledged assets, potentially requiring us to renegotiate our agreement on terms less favorable to us or to immediately cease operations. Further, if we are liquidated, the lenders’ rights to repayment would be senior to the rights of the holders of our common shares to receive any proceeds from the liquidation. The lenders could declare a default under the Trinity Loan Agreement upon the occurrence of specific events such as our failure to pay or our failure to comply with specified covenants, thereby requiring us to repay the loan immediately. Any declaration by the lenders of an event of default could significantly harm our business and prospects and could cause the price of our common shares to decline.
In order to service this indebtedness and any additional indebtedness we may incur in the future, we need to generate cash from our operating activities. Our ability to generate cash is subject, in part, to our ability to successfully execute our business strategy, as well as general economic, financial, competitive, regulatory and other factors beyond our control. We cannot assure you that our business will be able to generate sufficient cash flow from operations or that future borrowings or other financings will be available to us in an amount sufficient to enable us to service our indebtedness and fund our other liquidity needs. To the extent we are required to use cash from operations or the proceeds of any future financing to service our indebtedness instead of funding working capital, capital expenditures or other general corporate purposes, we will be less able to plan for, or react to, changes in our business, industry and in the economy generally. This may place us at a competitive disadvantage compared to our competitors that have less indebtedness.
Also, the anticipated benefit of any strategic alliance, joint venture or acquisition may not materialize or such strategic alliance, joint venture or acquisition may be prohibited. The Trinity Loan Agreement restricts our ability to pursue certain mergers, acquisitions, amalgamations or consolidations that we may believe to be in our best interest. Additionally, future acquisitions or dispositions could result in potentially dilutive issuances of our equity securities, the incurrence of debt, contingent liabilities or amortization expenses or write-offs of goodwill, any of which could harm our financial condition. We cannot predict the number, timing or size of future joint ventures or acquisitions, or the effect that any such transactions might have on our operating results.
We may acquire other complementary companies, products or technologies, through the licensing of products or technologies from third parties or other strategic alliances, and the failure to manage acquisitions, investments, licenses or other strategic alliances, or the failure to integrate them with our existing business could fail to result in a
45
commercial product or net sales, divert our management’s attention, result in additional dilution to our stockholders, cause us to incur significant expense and otherwise disrupt our business.
Our success depends on our ability to continually enhance and broaden our system offerings in response to changing surgeon and patient needs, competitive technologies and market pressures. Accordingly, from time to time we may consider opportunities to acquire, make investments in or license other technologies, products and businesses that may enhance our capabilities, complement our existing products and technologies or expand the breadth of our markets or customer base. Although we currently have no agreements or commitments to complete any such transactions, we may in the future seek to acquire or invest in businesses, applications or technologies that we believe could complement or expand our portfolio, enhance our technical capabilities or otherwise offer growth opportunities. However, we cannot assure you that we would be able to successfully complete any acquisition we choose to pursue on favorable terms, if at all, or that we would be able to successfully integrate any acquired business, product or technology in a cost-effective and non-disruptive manner. Potential and completed acquisitions, strategic investments, licenses and other alliances involve numerous risks, including:
•difficulty assimilating or integrating acquired or licensed technologies, products, employees or business operations;
•issues maintaining uniform standards, procedures, controls and policies;
•unanticipated costs associated with acquisitions or strategic alliances, including the assumption of unknown or contingent liabilities and the incurrence of debt or future write-offs of intangible assets or goodwill;
•diversion of management’s attention from our core business and disruption of ongoing operations;
•adverse effects on existing business relationships with suppliers, independent distributors, health care facilities, shoulder specialists and other health care providers;
•risks associated with entering new markets in which we have limited or no experience;
•potential losses related to investments in other companies;
•potential loss of key employees of acquired businesses;
•unanticipated or undisclosed liabilities of any target; and
•increased legal and accounting costs relating to the acquisitions or compliance with regulatory matters.
If we do acquire, make investments in or license other technologies, products and businesses, we may not ultimately obtain the expected benefits of such acquisition or investment, strengthen our competitive position or achieve our goals, and any acquisitions we complete could be viewed negatively by our customers, investors and industry analysts.
To date, the growth of our operations has been wholly organic, and we have limited experience in acquiring other businesses or technologies. We do not know if we will be able to identify acquisitions or strategic relationships we deem suitable, whether we will be able to successfully complete any such transactions on favorable terms, if at all, or whether we will be able to successfully integrate any acquired personnel, operations and technologies, or effectively manage the combined business following an acquisition. Our ability to successfully grow through strategic transactions depends upon our ability to identify, negotiate, complete and integrate suitable target businesses, technologies or products and to obtain any necessary financing. Acquisitions could also result in dilutive issuances of equity securities, the use of our available cash, or the incurrence of debt, which could harm our operating results. To finance any acquisitions, investments or strategic alliances, we may choose to issue shares of our common stock as consideration, which could dilute the ownership of our stockholders. If the price of our common stock is low or volatile, we may be unable to consummate any acquisitions, investments or strategic alliances using our common stock as consideration. Additional funds may not be available on terms that are favorable to us, or at all. In addition, if an acquired business fails to meet our expectations, our business, financial condition and results of operations may be negatively affected. These efforts could be expensive and time-consuming and may disrupt our ongoing business and prevent management from focusing on our operations.
46
We may require substantial additional funding, which may not be available to us on acceptable terms, or at all, and, if not available, may require us to delay, scale back, or cease our innovation efforts or operations.
We may require additional capital in the future to maintain and expand our operations. Our operations are capital-intensive and are expected to increase as we expand our dedicated commercial leadership team, research and development efforts and product offerings, among other growth initiatives. Changing circumstances could result in lower revenues or cause us to consume capital significantly faster than we currently anticipate, and we may need to raise capital sooner or in greater amounts than currently expected because of circumstances beyond our control. If we raise additional funds through the issuance of equity, equity-linked or debt securities, those securities may have rights, preferences or privileges senior to those of our common stock, and our existing stockholders may experience dilution. Any debt financing secured by us in the future could require that a substantial portion of our operating cash flow be devoted to the payment of interest and principal on such indebtedness, which may decrease available funds for other business activities, and could involve restrictive covenants relating to our capital-raising activities and other financial and operational matters, which may make it more difficult for us to obtain additional capital and to pursue business opportunities. Additionally, any future collaborations we enter into with third parties may provide capital in the near term but limit our potential cash flow and revenue in the future. If we raise additional funds through strategic partnerships and alliances and licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, intellectual property or products, or grant licenses on terms unfavorable to us. We cannot be certain that we will be able to obtain additional financing on favorable terms, if at all. If we cannot raise funds on acceptable terms, if and when needed, we may not be able to continue as a going concern or we may not be able to grow our business or respond to competitive pressures or unanticipated requirements, which could seriously harm our business.
Risks Related to Our Business and Industry
We operate in a very competitive business environment, and if we are unable to compete successfully against our existing or potential competitors, our business, financial condition and results of operations may be adversely affected.
Our existing implant systems are, and any new products or procedures we develop and commercialize will be, subject to intense competition. There are a number of competing products that can be used in shoulder arthroplasty procedures. The industry in which we operate is competitive, subject to change and sensitive to the introduction of new products, procedures or other market activities of industry participants. Our ability to compete successfully will depend on our ability to continue to train surgeons on the use of our implant systems and other technologies for shoulder surgical care and gain their acceptance of our systems or ecosystem, develop additional products to improve shoulder surgical care and expand our offerings that reach the market in a timely manner, receive adequate coverage and reimbursement from third-party payors and provide systems, technologies and instrumentation that are easier to use, safer, less invasive and perceived to be more effective than the products of our competitors. In addition, our ability to increase our customer base and achieve broader market acceptance of our products will depend to a significant extent on our ability to expand our marketing efforts. We plan to dedicate significant resources to our marketing and surgeon-to-surgeon collaboration programs, but if our marketing efforts and expenditures do not generate a corresponding increase in net revenue, it will negatively affect our business, financial condition and results of operations. In addition, we believe that developing and maintaining broad awareness of our systems in a cost-effective manner is critical to achieving broad acceptance and expanding our business in our target markets. Promotion activities may not generate sufficient or any awareness or increase net revenue, and even if they do, any increase in net revenue may not offset the costs and expenses we incur in building our brand and conducting such promotion activities. If we fail to successfully promote, maintain and protect our brand, we may fail to attract or retain the physician acceptance necessary to realize a sufficient return on our brand building efforts, or to achieve a level of brand awareness that supports broad adoption of our systems.
We compete with large, multinational companies, as well as with smaller orthopedic companies that offer innovations and improvements on traditional shoulder implants and technologies. For example, we compete with Arthrex, Enovis, Exectech, Johnson & Johnson, Smith & Nephew, Stryker, and Zimmer Biomet. Furthermore, conditions in our market could change rapidly and significantly as a result of technological advancements, partnerships, or acquisitions by competitors. We have seen and continue to see consolidation amongst our competitors. If our competitors have greater resources and access to funding than us, they may be able to finance the development of new technologies and products before we are able to do so, which may allow them to gain market share or enter new markets before us or provide lower-priced or better-quality offerings.
At any time, these competitors and other potential market entrants may develop new products, procedures or treatment alternatives that could render our products obsolete, uncompetitive or economically unviable. In addition, one or more of such competitors may gain a market advantage by developing and patenting competitive products, procedures or treatment
47
alternatives earlier than we can, obtaining regulatory clearances or approvals more rapidly than we can or selling competitive products at prices lower than ours. If medical research were to lead to the discovery of alternative therapies or technologies that improve or cure certain shoulder diseases, including through the use of pharmaceuticals or breakthrough bio-technological innovations or therapies, our profitability could suffer through a reduction in sales or a loss in market share to a competitor. The discovery of methods of prevention or the development of other alternatives to our systems, or novel methods of conducting shoulder arthroplasty or other procedures, could result in decreased demand for shoulder arthroplasty procedures and our systems, and, accordingly, could have a material adverse effect on our business, financial condition and results of operations. Many of our current and potential competitors have substantially greater sales and financial resources than we do. These competitors may also have more established distribution networks, a broader offering of products, entrenched relationships with surgeons and distributors or greater experience in launching, marketing, distributing and selling products or treatment alternatives.
We also compete with our competitors to engage the services of independent distributors, both those presently working with us and those with whom we hope to work with as we expand. These independent distributors may also sell shoulder care products that our competitors offer. In addition, we compete with our competitors in acquiring technologies and technology licenses complementary to our products or procedures or advantageous to our business. If we are unable to compete successfully against our existing or potential competitors, our business, financial condition and results of operations may be adversely affected, and we may not be able to grow at our expected rate, if at all.
If we fail to develop and retain an effective commercial organization, or if we are unable to successfully expand our organization, it could negatively impact our sales, and we may not generate sufficient net revenue to sustain profitability.
Our net revenue and profitability is directly dependent upon the sales and marketing efforts of our commercial organization and the effectiveness of our CEME team. Our commercial organization is comprised of a team of dedicated commercial leaders, our CEME team and a network of independent distributors. In order to expand our business, we plan to expand our dedicated commercial leadership team with individuals who have strong technical backgrounds specializing in sales and marketing of products for shoulder arthroplasty procedures and within the shoulder surgical care market. As we increase our marketing efforts, we will need to retain, develop and grow the number of team members that we employ. We intend to invest in recruiting and training dedicated commercial leadership team members and clinical representatives as we expand our business. There is significant competition for personnel experienced in relevant medical device sales and in shoulder surgical care in particular. Once hired, the training process can be lengthy because it requires significant education for new dedicated commercial leadership team members and clinical specialists to achieve the level of clinical competency and expertise that is expected by surgeons that use our systems. In addition, team members typically require at least one year if not longer in the field to grow their network of accounts and achieve the productivity levels we expect them to reach in any individual territory. Our future success will depend largely on our ability to continue to hire, train, retain and motivate skilled members of our commercial organization with significant technical knowledge. If we are unable to attract, motivate, develop and retain a sufficient number of such team members, or if these team members do not achieve the productivity levels we expect them to reach, our net revenue will not grow at the rate we expect and our financial performance will suffer. Also, to the extent we hire personnel from our competitors, we may have to wait until applicable non-competition provisions have expired before deploying such personnel in restricted territories or incur costs to relocate personnel outside of such territories, and we have been in the past, and may be subject to future allegations that these new hires have been improperly solicited, and that they have divulged to us proprietary or other confidential information of their former employers. Additionally, because the market for our target team members is competitive, our competitors may try to hire our team members from us. If successful, we would be required to dedicate resources to recruiting, filling and training those vacant positions. Any of these risks may adversely affect our business.
We rely in part on independent distributors to sell our products to our customers, and if we are unable to maintain and expand our network of independent distributors, we may be unable to generate anticipated sales.
Within our commercial organization and as part of our commercial approach, we use independent distributors to sell our implant systems to hospitals, ASCs and other end users and to assist us in promoting market acceptance of, and creating demand for, our systems. If we are unable to come to commercially reasonable terms with a distributor and/or its agents, we may not generate the expected level of sales and may need to spend more of our capital resources to hire sales personnel as employees or to engage one or more alternative distributors in a particular region or market. In addition, our independent distributors that we contract with are not all exclusive to us and there is a risk that an independent distributor will give higher priority to the products of other medical device companies, including products directly competitive with our systems or may be required by larger medical devices companies to stop offering our systems. Though we have established initiatives to further focus our independent distributor channel on our systems, these initiatives may not
48
translate to an increase in sales or penetration or may underperform relative to our expectations. In addition, we may not have insight into competitive lines at our distributors, including other commercial arrangements between such distributors and our competitors to sell our competitor’s products. As such, there can be no assurance that an agent will devote the resources necessary to provide effective sales and promotional support to our systems. In addition, if an agent terminates its relationship with us and is retained by one of our competitors, we may be unable to prevent them from helping competitors solicit business from our existing customers and surgeons, which could adversely affect our sales and growth. In addition to expanding our internal commercial team members, we expect to continue to utilize and expand our relationships with independent distributors and agents to meet our growing business needs and accomplish our business strategies.
Our business plan relies on certain assumptions about the market for our implant systems, however, the size and expected growth of our addressable market has not been established with precision and may be smaller than we estimate, and even if the addressable market is as large as we have estimated, we may not be able to capture additional market share.
Our estimates of our addressable market are based on publicly-available information, a number of internal and third-party estimates and assumptions, including the prevalence of aTSA and rTSA procedures and management’s knowledge and experience in the shoulder surgical care market. For example, we believe that the aging of the general population and increasingly active lifestyles will continue and that these trends will increase the demand for effective devices and implant systems to be used in connection with shoulder arthroplasty procedures. Furthermore, we currently only operate in the United States, which is the largest market for shoulder arthroplasties, and any contraction in this market or incorrect assumptions about the current and future development of the market would have a material impact on our results of operations and financial condition. While we believe our assumptions and the data underlying our estimates are reasonable, these assumptions and our estimates may not be correct. The projected growth of annual shoulder arthroplasty procedures, including in ASCs, and demand for our systems could materially differ from actual demand if our assumptions regarding these trends and acceptance of our systems by the medical community prove to be incorrect or do not materialize, or if non-surgical treatments or other surgical techniques gain more widespread acceptance as a viable alternative to our systems or products. In addition, even if the number of people who undergo shoulder arthroplasty procedures increases as we expect, technological or medical advances could provide alternatives to address shoulder conditions and reduce demand for shoulder arthroplasty procedures or for our particular systems. As a result, our estimates of the addressable market for our current or future products may prove to be incorrect. Even if the total addressable market for our current and future products is as large as we have estimated, we may not be able to penetrate this market to capture additional market share for the reasons discussed in this “Risk Factors” section. The estimated market opportunities included in this Annual Report represent the total overall revenue opportunity that we believe is available for our systems if 100% market share is achieved by us, and are not a representation that we will achieve any such market share. If the actual number of people who suffer from shoulder conditions who would benefit from shoulder arthroplasty procedures with our systems, the price at which we can sell our current and future products or the addressable market for our systems and future products is smaller than we estimate, or if the total addressable market is as large as we have estimated but we are unable to capture additional market share, it could have a material adverse effect on our business, financial condition and results of operations.
Our business is dependent upon the adoption of our implant systems by hospitals, ASCs, surgeons and patients.
Our future growth and profitability depend on our ability to increase physician awareness of our implant systems or ecosystem and on the willingness of surgeons and hospitals and ASCs to adopt our implant systems. Surgeons may not adopt our technologies unless they are able to determine, based on experience, clinical data, medical society and peer-surgeon recommendations and other analyses, that our implant systems provide a safe and effective treatment for shoulder care. Even if we can raise awareness among surgeons, such surgeons, hospitals and ASCs may be slow in changing their medical treatment practices and may be hesitant to select our systems for a variety of reasons, including:
•lack of experience with us and concerns that we are relatively new to market;
•long-standing relationships with companies and distributors that sell other products;
•lack of availability of adequate third-party payor coverage or reimbursement;
•competitive response and negative selling efforts from providers of alternative treatments;
•perception regarding the time commitment and skill development that may be required to gain familiarity and proficiency with our systems, technologies and instrumentation;
49
•perceived liability risk generally associated with the use of new products and treatment options in both hospitals as well as ASCs that lack ambulatory services and may expose patients to greater risk and us and care providers to increased potential liability;
•lack or perceived lack of sufficient clinical evidence, including long-term data, supporting clinical benefits or the cost-effectiveness of our systems over existing treatments; and
•the failure of key opinion leaders to provide recommendations regarding our systems, or to assure surgeons, patients and healthcare payors of the benefits of our systems as an attractive alternative to other treatment options.
To effectively market and sell our systems and other future products, we will need to continue to educate the medical community about the safety, efficacy, necessity and efficiency of our systems and about the patient population that would potentially benefit from using our systems. We will also need to educate the medical community, including at hospitals and ASCs, as to the broader benefits and efficiencies of our systems and our ecosystem. We cannot assure you that we will achieve broad education or market acceptance. In addition, some surgeons may choose to utilize our systems on only a subset of their total patient population or may not adopt our offerings at all. If we are not able to effectively demonstrate that our offerings are beneficial for a broad range of patients, adoption will be limited and may not occur as rapidly as we anticipate or at all, which would have a material adverse effect on our business, financial condition and results of operations. We cannot assure you that our implant systems will achieve broad market acceptance among hospitals, ASCs, surgeons or patients. Any such failure to satisfy demand or to achieve meaningful market acceptance and penetration will harm our future prospects and have a material adverse effect on our business, financial condition and results of operations.
The seasonality of our business creates variance in our quarterly revenue, which makes it difficult to compare or forecast our financial results.
Our revenue fluctuates on a seasonal basis, which affects the comparability of our results between periods. In particular, we have experienced and expect to continue to experience seasonality in our business. For example, the orthopedic industry traditionally experiences lower sales volumes in the months in and surrounding the third calendar quarter than throughout the rest of the year as elective procedures generally decline during the summer months due to warmer weather and its corresponding impact on individual lifestyles. Medical device companies historically experience a decline in the number of orthopedic implant surgeries in the summer months, and we may experience similar seasonality in the future. We expect these seasonal factors to become more pronounced in the future as our business grows. These seasonal variations are difficult to predict accurately, may vary amongst different markets and at times may be entirely unpredictable, which introduces additional risk into our business as we rely upon forecasts of customer demand to build inventory in advance of anticipated sales. In addition, we believe our limited history commercializing our products combined with our rapid growth has, in part, made our seasonal patterns more difficult to discern, making it more difficult to predict future seasonal patterns.
Our long-term growth depends on our ability to enhance our implant systems, expand our indications and develop and commercialize additional products in a timely manner. If we cannot innovate, we may not be able to develop or exploit new products in time to remain competitive.
The market for our systems is highly competitive, dynamic, and marked by rapid and substantial technological development and product innovation. New entrants or existing competitors could attempt to develop products that compete directly with ours. Our success depends on our ability to continually enhance and broaden our product offerings in response to changing surgeon and patient needs, competitive technologies and market pressures. For us to remain competitive, it is important to develop and bring to market new products. Any promotional activities for new products may not generate patient or physician awareness or increase revenue, and even if they do, any increase in revenue may not offset the costs and expenses we incur in such product development. Demand for our systems and other future products could be diminished by equivalent or superior products and technologies offered by competitors. If we are unable to innovate successfully, our products could become obsolete and our revenue would decline as our customers purchase our competitors’ products. Developing products is expensive and time-consuming and could divert management’s attention away from our core business. The success of any new product offering or product enhancements to our existing offerings will depend on several factors, including our ability to:
•assemble sufficient resources to acquire or discover additional products;
•properly identify and anticipate surgeon and patient needs;
•develop and introduce new products and product enhancements in a timely manner;
50
•avoid infringing upon the intellectual property rights of third parties;
•demonstrate, if required, the safety and efficacy of new products;
•obtain the necessary regulatory clearances, approvals or certifications for new products or product modifications;
•be fully compliant with the FDA regulations and, to the extent we expand internationally, be fully compliant with applicable foreign regulations related to marketing of new or modified devices;
•produce new products in commercial quantities at an acceptable cost;
•provide adequate training to potential users of our products;
•receive adequate coverage and reimbursement for procedures performed with our products; and
•develop an effective and dedicated commercial leadership and marketing team.
If we are unable to develop or improve products, applications or features due to constraints, such as insufficient cash resources, high employee turnover, inability to hire and retain personnel with sufficient technical skills or a lack of other research and development resources, we may not be able to maintain our competitive position compared to other companies. Furthermore, many of our competitors have the capability to devote a considerably greater amount of funds to their research and development programs than we do, and those that do not may be acquired by larger companies that could allocate greater resources to research and development programs. Our failure or inability to devote adequate research and development resources or compete effectively with the research and development programs of our competitors could harm our business.
Further, our new products may not gain a substantial degree of market acceptance among surgeons, patients or healthcare providers. Our failure to gain widespread acceptance of our new products could negatively affect our business, financial condition and results of operations.
We may expend our limited resources to pursue particular improvements or enhancements or new products and fail to capitalize on development opportunities that may be more profitable or for which there is a greater likelihood of success.
Because we have limited financial and managerial resources, we may forego or delay pursuit of opportunities with developments or initiatives that later prove to have greater commercial potential. Our resource allocation decisions may cause us to fail to timely capitalize on viable commercial products or profitable market opportunities. Furthermore, our spending on current and future research and development programs and products for specific indications may not yield any commercially viable products. If we do not accurately evaluate the commercial potential or target market for a particular product, we may relinquish valuable rights to that product through collaboration, licensing or other royalty arrangements in cases in which it would have been more advantageous for us to retain sole development and commercialization rights to such product.
We face the risk of product liability claims that could be expensive, divert management’s attention and harm our reputation and business. We may not be able to maintain adequate product liability insurance.
Our business exposes us to the risk of product liability claims that are inherent in the testing, manufacturing and marketing of medical devices. This risk exists even if a device is cleared or approved for commercial sale by the FDA or approved and manufactured in facilities licensed and regulated by the FDA. Our products are designed to affect, and any future enhancements to our products will be designed to affect, important bodily functions and processes. Any side effects, manufacturing defects, misuse or abuse associated with our systems could result in patient injury or death. The medical device industry has historically been subject to extensive litigation over product liability claims, and we may face product liability suits in the future. We may be subject to product liability claims if our systems cause, or merely appear to have caused, patient injury or death. In addition, an injury that is caused by the activities of our suppliers, such as those who provide us with components and raw materials, may be the basis for a claim against us. Product liability claims may be brought against us by patients, healthcare providers or others selling or otherwise coming into contact with our systems and other future products, among others. If we cannot successfully defend ourselves against product liability claims, we will incur substantial liabilities and reputational harm. If any of our products become the subject of a product liability claim, legal defenses are costly, regardless of the outcome. Thus, we may experience increased legal expenses as we defend any
51
such matter, and we could incur liabilities associated with adverse outcomes that exceed our insurance coverage. In addition, regardless of merit or eventual outcome, product liability claims may result in:
•distraction of management’s attention from our primary business;
•the inability to commercialize our products and develop enhancements to our products;
•decreased demand for our products;
•damage to our business reputation and brand;
•product recalls or withdrawals from the market;
•withdrawal of clinical trial participants;
•substantial monetary awards to patients or other claimants; or
•loss of sales.
Additionally, we could experience a material design or manufacturing failure in our products, a quality system failure, other safety issues or heightened regulatory scrutiny that would warrant a recall of some of our products. While we may attempt to manage our product liability exposure by proactively recalling or withdrawing from the market any defective products, any recall or market withdrawal of our products may delay the supply of those products to our customers and may impact our reputation. We may not be successful in initiating appropriate market recall or market withdrawal efforts that may be required in the future and these efforts may not have the intended effect of preventing product malfunctions and the accompanying product liability that may result. Such recalls and withdrawals may also be used by our competitors to harm our reputation for safety or be perceived by patients as a safety risk when considering the use of our products. Product liability lawsuits and claims, safety alerts and product recalls and withdrawals, regardless of their ultimate outcome, could have a material adverse effect on our business, financial condition and results of operations.
Although we have product liability insurance that we believe is appropriate, this insurance is subject to deductibles and coverage limitations. Our current product liability insurance may not continue to be available to us on acceptable terms, if at all, and, if available, coverage may not be adequate to protect us against any future product liability claims. If we are unable to obtain insurance at an acceptable cost or on acceptable terms or otherwise protect against potential product liability claims, we could be exposed to significant liabilities. While we have not been materially impacted by product liability claims, lawsuits and recalls to date, we may be materially impacted by such events in the future, and any such claim, lawsuit, recall or other claim with respect to uninsured liabilities or for amounts in excess of insured liabilities could have a material adverse effect on our business, financial condition and results of operations. Further, such product liability matters may negatively impact our ability to obtain insurance coverage or cost-effective insurance coverage in future periods.
Industry trends have resulted in increased downward pricing pressure on medical services and products, which may affect our ability to sell our products at prices necessary to support our current business strategy.
The trend toward health care cost containment through aggregating purchasing decisions and industry consolidation, along with the growth of managed care organizations, is placing increased emphasis on the delivery of more cost-effective medical therapies. For example:
•There has been consolidation among health care facilities and purchasers of medical devices, particularly in the United States. One of the results of such consolidation is that group purchasing organizations (“GPOs”), integrated delivery networks and large single accounts use their market power to consolidate purchasing decisions, which intensifies competition to provide products and services to health care providers and other industry participants, resulting in greater pricing pressures and the exclusion of certain suppliers from important market segments.
•Surgeons increasingly have moved from independent, outpatient practice settings toward employment by or affiliation with hospitals and other larger health care organizations, which aligns surgeons’ product choices with the institutional providers’ price sensitivities and adds to pricing pressures. Hospitals and health care facilities have introduced and may continue to introduce new pricing structures into their contracts to contain health care costs, including fixed price formulas and capitated and construct pricing. Such pricing structures may create
52
incentive programs for surgeons that are less advantageous to our business model, including capitation models where surgeons are paid a fixed amount per patient, per period.
•Certain hospitals provide financial incentives to doctors for reducing hospital costs (known as gainsharing), rewarding physician efficiency (known as physician profiling) and encouraging partnerships with health care service and goods providers to reduce costs.
•Existing and proposed laws, regulations and industry policies, in both domestic and international markets, regulate or seek to increase regulation of sales and marketing practices and the pricing and profitability of companies in the health care industry.
More broadly, provisions of the Affordable Care Act (“ACA”) could meaningfully change the way health care is developed and delivered in the United States, and may adversely affect our business and results of operations. For further discussion of these challenges, see the section titled “ —Risks Related to Regulatory Matters—Legislative or regulatory reforms may have a material adverse effect on us.” We cannot predict accurately what health care programs and regulations will ultimately be implemented at the federal or state level, or the effect of any future legislation or regulation in the United States or elsewhere. However, any changes that have the effect of reducing reimbursement by government health care programs and other third-party payors for procedures using our systems or reducing medical procedure volumes could have a material and adverse effect on our business, financial condition and results of operations. Any decline in the amount that payors reimburse our customers for our systems could make it difficult for customers to continue using, or to adopt, our systems and could create additional pricing pressure for us. If we are forced to lower the price we charge for our systems, or if we add more components to our systems, our gross margins will decrease, which will adversely affect our ability to invest in and grow our business. If we are unable to maintain our prices, or if our costs increase and we are unable to offset such increase with an increase in our prices, our margins could erode. While we have not experienced a material impact from any such industry trends to date, there is no assurance that our net revenue will not be subject to these headwinds in the future.
In addition, the largest medical device companies with multiple product franchises have increased their effort to leverage and contract broadly with customers across franchises by providing volume discounts and multi-year arrangements that could prevent our access to these customers or make it difficult, or impossible, to compete on price.
Our employees and independent contractors, including independent distributors and any other consultants, any future service providers and other vendors, may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could have an adverse effect on our results of operations.
We are exposed to the risk that our employees and independent contractors, including independent distributors and any other consultants, any future commercial collaborators, and other vendors may engage in misconduct or other illegal activity. Misconduct by these parties could include intentional, reckless and/or negligent conduct or other unauthorized activities that violate: federal, state or local laws and regulations, as well as the laws, regulations and rules of regulatory bodies such as the FDA; manufacturing standards; U.S. federal and state health care fraud and abuse laws and regulations, data privacy laws and other similar non-U.S. laws; or laws that require the true, complete and accurate reporting of financial information or data. For example, we are aware of limited instances in which our dedicated commercial leadership team and independent distributors have recommended using our systems for off-label uses, including using certain aTSA products for rTSA procedures and vice versa. It is not always possible to identify and deter misconduct by employees and other third parties, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with such laws or regulations. In addition, we are subject to the risk that a person or government could allege such fraud or other misconduct, even if none occurred. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business and financial results, including, without limitation, the imposition of significant civil, criminal and administrative penalties, damages, monetary fines, disgorgements, possible exclusion from participation in Medicare, Medicaid and other U.S. health care programs, other sanctions, imprisonment, contractual damages, reputational harm, diminished profits and future earnings and curtailment of our operations, any of which could adversely affect our ability to operate our business and our results of operations.
If rising prices or unavailability of raw materials continue to persist, our business and results of operations may be adversely affected.
Volatility in raw material prices and availability can impact our business and finances due to numerous factors beyond our control, including general, domestic, and international economic conditions, labor costs, production levels, competition,
53
consumer demand, import duties, inflation and currency exchange rates. This volatility can significantly affect the availability and cost of raw materials that our suppliers purchase and are ultimately used in our systems, and may therefore have a material adverse effect on our business, results of operations, and financial condition.
In addition, the current U.S. administration has expressed strong concerns about imports from countries that it perceives as engaging in unfair trade practices, and has imposed tariffs or other restrictions on products, components or raw materials sourced from those countries. Moreover, these new tariffs, or other changes in U.S. trade policy, have triggered and may in the future trigger retaliatory actions by affected countries. For example, there have been and continue to be further indications that there may be an increase in tariff rates on various types of goods imported from Canada and Europe that could apply to the raw materials we require in our products, including certain types of metal powders used for coating our products. In the event that any such possible tariff increases remain in place or become enacted in the future, they could significantly increase the cost of materials and components that our suppliers import and use in our systems, which in turn could increase our supply costs. At this time, there can be no assurance that we will be able to pass any portion of such increases on to customers. We currently do not hedge against our exposure to changing raw material prices and are not aware as to whether our suppliers hedge. As a result, fluctuations in raw material prices could have a material adverse effect on our business, results of operations, and financial condition. Supply shortages or changes in availability for any particular type of raw material can delay supplier volume and production capabilities or cause increases in the cost of manufacturing our products. We may be negatively affected by changes in availability and pricing of raw materials, which could negatively impact our results of operations.
Risks Related to Administrative, Organizational and Commercial Operations and Growth
If hospitals, ASCs, and other health care facilities do not approve the use of our implant systems, our sales may not increase.
In order for surgeons to use our systems at hospitals, ASCs and other health care facilities, we are often required to obtain approval from those hospitals, ASCs and health care facilities. Typically, hospitals, ASCs and health care facilities review the comparative effectiveness and cost of products used in the facility. The makeup and evaluation processes for health care facilities vary considerably, and it can be a lengthy, costly and time-consuming effort to obtain approval by the relevant health care facilities. Additionally, hospitals, ASCs and other health care facilities, which manage purchasing for multiple facilities, may also require us to enter into a purchase agreement and satisfy numerous elements of their administrative procurement process, which can also be a lengthy, costly and time-consuming effort. If we do not obtain access to hospitals, ASCs, GPOs and other health care facilities in a timely manner, or at all, via their approvals or purchase contract processes, or otherwise, or if we are unable to obtain approvals or secure contracts in a timely manner or on terms that are economically viable to our business, or at all, our operating costs will increase, our sales may decrease and our operating results may be adversely affected. Furthermore, we may expend significant efforts on these costly and time-consuming processes but may not be able to obtain necessary approvals or secure a purchase contract from such hospitals, ASCs or health care facilities.
Performance issues, service interruptions or price increases by shipping carriers could adversely affect our business and harm our reputation and ability to provide our systems on a timely basis.
Expedited, reliable shipping is essential to our operations. We rely heavily on providers of transport services for reliable and secure point-to-point transport of our systems to our customers and for tracking of these shipments, including our InSet tray systems that are delivered directly to hospitals and ASCs, employees or independent distributors. Should a carrier encounter delivery performance issues such as loss, damage or destruction of our systems, it would be costly to replace our systems in a timely manner, could cause surgeries using our systems to be delayed or canceled and such occurrences may damage our reputation and lead to decreased demand for our systems and increased cost and expense to our business. Once a product leaves our facilities, we are no longer in control of the care of such shipped products and any damage that may occur during transit may not be readily detectable by the customer, including any penetrations of sterile barriers, temperature swings that may cause package seal rupturing or exposure to fine particulates or other debris that is not easily detectable. In addition, any significant increase in shipping rates could adversely affect our operating margins and results of operations. Similarly, strikes, severe weather, natural disasters, including fires and hurricanes, or other service interruptions affecting delivery services we use would adversely affect our ability to process orders for our systems on a timely basis.
54
If coverage or adequate levels of reimbursement from third-party payors for procedures using our implant systems, or any future products we may seek to commercialize, are not obtained or maintained, shoulder specialists and patients may be reluctant to use our systems and our business will suffer.
In the United States, health care providers such as hospitals and ASCs who purchase our implant systems generally rely on third-party payors, principally federally-funded Medicare, state-funded Medicaid and private health insurance plans, to pay for all or a portion of the cost of shoulder procedures and products utilized in those procedures. We may be unable to sell our implant systems, or any future products we may seek to commercialize, on a profitable basis if third-party payors deny coverage or reduce their current levels of reimbursement for procedures using our systems. Our sales depend largely on governmental health care programs and private health insurers reimbursing providers for procedures using our implant systems. Significant changes to operations at, funding of, or restructuring of such governmental authorities, including but not limited to a government shutdown, decreases in staff who are able to provide reimbursement services, reductions or other changes in funding provided to such governmental authorities, and changes in policy and enforcement priorities, may adversely affect our business. Hospitals, ASCs and other health care providers may not purchase our products if they do not receive adequate reimbursement from third-party payors for procedures using our products. Payors continue to review their coverage policies for existing and new therapies and may deny coverage for treatments that include the use of our systems or any future products we may seek to commercialize. Third-party payors, whether governmental or commercial, are developing increasingly sophisticated methods of controlling healthcare costs. In addition, no uniform policy of coverage and reimbursement for procedures using our solution exists among third-party payors. Therefore, coverage and reimbursement for procedures using our products can differ from payor to payor.
In addition, some health care providers in the United States have adopted or are considering bundled payment methodologies and/or managed care systems in which providers contract to provide comprehensive health care for a fixed cost per person. Health care providers may attempt to control costs by authorizing fewer elective surgical procedures, including shoulder surgeries, or by requiring the use of the least expensive procedure available. In addition, third-party payors increasingly are requiring evidence that medical devices are cost-effective, and if we are unable to meet this requirement, the third-party payor may not cover procedures using our products, which could reduce sales of our products to health care providers who depend upon third-party payor reimbursement for payment. Changes in coverage policies or health care cost containment initiatives that limit or restrict reimbursement for procedures using our systems may have an adverse effect on our business. For example, the CMS only recently approved total shoulder arthroplasty for reimbursement when performed in an ASC on January 1, 2024, and any change or reversal in this decision would adversely affect our results of operations and financial condition.
We depend on third-party contract manufacturers and suppliers, some of which are single source, to produce and package all elements comprising our shoulder implant systems, and if these suppliers and manufacturers fail to supply us, our products or their components or subcomponents in sufficient quantities or at all, or in accordance with applicable regulatory requirements and our specifications, it will have a material adverse effect on our business, financial condition, and results of operations.
We utilize qualified medical device contract manufacturers and suppliers, the majority of which are single sources, to produce, package and sterilize the majority of the elements comprising our systems. For us to be successful, our suppliers must be able to provide us with products and components in substantial quantities, in compliance with regulatory requirements, in accordance with agreed upon specifications, at acceptable costs and quality, and on a timely basis. An interruption in our commercial operations could occur if we encounter delays or difficulties in securing these components, and if we cannot then obtain an acceptable substitute. Our significant single source suppliers include Avalign Technologies, Inc., which manufactures our humeral stems, Micropulse, Inc., which manufactures and supplies our InSet Glenoid products, Trifecta Medical Group, which manufactures our instrument cases and trays and Revelation Medical Devices, which manufactures the surgical instruments used during procedures involving our systems. While we estimate replacing these single source suppliers could take up to between six and twelve months, in some of these examples alternative second source suppliers may not be readily available. Furthermore, while we have two sterilization partners, we utilize a single supplier to sterilize all of our polyethylene implants. We also do not have long-term supply agreements, and we typically purchase our products on a purchase order basis, which means that aside from any binding purchase orders we have from time to time, our supplier could cease supplying to us or change the terms on which it is willing to continue supplying to us at any time. Although we have identified alternate third parties who could provide this manufacturing service and expect to reduce our reliance on single source manufacturers and third parties we use for sterilization in the foreseeable future, we cannot guarantee that we would be able to contract with such alternate third parties within a reasonable amount of time or at all, or upon similar pricing and volume terms, nor can we be assured that any such third party would be capable of producing products in sufficient volume and quality. For example, a replacement supplier may discover challenges with our products or we may not perfectly specify our product designs for such new suppliers’ manufacturing systems, which
55
may require us to refine our products and/or incur additional research and development expense, which could delay our sales and have an adverse effect on our financial condition and results of operations. Furthermore, if we are required to change the manufacturer of a critical component of any of our systems, we will be required to verify that the new manufacturer maintains facilities, procedures and operations that comply with our quality standards and applicable regulatory requirements, which could further impede the manufacture of our systems in a timely manner or at all. A change in manufacturer could trigger the requirement to submit and obtain a new 510(k) clearance from the FDA, or similar international regulatory authorization before we implement the change, which could cause substantial delays. Any event, including those listed above, other circumstances that result in a prolonged business disruption or shutdown to one or more of single source suppliers’ facilities, or the facilities of our other vendors and service providers, or a deterioration in our relationship with any of our single source suppliers, or any of our other vendors or service providers, in each case, could create conditions that prevent, or significantly and adversely affect, our sales, increase our expenses, create potential liabilities or damage our reputation, any of which could have an adverse effect on our business, financial condition and results of operations. Our marketing applications and other regulatory submissions may identify the facilities used by our third-party manufacturers for the manufacture of our systems, and these facilities are subject to inspections. We do not control the manufacturing process of, and are completely dependent on, third-party manufacturers for compliance with current good manufacturing practice (“cGMP”) requirements for manufacture of our systems. If these third-party manufacturers cannot successfully manufacture material that conforms to our specifications and the strict regulatory requirements of the FDA or any comparable foreign regulatory authority, we will not be able to secure and/or maintain marketing authorization or certification of our products manufactured in their manufacturing facilities.
We seek to strategically maintain sufficient levels of inventory to help mitigate supply disruption, usually holding sufficient inventory to allow for manufacturing from approximately 90 days following the loss of a supplier, to accommodate varying demand mix and to achieve more efficient volume-based pricing on our components; however, we may not be accurate in our estimates which could result in insufficient inventory to meet demand or excess inventory and the risk of inventory obsolescence and expiration. Further, while we have entered into several supply agreements in an effort to reinforce our supply chain, there is no guarantee the counter-party suppliers will adhere to the terms of these agreements.
We do not have complete control over all aspects of the manufacturing process of, and are dependent on, our manufacturing partners for compliance with current cGMP regulations applicable to our systems. Third-party manufacturers may not be able, or may fail, to comply with cGMP regulations. If our third-party manufacturers cannot successfully manufacture products that conform to our specifications and the strict regulatory requirements of the FDA, they will not be able to secure and/or maintain registration for their manufacturing facilities. In addition, we do not have complete control over the ability of our third-party manufacturers to maintain adequate quality control, quality assurance and qualified personnel. The failure of our manufacturing partners to maintain acceptable quality requirements could result in quality issues, including recalls. If one of our manufacturing partners fails to maintain acceptable quality requirements or perform as agreed, we may have to identify and qualify a new supplier or develop in-house manufacturing capabilities, which could require significant capital investments.
Our suppliers may encounter problems during manufacturing for a variety of reasons, including, for example, failure to follow specific protocols and procedures, failure to comply with applicable legal and regulatory requirements, equipment malfunction and environmental factors, including damages to their facilities as a result of fires, storms or hurricanes, failure to properly conduct their own business affairs, and infringement of third-party intellectual property rights, any of which could delay or impede their ability to meet our requirements. Our suppliers may also not prioritize the production of our systems compared to the suppliers’ larger customers so we may experience longer delays in receiving our requested orders. Our reliance on a third-party manufacturer and third-party suppliers also subjects us to other risks that could harm our business that we would not be subject to if we manufactured ourselves, including, among others:
•we may not be a major customer of many of our suppliers, and these suppliers may therefore give other customers’ needs higher priority than ours, including some of our competitors who use the same manufacturing partners;
•third parties may threaten or enforce their intellectual property rights against our suppliers, which may cause disruptions or delays in shipment, or may force our suppliers to cease conducting business with us;
•we may not be able to obtain an adequate supply of components in a timely manner or on commercially reasonable terms;
56
•our suppliers, especially new suppliers, may make errors in manufacturing that could negatively affect the efficacy or safety of our systems or cause delays in shipment;
•we may have difficulty locating and qualifying alternative suppliers and our existing suppliers could be acquired by companies that have limiting or exclusive relationships with our competitors;
•switching components or suppliers may require product redesign and possibly submission to the FDA, or notified bodies, which could significantly impede or delay our commercial activities;
•one or more of our single-source suppliers may be unwilling or unable to supply components of our systems;
•other customers, including our competitors, may use fair or unfair negotiation tactics or pressures to impede our use of the suppliers;
•the occurrence of a fire, natural disaster or other catastrophe, or the occurrence of geopolitical conflicts, as well as any sanctions or other actions resulting therefrom impacting one or more of our suppliers may affect their ability to deliver products to us in a timely manner;
•the occurrence of pandemics, epidemics and other public health emergencies may impact a manufacturing facility by limiting operating capacity or sideline critical employees involved in the manufacturing processes thereby affecting their ability to deliver products to us in a timely manner;
•our suppliers may encounter financial or other business hardships unrelated to our demand, which could inhibit their ability to fulfill our orders and meet our requirements;
•our suppliers may not maintain the confidentiality of our proprietary information or may mislabel raw materials resulting in inaccurate certifications; and
•higher manufacturing and product costs than more vertically integrated companies.
Any of these factors could cause delay or suspension of commercialization and marketing, regulatory submissions or required approvals, clearances or certifications, or cause us to incur higher costs. Furthermore, if our contract manufacturers fail to deliver the required commercial quantities of finished products on a timely basis and at commercially reasonable prices and we are unable to find one or more replacement manufacturers capable of production at a substantially equivalent cost, in substantially equivalent volumes and quality, and on a timely basis, we would likely be unable to meet demand for our systems and we would lose potential revenue. Any difficulties in locating and hiring third-party manufacturers, or in the ability of third-party manufacturers to supply quantities of our products at the times and in the quantities we need, could have a material adverse effect on our business. It may take a significant amount of time and resources (including costs) to establish an alternative source of supply for our products and to have any such new source authorized by the FDA or other bodies. Given our reliance on certain single-source suppliers, we are especially susceptible to supply shortages because we do not have alternate suppliers currently available that we could contract with in a reasonable amount of time or at all, or upon similar pricing and volume terms, nor can we be assured that any such third party would be capable of producing in sufficient volume and of sufficient quality.
Moreover, some third parties are located in markets subject to infrastructure problems and exposed to significant natural disasters, such as hurricanes and wildfires. Failure of third parties to meet their contractual, regulatory, and other obligations may have a material adverse effect on our business, financial condition, and results of operations. Any of these matters could materially and adversely affect our business, financial condition, and results of operations.
Our results of operations will be materially harmed if we are unable to accurately forecast demand for our implant systems and manage our inventory.
In order to market and sell effectively, we must maintain significant levels of inventory of systems and surgical instrumentation. As a result, a significant amount of our cash used in operations has been associated with maintaining these levels of inventory. To ensure adequate inventory supply, we must forecast inventory needs and manufacturing orders based on our estimates of future demand. Our ability to accurately forecast demand for our systems could be negatively affected by many factors, including our failure to accurately manage our expansion strategy, product introductions by competitors, an increase or decrease in customer demand for our systems or for products of our competitors, our failure to accurately forecast customer acceptance of new products, unanticipated changes in general market conditions or regulatory matters and weakening of economic conditions or consumer confidence in future economic conditions. Inventory levels in
57
excess of customer demand may result in inventory write-downs or write-offs, which would cause our gross margin to be adversely affected and could impair the strength of our brand. Furthermore, as we continue to grow and expand, we require increasing levels of inventory which may subject us to greater financial risk in the event of a recall where such inventory is no longer commercially viable. Conversely, if we underestimate customer demand for our systems, our manufacturers and suppliers may not be able to deliver systems to meet our requirements, which could result in damage to our reputation and customer relationships. In addition, if we experience a significant increase in demand, additional supplies of raw materials or additional manufacturing capacity may not be available at our existing manufacturers and suppliers when required on terms that are acceptable to us, or at all, which will negatively affect our business, financial condition and results of operations.
We seek to maintain sufficient levels of inventory in order to protect ourselves from supply interruptions. As a result, we are subject to the risk that a portion of our inventory will become obsolete or expire, which could have a material adverse effect on our earnings and cash flows due to the resulting costs associated with the inventory obsolescence charges and costs required to replace such inventory.
We may enter into collaborations, in-licensing arrangements, joint ventures, strategic alliances or partnerships with third parties that may not result in the development of commercially viable products, product improvements or the generation of significant future revenues.
In the ordinary course of our business, we may enter into collaborations, in-licensing arrangements, joint ventures, strategic alliances, partnerships or other arrangements to develop new products or product improvements and to pursue new markets. Proposing, negotiating and implementing collaborations, in-licensing arrangements, joint ventures, strategic alliances or partnerships may be a lengthy and complex process. Other companies, including those with substantially greater financial, marketing, sales, technology or other business resources, may compete with us for these opportunities or arrangements. We have entered into a second amended and restated software license agreement with Genesis Software, pursuant to which we have exclusive rights to use certain preoperative surgery planning platform software.
We may not identify, secure, or complete any such transactions or arrangements in a timely manner, on a cost-effective basis, on acceptable terms or at all. We have limited institutional knowledge and experience with respect to these business development activities, and we may also not realize the anticipated benefits of any such transaction or arrangement. In particular, these collaborations may not result in the development of products that achieve commercial success or viable product improvements or result in significant revenues and could be terminated prior to developing any products. For example, while our license agreement with Genesis Software provides us with a preoperative software planning platform that is used by surgeons preparing for procedures, we do not generate any separate revenue specifically from ProVoyance.
Additionally, we may not be in a position to exercise sole decision-making authority regarding the transaction or arrangement, which could create the potential risk of creating impasses on decisions, and our future collaborators may have economic or business interests or goals that are, or that may become, inconsistent with our business interests or goals. It is possible that conflicts may arise with our collaborators, such as conflicts concerning the achievement of performance milestones, or the interpretation of significant terms under any agreement, such as those related to financial obligations or the ownership or control of intellectual property developed during the collaboration. If any conflicts arise with any future collaborators, they may act in their self-interest, which may be adverse to our best interest, and they may breach their obligations to us. For example, we have and will continue to enter into agreements with third parties that are owned or controlled by certain of our officers and directors. Robert Ball, our Chief Executive Officer and Executive Chairman, and is a co-founder and director of Genesis Innovation Group, Inc. (“Genesis Innovation”) and Genesis Software. See the section titled “—Risks Related to Administrative, Organizational and Commercial Operations and Growth—Certain of our officers and directors also serve as directors of, and are affiliates of, Genesis Innovation, Genesis Software and cultivate(MD), which may give rise to a perceived or actual conflict of interest which may adversely impact your interests.” In addition, we may have limited control over the amount and timing of resources that any future collaborators devote to our or their future products.
Disputes between us and our collaborators may result in litigation or arbitration which would increase our expenses and divert the attention of our management. Further, these transactions and arrangements will be contractual in nature and will generally be terminable under the terms of the applicable agreements and, in such event, we may not continue to have rights to the products relating to such transaction or arrangement or may need to purchase such rights at a premium. If we enter into in-bound intellectual property license agreements, we may not be able to fully protect the licensed intellectual property rights or maintain those licenses. Future licensors could retain the right to prosecute and defend the intellectual property rights licensed to us, in which case we would depend on the ability of our licensors to obtain, maintain and enforce intellectual property protection for the licensed intellectual property. These licensors may determine not to pursue
58
litigation against other companies or may pursue such litigation less aggressively than we would. Further, entering into such license agreements could impose various diligence, commercialization, royalty or other obligations on us. Future licensors may allege that we have breached our license agreement with them, and accordingly seek to terminate our license, which could adversely affect our competitive business position and harm our business prospects.
AI presents risks and challenges that can impact our business including by posing security risks to our confidential information, proprietary information and personal data.
We incorporate AI solutions into our platform, and these applications are increasingly important to our operations over time. AI presents risks such as inaccuracy, bias, toxicity, intellectual property infringement or misappropriation, data privacy and cybersecurity and data provenance. In addition, AI utilizes machine learning and predictive analytics, which in some cases present flawed, biased, and inaccurate results, and may have errors or inadequacies that are not easily detectable and may also be subject to data herding and interconnectedness (i.e., multiple market participants utilizing the same data), in each case adversely impacting our business. The regulatory framework for AI is rapidly evolving as many federal, state, and foreign government bodies and agencies have introduced or are currently considering additional laws and regulations. Additionally, existing laws and regulations could affect the operation of our AI. As a result, implementation standards and enforcement practices are likely to remain uncertain for the foreseeable future, and we cannot yet determine the impact of future laws, regulations, standards, or market perception of their requirements on our business and our response. It is possible that new laws and regulations will be adopted in the United States and in other jurisdictions outside the United States, or that existing laws and regulations, including competition and antitrust laws, may be interpreted in ways that would limit our ability to use AI for our business, or require us to change the way we use AI in a manner that negatively affects the performance of our technologies, services, and business and the way in which we use AI. We may need to expend resources to adjust our technologies or services in certain jurisdictions if the laws, regulations, or decisions are not consistent across jurisdictions. Further, the cost to comply with such laws, regulations, or decisions and/or guidance interpreting existing laws, could be significant and would increase our operating expenses (such as by imposing additional reporting obligations regarding our use of AI). Such an increase in operating expenses, as well as any actual or perceived failure to comply with such laws and regulations, could adversely affect our business, financial condition, and results of operations.
These issues, combined with an uncertain regulatory environment, may further result in reputational harm, liability, or other adverse consequences to our business operations. Incorporation of generative AI tools by vendors into their offerings without disclosing this use to us would expose us to potential liability, and the providers of these generative AI tools may not meet existing or rapidly evolving regulatory or industry standards with respect to privacy and data protection and may inhibit our or our vendors’ ability to maintain an adequate level of service and experience. If our vendors, or our third-party partners experience an actual or perceived breach or privacy or security incident because of the use of generative AI, we may lose valuable intellectual property and confidential information and our reputation and the public perception of the effectiveness of our security measures could be harmed. Further, bad actors around the world use increasingly sophisticated methods, including the use of AI, to engage in illegal activities involving the theft and misuse of personal information, confidential information, and intellectual property. Any of these outcomes could damage our reputation, result in the loss of valuable property and information, and adversely impact our business.
We may not be able to establish or strengthen our brand.
We believe that establishing and strengthening our brand, including our ecosystem, is important to achieving widespread acceptance of our implant systems, particularly because of the highly competitive nature of the market for similar products. We believe the quality, breadth and reliability of our systems is critical to building physician support for our systems, and any negative publicity regarding the quality or reliability of our systems or shoulder arthroplasty procedures generally could significantly damage our reputation. In the course of conducting our business, we must adequately address quality issues that may arise with our systems and technologies. Although we have established internal procedures designed to minimize risks that may arise from quality issues, we may not be able to eliminate or mitigate occurrences of these issues and associated liabilities. In addition, even in the absence of quality issues, we may be subject to claims and liability if the performance of our systems do not meet the expectations of physicians or patients or any improper use of our systems or technologies that results in injury or unsatisfactory results.
In addition, promoting and positioning our brand will depend largely on the success of our medical education efforts and our ability to educate shoulder specialists and patients. These brand promotion activities may not yield increased sales and, even if they do, any sales increases may not offset the expenses we incur to promote our brand.
59
If we fail to successfully promote and maintain our brand, or if we incur substantial expenses in an unsuccessful attempt to promote and maintain our brand, our shoulder solutions may not be accepted by physicians or patients, which would adversely affect our business, results of operations and financial condition.
Our inability to maintain contractual relationships with health care professionals could have a negative impact on our research and development and medical education programs.
We maintain contractual relationships with respected physicians and medical personnel in hospitals, private practice and universities who assist in clinical studies, product research, data collection for our registries and development and in the training of shoulder specialists on the safe and effective use of our products. We continue to place emphasis on the validation of the benefits of our implant systems through clinical studies, the development of proprietary products and product improvements as well as providing high quality training and support. If we are unable to maintain these relationships, our ability to develop and market new and improved products and train on the use of those products could decrease, and future operating results could be unfavorably affected. At the same time, the medical device industry’s relationship with physicians is under increasing scrutiny by the U.S. Department of Health and Human Services Office of Inspector General (“OIG”), the U.S. Department of Justice (“DOJ”), the state attorneys general and other foreign and domestic government agencies. Our failure to comply with requirements governing the industry’s relationships with physicians or an investigation into our compliance by the OIG, the DOJ, state attorneys general and other government agencies, could negatively affect our business, financial condition and results of operations. See the section titled “—Risks Related to Regulatory Matters—Our relationships with customers, physicians and third-party payors are subject to federal and state health care fraud and abuse laws, false claims laws, physician payment transparency laws and other health care laws and regulations. If we or our employees, independent contractors, consultants, commercial partners, or vendors violate these laws we could face substantial penalties.”
We may be unable to continue to successfully demonstrate to shoulder specialists or key opinion leaders the merits of our implant systems and technologies compared to those of our competitors, which may make it difficult to achieve market acceptance.
Shoulder specialists play the primary role in determining the course of treatment and, ultimately, the type of products that will be used to treat a patient. As a result, our success depends, in large part, on our ability to effectively market and demonstrate to shoulder specialists the merits of our systems and methodologies compared to those of our competitors. Acceptance of our systems and methodologies depends on educating shoulder specialists as to the distinctive characteristics, clinical benefits, safety, cost-effectiveness and efficiencies of our systems and technologies as compared to those of our competitors, and on training surgeons in the proper use of our systems and offerings. If we are not successful in convincing surgeons of the merits of our systems and methodologies or educating them on the use of our systems, they may not use our systems or may not use them effectively and we may be unable to increase our sales, sustain our growth or achieve and sustain profitability.
Also, since certain of our offerings and products are relatively new, some surgeons may be reluctant to change their surgical treatment practices for the following reasons, among others:
•lack of experience with our systems and technologies;
•existing relationships with competitors and distributors that sell competitive products;
•lack or perceived lack of evidence supporting additional patient benefits;
•perceived liability risks generally associated with the use of new systems and technologies;
•less attractive availability of coverage and reimbursement by third-party payors compared to procedures using competitive products and other techniques;
•costs; and
•the time commitment that may be required for training.
These reasons may affect the pace of adoption of our systems and future products that we may offer.
In addition, we believe recommendations and support of our systems and technologies by surgeons, shoulder specialists and other key opinion leaders in our industry are essential for market acceptance. If we do not receive support from such surgeons, shoulder specialists and other key opinion leaders, if long-term data does not show the benefits of
60
using our systems or if the benefits offered by our systems are not sufficient to justify their cost, shoulder specialists, hospitals, ASCs and other health care facilities may not use our systems and we might be unable to achieve market acceptance.
Our systems are designed to satisfy long-term demands for patients and, given the length of time we have been commercially active, we do not have significant long-term use cases available. In the orthopedic industry, participants have been able to demonstrate favorable short-term outcomes that later went on to fail and not provide adequate long-term performance. Any such failure to demonstrate long-term satisfaction for our systems in the future could have adverse affect on our brand, demand for our systems and our results of operations and financial condition.
If surgeons fail to safely and appropriately use our implant systems, or if we are unable to train surgeons on the safe and appropriate use of our systems, we may be unable to achieve our expected sales, growth or profitability.
An important part of our ecosystem includes our ability to screen for and identify surgeons who have the requisite training and experience to safely and appropriately use our implant systems and to train a sufficient number of these surgeons and provide them with adequate instruction in the use of our systems. There is a training process involved for surgeons to become proficient in the use of our systems. This training process may take longer or be more expensive than expected and may therefore affect our ability to increase sales. Convincing surgeons to dedicate the time and energy necessary for adequate training is challenging, and we may not be successful in these efforts. Recent changes to federal guidance regarding medical education programs under the federal Anti-Kickback Statute also could limit our ability to train surgeons, and such programs could be subject to challenge under the federal Anti-Kickback Statute. See the section titled “—Risks Related to Regulatory Matters—Our relationships with customers, physicians and third-party payors are subject to federal and state health care fraud and abuse laws, false claims laws, physician payment transparency laws and other health care laws and regulations. If we or our employees, independent contractors, consultants, commercial partners, or vendors violate these laws we could face substantial penalties.”
Furthermore, if clinicians are not properly trained, they may misuse or ineffectively use our systems. Any such improper use may result in unsatisfactory outcomes, patient injury, negative publicity or lawsuits against us, any of which could harm our reputation and affect future sales. Accordingly, if surgeons fail to safely and appropriately use our systems, use them for off-label purposes, or if we are unable to train them on the safe and appropriate use of our systems, we may be unable to achieve our desired sales, growth or profitability. Furthermore, despite the requisite training, surgeons may continue to deliberately use our systems in ways not in conformance to our instructions or methods for which our systems are designed, which could lead to unsuccessful patient outcomes which may harm our brand, demand and results of operations.
The loss of any member of our executive management team or our inability to attract and retain highly skilled members of our dedicated commercial leadership and marketing teams as well as certain third-party engineers could have a material adverse effect on our business, financial condition and results of operations.
Our success depends on the skills, experience and performance of the members of our executive management team including Robert Ball, our Chief Executive Officer & Executive Chairman, and Matthew Ahearn, our Chief Operating Officer, as well as David Blue, our Chief Customer Experience Officer, Jeffrey Points, our Chief Financial Officer and Jon Osborne, our Senior Vice President of Commercial Development. The individual and collective efforts of our executive management team will be important as we continue to commercialize our existing systems, develop new products and technologies and expand our commercial activities. The loss or incapacity of existing members of our executive management team could have a material adverse effect on our business, financial condition and results of operations if we experience difficulties in hiring qualified successors. We do not maintain “key person” insurance for any of our executives or key employees.
Our commercial, quality and research and development programs and operations depend on our ability to attract and retain highly skilled team members. We may be unable to attract or retain qualified team members. All of our employees are at-will, which means that either we or the employee may terminate his or her employment at any time. The loss of key employees, failure of any key employee to perform, our inability to attract and retain skilled employees, as needed, or our inability to effectively plan for and implement a succession plan for key employees could have a material adverse effect on our business, financial condition and results of operations.
Furthermore, we rely on third parties, including Genesis Innovation and Genesis Software, for engineering and design services. The loss of any such contractual arrangement with Genesis Innovation or Genesis Software, including access to the professional services of such engineers, could have a material adverse effect on our business, financial condition and results of operations.
61
Certain of our officers and directors also serve as directors of, and are affiliates of, Genesis Innovation, Genesis Software and cultivate(MD), which may give rise to a perceived or actual conflict of interest which may adversely impact your interests.
We have and will continue to operate under agreements with third parties that are owned or otherwise affiliated with certain of our officers and directors. For example, Robert Ball, our Chief Executive Officer and Executive Chairman, is a co-founder and director of Genesis Innovation, Genesis Software and Genesis Investment Holdings, LLC (“Genesis Investment Holdings”). Genesis Investment Holdings is affiliated with cultivate(MD) Holdings, LLC (“cultivate(MD)”). In addition, Mr. Ball, Matthew Ahearn, our Chief Operating Officer and a member of our board of directors, and David Blue, our Chief Customer Experience Officer, are directors of cultivate(MD), which is an investor in Genesis Software. Mr. Ahearn is also a director of Genesis Investment Holdings. Pursuant to our consulting agreement with Genesis Innovation, Genesis Innovation has agreed to provide consulting services for concept development, intellectual property creation, surgeon relationship management and large-scale project management. We have entered into a software licensing agreement with Genesis Software. As a result of these roles and affiliations with Genesis Innovation and Genesis Software, a conflict of interest could rise in which our interests are different than those of Genesis Innovation and/or Genesis Software or in which the interests of certain of our officers and directors in relation to Genesis Innovation and Genesis Software are different than the interests in us and our stockholders. The interests of these officers and directors may not be fully aligned with yours, which could lead to actions that are not in your best interests. Furthermore, such officers and directors could be seen as benefiting directly or indirectly from transactions in which we will pay Genesis Innovation and/or Genesis Software for their services and offerings under our respective agreements with each party regardless of whether such agreements are believed to be in the best interests of our stockholders. In such cases, it may be difficult to manage the conflict of interest through the oversight of our board of directors.
This could give rise to a conflict of interest in which our interests are different than those of Genesis Innovation or Genesis Software or in which the interests of our officers and directors in relation to Genesis Innovation and Genesis Software are different than the interests of the company and its stockholders. In such cases, we may be unable to effectively manage the conflict of interest through the oversight of our board of directors.
Any future international expansion will subject us to additional costs and risks that may have a material adverse effect on our business, financial condition and results of operations.
Historically, all of our sales have been to customers in the United States. Our long-term strategy may include increasing our international presence, including securing regulatory approvals in targeted countries outside the United States. This strategy may include establishing and maintaining physician outreach and education capabilities outside of the United States and expanding our relationships with international payors. To the extent we enter into international markets in the future, there are significant costs and risks inherent in conducting business in international markets, including:
•Difficulties in staffing and managing our international operations;
•Multiple, conflicting and changing laws and regulations such as tax laws, privacy laws, export and import restrictions, employment laws, regulatory requirements and other governmental approvals, permits and licenses;
•Reduced or varied protection for intellectual property rights in some countries;
•Obtaining regulatory clearance where required for our products in various countries;
•Requirements to maintain data and the processing of that data on servers located within such countries;
•Complexities associated with managing multiple payor reimbursement regimes, government payors or patient self-pay systems;
•Limits on our ability to penetrate international markets if we are required to manufacture locally;
•Financial risks, such as longer payment cycles, difficulty collecting accounts receivable, foreign tax laws and complexities of foreign value-added tax systems, the effect of local and regional financial pressures on demand and payment for our products and exposure to foreign currency exchange rate fluctuations;
•Restrictions on the site-of-service for use of our products and the economics related thereto for physicians, providers and payors;
62
•Natural disasters, political and economic instability, including wars, terrorism, political unrest, outbreak of disease, pandemics and epidemics, boycotts, curtailment of trade and other market restrictions; and
•Regulatory and compliance risks that relate to maintaining accurate information and control over activities subject to regulation under the United States Foreign Corrupt Practices Act of 1977 (“FCPA”), U.K. Bribery Act of 2010 and comparable laws and regulations in other countries.
We have limited experience with regulatory environments and market practices internationally, and we may not be able to penetrate or successfully operate in new markets. We may also encounter difficulty expanding into international markets because of limited brand recognition in certain parts of the world, leading to delayed acceptance of our products by surgeons and their patients, hospitals, ASCs and payors in these international markets. For example, any perceived advantages of our products in the United States may not be perceived the same internationally. If we are unable to expand internationally and manage the complexity of international operations successfully, it could have a material adverse effect on our business, financial condition and results of operations. If our efforts to introduce our products into foreign markets are not successful, we may have expended significant resources without realizing the expected benefit. Ultimately, the investment required for expansion into foreign markets could exceed the results of operations generated from this expansion.
Risks Related to Our Intellectual Property
If we are unable to obtain and maintain significant patent or other intellectual property protection for our products, or if the scope of our patents and other intellectual property rights do not adequately protect our products, our competitors could develop and commercialize products similar or identical to ours and we may be unable to gain significant market share and be unable to operate our business profitably.
We have and will rely on patents, trade secrets, copyrights, know-how, trademarks, license agreements and contractual provisions to establish our intellectual property rights and protect our products. These legal means, however, afford only limited protection and may not completely protect our rights. Any failure to obtain or maintain patent and other intellectual property protection with respect to our products could harm our business, financial condition and results of operations. We generally seek to protect our proprietary position by filing patent applications that are important to our business. We also seek to protect our proprietary position by acquiring or in-licensing relevant issued patents or pending patent applications or other intellectual property or proprietary rights from third parties.
We cannot assure you that our intellectual property position will not be challenged or that all patents for which we have applied will be granted. The validity and breadth of claims in patents involve complex legal and factual questions and, therefore, may be highly uncertain. Uncertainties and risks that we face include the following:
•our pending or future patent applications may not result in the issuance of patents;
•the scope of any existing or future patent protection may not exclude competitors or provide competitive advantages to us;
•our patents may not be held valid or enforceable if subsequently challenged;
•other parties may claim that our products and designs infringe the proprietary rights of others—even if we are successful in defending our patents and proprietary rights, the cost of such litigation may adversely affect our business; and
•other parties may develop similar products, duplicate our products, or design around our patents.
The patent prosecution process is expensive and time-consuming, and we may not be able to file, prosecute, maintain, enforce or license all necessary or desirable patents or patent applications at a reasonable cost or in a timely manner, or in all jurisdictions. Although we enter into non-disclosure and confidentiality agreements with parties who have access to confidential or patentable aspects of our research and development output, any of these parties may breach such agreements and disclose such output before a patent application is filed, thereby jeopardizing our ability to seek and obtain patent protection. We may choose not to seek patent protection for certain innovations and may choose not to pursue patent protection in certain jurisdictions, and under the laws of certain jurisdictions, patents or other intellectual property rights may be unavailable or limited in scope. It is also possible that we will fail to identify patentable aspects of our developments before it is too late to obtain patent protection. In addition, our ability to obtain and maintain valid and enforceable patents depends in part on whether the differences between our inventions and the prior art allow our
63
inventions to be patented over the prior art. Furthermore, the publication of discoveries in scientific literature often lags behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until 18 months after filing, or in some cases not at all. Therefore, we cannot be certain that we were the first to file for patent protection of such inventions.
In addition, the laws of foreign jurisdictions may not protect our rights to the same extent as the laws of the United States. For example, most countries outside of the United States do not allow patents for methods of treating the human body. This may preclude us from obtaining method patents outside of the United States having similar scope to those we have obtained or may obtain in the future in the United States. This includes certain key method patents covering the InSet system. Changes in either the patent laws or interpretation of the patent laws in the United States and other countries may diminish our ability to protect our inventions, obtain, maintain and enforce our patent rights and, more generally, could affect the value of our patents. Additionally, we cannot predict whether the patent applications we are currently pursuing will issue as patents in any particular jurisdiction or whether the claims or any issued patents will provide sufficient protection from competitors or third parties.
Moreover, we may be subject to a third-party pre-issuance submission of prior art to the U.S. Patent and Trademark Office (“USPTO”) or patent offices in foreign jurisdictions, or become involved in opposition, derivation, reexamination, inter partes review, post-grant review or interference proceedings challenging our patent rights or the patent rights of others. An adverse determination in any such submission, proceeding or litigation could reduce the scope of, or invalidate, our patent rights, and allow third parties to commercialize our technology and compete directly with us, without payment to us.
The patent positions of companies may involve complex legal and factual questions that have been the subject of much litigation in recent years, and, therefore, the scope of any patent claims that we have or may obtain cannot be predicted with certainty. Accordingly, we cannot provide any assurances about which of our patent applications will issue, the breadth of any resulting patent, whether any of the issued patents will be found to be infringed, invalid, or unenforceable or will be threatened or challenged by third parties, or that any of our issued patents have, or that any of our currently pending or future patent applications that mature into issued patents will include, claims with a scope sufficient to protect our products, services, or technology. Our pending and future patent applications may not result in the issuance of patents or, if issued, may not issue in a form that will be advantageous to us. The coverage claimed in a patent application can be significantly reduced before the patent is issued, and its scope can be reinterpreted after issuance. We cannot offer any assurances that the breadth of our issued patents will be sufficient to stop a competitor from developing, manufacturing, and commercializing one or more products, services, or technologies in a non-infringing manner that would be competitive with one or more of our products, services, or technologies, or otherwise provide us with any competitive advantage. Furthermore, any successful challenge to these patents or any other patents owned by or licensed to us after patent issuance could deprive us of rights necessary for our commercial success. Further, there can be no assurance that we will have adequate resources to enforce our patents.
The issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, and our patents may be challenged in the courts or patent offices in the United States and abroad, and as a result, may not provide us with adequate proprietary protection or competitive advantage against competitors with similar products. Such challenges may result in loss of exclusivity or freedom to operate or in patent claims being narrowed, invalidated or held unenforceable, in whole or in part, which could limit our ability to stop others from using or commercializing similar or identical products and techniques without payment to us, or limit the duration of the patent protection of our technology.
We cannot ensure that we do not infringe any patents or other proprietary rights held by third parties. If our products were found to infringe any proprietary right of another party, we could be required to pay significant damages or license fees to such party and/or cease production, marketing and distribution of those products. Litigation may also be necessary to defend infringement claims of third parties or to enforce patent rights we hold or to protect trade secrets or techniques we own. In addition, because patent applications can take many years to issue and because publication schedules for pending applications vary by jurisdiction, there may be applications now pending of which we are unaware and which may result in issued patents which our current or future products infringe. Also, because the claims of published patent applications can change between publication and patent grant, there may be published patent applications that may ultimately issue with claims that we infringe.
We also rely on trade secrets and other unpatented proprietary technology. There can be no assurances that we can meaningfully protect our rights in our trade secrets and other unpatented proprietary technology or that others will not independently develop substantially equivalent proprietary products or processes or otherwise gain access to our proprietary technology. We seek to protect our trade secrets and proprietary know-how and technology, in part, with
64
confidentiality and invention assignment agreements with employees and consultants that include customary intellectual property assignment obligations. There can be no assurances, however, that the agreements will not be breached, adequate remedies for any breach would be available or competitors will not discover our trade secrets or independently develop comparable intellectual property. Further, litigation may be necessary to obtain ownership or to defend against claims challenging ownership. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to our management and other employees, and such claims could harm our business, financial condition and results of operations.
Some of our patents and patent applications may in the future be jointly owned with third parties. If we are unable to obtain an exclusive license to any such third-party joint owners’ interest in such patents or patent applications, such co-owners may be able to license their rights to other third parties, including our competitors, and our competitors could market competing products and technology. In addition, we may need the cooperation of any such joint owners of our patents in order to enforce such patents against third parties, and such cooperation may not be provided to us. Any of the foregoing could harm our business, financial condition and results of operations.
Obtaining and maintaining intellectual property protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental agencies, and our intellectual property protection could be reduced or eliminated for non-compliance with these requirements.
The USPTO, the United States Copyright Office (“USCO”) and various foreign governmental agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the intellectual property application process. In addition, periodic maintenance fees, renewal fees, annuity fees and various other government fees on intellectual property registrations often must be paid to the USPTO, USCO and foreign agencies over the lifetime of the intellectual property registration and/or application and any intellectual property rights we may obtain in the future. While an unintentional lapse of an intellectual property registration or application can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the registration or application, resulting in partial or complete loss of intellectual property rights in the relevant jurisdiction. Non-compliance events that could result in abandonment or lapse of a registration or application include, but are not limited to, failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. If we fail to maintain the intellectual property registrations and applications covering our products, we may not be able to stop a competitor from developing and marketing products that are the same as or similar to our products, which would have a material adverse effect on our business.
We rely on intellectual property licensed from others and may become dependent on patents or other intellectual property licensed from others in the future. If we lose our licenses for intellectual property that is important to our business, we may not be able to continue developing or selling our products.
We rely on certain licenses that give us rights to third-party intellectual property that is necessary or useful to our business, including pursuant to the License Agreement. Our rights to use such intellectual property rights in our business are or may in the future be subject to the continuation of and our compliance with the terms of the license agreements between us and each of our licensors. In addition, the agreements under which we in-license intellectual property or technology from third parties are or may in the future be complex, and certain provisions in such agreements may be susceptible to multiple interpretations. The resolution of any contract interpretation disagreement that may arise could narrow what we believe to be the scope of our rights to the relevant intellectual property or technology or increase what we believe to be our financial or other obligations under the relevant agreement, either of which could have a material adverse effect on our business, financial condition, results of operations and prospects. Moreover, if disputes over intellectual property that we have in-licensed, or in-license in the future, prevent, or impair our ability to maintain our licensing arrangements on commercially acceptable terms, we may be unable to successfully develop and commercialize the affected product candidates, which could have a material adverse effect on our business, financial condition, results of operations, and prospects. License agreements may impose various obligations on us, including the obligation to pay certain royalties. For example, we are required to pay certain royalties under our License Agreement with Genesis Software. One or more of our licensors may allege that we have breached our license agreement with them, and accordingly seek to terminate our license. As a result, we may not be able to market products that were covered by the license, which would result in the loss of significant rights, restrict our ability to commercialize certain of our products and could adversely affect our competitive business position and harm our business prospects. In addition, any claims brought against us by our licensors could be costly and time-consuming and would divert the attention of our management and key personnel from our business operations.
65
We may not identify relevant third-party patents or may incorrectly interpret the relevance, scope or expiration of a third-party patent, which might adversely affect our ability to develop and market our products.
We cannot guarantee that any of our patent searches or analyses, including but not limited to the identification of relevant patents, analysis of the scope of relevant patent claims or determination of the expiration of relevant patents, are complete or thorough, nor can we be certain that we have identified each and every third-party patent and pending application in the United States, Europe and elsewhere that is relevant to or necessary for the commercialization of our products in any jurisdiction. Patent applications in the United States, the European Union (the “EU”) and elsewhere are published approximately 18 months after the earliest filing for which priority is claimed, with such earliest filing date being commonly referred to as the priority date. Additionally, pending patent applications that have been published can, subject to certain limitations, be later amended in a manner that could cover our products or the use of our products. After issuance, the scope of patent claims remains subject to construction as determined by an interpretation of the law, the written disclosure in a patent and the patent’s prosecution history. Our interpretation of the relevance or the scope of a patent or a pending application may be incorrect, which may negatively impact our ability to market our products. We may incorrectly determine that our products are not covered by a third-party patent or may incorrectly predict whether a third party’s pending application will issue with claims of relevant scope. Our determination of the expiration date of any patent in the United States, the EU or elsewhere that we consider relevant may be incorrect, which may negatively impact our ability to develop and market our products. Our failure to identify and correctly interpret relevant patents may negatively impact our ability to develop and market our products, if approved.
We are presently party to lawsuits involving patents and other intellectual property and the possibility exists that we may in the future be party to other lawsuits or administrative proceedings involving patents or other intellectual property. If we were to lose any intellectual property lawsuits, a court could require us to pay significant damages and/or prevent us from selling our products.
The medical device industry is highly competitive and litigious with respect to patents, trademarks, trade secrets, and other intellectual property rights. Companies in the medical device industry have used intellectual property litigation to gain a competitive advantage. Our commercial success depends in part upon our ability and that of our contract manufacturers and suppliers to manufacture, market, and sell our products, and to use our proprietary technologies without infringing, misappropriating or otherwise violating the proprietary rights or intellectual property of third parties. We may in the future become party to, or be threatened with, adversarial proceedings or litigation regarding intellectual property rights with respect to our products and any future products and technology, whether or not we are actually infringing, misappropriating or otherwise violating the rights of third parties. While we take reasonable steps to ensure that we and our suppliers do not infringe upon, misappropriate, or otherwise violate the intellectual property rights of others, there may be pertinent intellectual property rights of others which we or our suppliers are presently unaware.
We are presently a party to a lawsuit involving Catalyst OrthoScience. In addition, we may in the future be party to other lawsuits or other administrative proceedings, including interference proceedings, post grant review and inter partes review before the USPTO, involving our patents or other intellectual property, regardless of merit. Third parties may assert infringement claims against us based on existing patents or patents that may be granted in the future, regardless of their merit. There is a risk that third parties may choose to engage in litigation with us to enforce or to otherwise assert their patent rights against us. Even if we believe such claims are without merit, a court of competent jurisdiction could hold that these third-party patents are valid, enforceable and infringed, which could have a negative impact on our ability to commercialize our current and any future product candidates. In order to successfully challenge the validity of any such U.S. patent in federal court, we would need to overcome a presumption of validity. As this is a high burden and requires us to present clear and convincing evidence as to the invalidity of any such U.S. patent claim, there is no assurance that a court of competent jurisdiction would invalidate the claims of any such U.S. patent. Conversely, a patent owner need only prove infringement by a preponderance of the evidence, which is a lower burden of proof. Moreover, given the vast number of patents in our field of technology, we cannot be certain that we do not infringe existing patents or that we will not infringe patents that may be granted in the future. While we may decide to initiate proceedings to challenge the validity of these or other patents in the future, we may be unsuccessful, and courts or patent offices in the United States and abroad could uphold the validity of any such patent. Furthermore, because patent applications can take many years to issue and may be confidential for 18 months or more after filing, and because pending patent claims can be revised before issuance, there may be applications now pending which may later result in issued patents that may be infringed by the manufacture, use or sale of our product candidates. Regardless of when filed, we may fail to identify relevant third-party patents or patent applications, or we may incorrectly conclude that a third-party patent is invalid or not infringed by our product candidates or activities. If a patent holder believes that our product candidate or technology platform infringes its patent, the patent holder may sue us even if we have received patent protection for our technology. Moreover, we may face patent infringement claims from nonpracticing entities that have no relevant product revenue and against whom our own patent
66
portfolio may thus have no deterrent effect. If a patent infringement suit were threatened or brought against us, we could be forced to stop or delay research, development, manufacturing or sales of the product or product candidate that is the subject of the actual or threatened suit.
If we are found to have infringed a third party’s intellectual property rights, we could be required to obtain a license from such third party to continue selling, developing and marketing our products and techniques. However, we may not be able to obtain any required license on commercially reasonable terms or at all. The acquisition or licensing of third-party intellectual property rights is a competitive area, and our competitors may pursue strategies to acquire or license third-party intellectual property rights that we may consider attractive or necessary. If we are unable to successfully obtain rights to required third party intellectual property rights or maintain the existing intellectual property rights we have, we may have to abandon development of the relevant products or redesign those products that contain the allegedly infringing intellectual property, which could harm our business, financial condition and results of operations. Even if we were able to obtain a license, it could be non-exclusive, thereby giving our competitors access to the same technologies licensed to us. We could be forced, including by court order, to cease commercializing the infringing technology or product. In addition, we could be found liable for monetary damages, including treble damages and attorneys’ fees if we are found to have willfully infringed a patent. A finding of infringement could force us to cease some of our business operations, which could materially harm our business. Claims that we have misappropriated the confidential information or trade secrets of third parties could have a similar negative impact on our business. Intellectual property litigation may lead to unfavorable publicity that harms our reputation and causes the market price of our common shares to decline. A legal proceeding, regardless of the outcome, could drain our financial resources and divert the time and effort of our management. Protracted litigation to defend or prosecute our intellectual property rights could result in our customers or potential customers deferring or limiting their purchase or use of the affected products until resolution of the litigation.
Because competition in our industry is intense, competitors may infringe or otherwise violate our issued patents, patents of our licensors or other intellectual property. To counter infringement or unauthorized use, we may be required to file infringement claims, which can be expensive and time consuming, and could distract our technical and management personnel from their normal responsibilities. Any claims we assert against perceived infringers could provoke these parties to assert counterclaims or file administrative actions against us alleging that we infringe their patents. In addition, in a patent infringement proceeding, a court may decide that a patent of ours is invalid or unenforceable, in whole or in part, construe the patent’s claims narrowly or refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover the technology in question. An adverse result in any litigation proceeding or administrative action could put one or more of our patents at risk of being invalidated or interpreted narrowly and could put our related pending patent applications at risk of not issuing. Our competitors may assert invalidity on various grounds, including lack of novelty, obviousness or that we were not the first applicant to file a patent application related to our product. We may elect to enter into license agreements in order to settle patent infringement claims or to resolve disputes before litigation, and any such license agreements may require us to pay royalties and other fees that could be significant. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure.
Our competitors, many of which have made substantial investments in patent portfolios, trade secrets, trademarks and competing technologies, may have applied for or obtained, or may in the future apply for or obtain, patents or trademarks that may prevent, limit or otherwise interfere with our ability to make, use, sell and/or export our products or to use our technologies or product names. Moreover, individuals and groups that are non-practicing entities, commonly referred to as “patent trolls,” purchase patents and other intellectual property assets for the purpose of making claims of infringement in order to extract settlements. From time to time, we may receive threatening letters, notices or “invitations to license,” or may be the subject of claims that our products and business operations infringe or violate the intellectual property rights of others. The defense of these matters can be time consuming, costly to defend in litigation, divert management’s attention and resources, damage our reputation and brand and cause us to incur significant expenses or make substantial payments. Further, the outcome of litigation is uncertain and such litigation could result in us being forced to stop making, selling or using products or technologies that allegedly infringe the asserted intellectual property; pay substantial damages or royalties to the party whose intellectual property rights we may be found to be infringing; redesign those products that contain the allegedly infringing intellectual property, which could be costly, disruptive, or infeasible; and or attempt to obtain a license to the relevant intellectual property from third parties, which may not be available on reasonable terms, or at all, or, from third parties. In addition, third parties may assert infringement claims against our customers. These claims may require us to initiate or defend protracted and costly litigation on behalf of our customers or indemnify our customers for any costs associated with their own initiation or defense of infringement claims, regardless of the merits of these claims. If any of these claims succeed or settle, we may be forced to pay damages or settlement payments on behalf of our customers or may be required to obtain licenses for the products they use. If we cannot obtain all necessary licenses on commercially reasonable terms, our customers may be forced to stop using our products.
67
Because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during litigation. There could also be public announcements of the results of hearing, motions, or other interim developments. If securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of shares of our common stock. Even if we ultimately prevail, a court may decide not to grant an injunction against further infringing activity and instead award only monetary damages, which may not be an adequate remedy. Furthermore, even if resolved in our favor, the monetary cost of such litigation and the diversion of the attention of our management could outweigh any benefit we receive as a result of the proceedings. Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could have a material adverse effect on our business. Any of the foregoing may cause us to incur substantial costs and could place a significant strain on our financial resources, divert the attention of management from our core business, and harm our reputation.
If we fail to execute invention assignment agreements with our employees and contractors involved in the development of intellectual property or are unable to protect the confidentiality of our trade secrets, the value of our products and our business and competitive position could be harmed.
In addition to patent protection, we also rely on the protection of trademarks, copyrights, trade secrets, know-how and confidential and proprietary information. We generally enter into confidentiality and invention assignment agreements with our employees, consultants and third parties upon their commencement of a relationship with us. However, we may not enter into such agreements with all employees, consultants and third parties who have been involved in the development of our intellectual property. We may also be subject to claims that our current or former employees, contractors, or other third parties have an ownership interest in our current or future patents, patent applications, or other intellectual property rights, including as an inventor or co-inventor. We may be subject to ownership or inventorship disputes in the future arising, for example, from conflicting obligations of employees, consultants, or others who were or are involved in developing our products, services, or technologies. In addition, these agreements may not provide meaningful protection against the unauthorized use or disclosure of our trade secrets or other confidential information, and adequate remedies may not exist if unauthorized use or disclosure were to occur. The exposure of our trade secrets and other proprietary information would impair our competitive advantages and could have a material adverse effect on our business, financial condition and results of operations. In particular, a failure to protect our proprietary rights may allow competitors to copy our products and procedures, which could adversely affect our pricing and market share. We may need to share our proprietary information, including trade secrets, with our current and future business partners, collaborators, contractors and others located in countries at heightened risk of theft of trade secrets, including through direct intrusion by private parties or foreign actors, and those affiliated with or controlled by state actors. As a result, we may encounter significant problems in protecting and defending our intellectual property both in the United States and abroad. The failure to obtain or maintain trade secret protection could adversely affect our competitive business position. Further, other parties may independently develop substantially equivalent know-how and technology. In addition to contractual measures, we try to protect the confidential nature of our proprietary information using commonly accepted physical and technological security measures. Such measures may not, for example, in the case of misappropriation of a trade secret by an employee or third party with authorized access, provide adequate protection for our proprietary information. Our security measures may not prevent an employee or consultant from misappropriating our trade secrets and providing them to a competitor, and recourse we take against such misconduct may not provide an adequate remedy to protect our interests fully. Unauthorized parties may also attempt to copy or reverse engineer certain aspects of our products that we consider proprietary. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret can be difficult, expensive and time-consuming, and the outcome is unpredictable.
Even though we use commonly accepted security measures, trade secret violations are often a matter of state law, and the criteria for protection of trade secrets can vary among different jurisdictions. In addition, trade secrets may be independently developed by others in a manner that could prevent legal recourse by us. While we have agreements with our employees, consultants and third parties that obligate them to assign their inventions to us, these agreements may not be self-executing, not all employees or consultants may enter into such agreements, or employees or consultants may breach or violate the terms of these agreements, and we may not have adequate remedies for any such breach or violation. If any of our intellectual property or confidential or proprietary information, such as our trade secrets, were to be disclosed or misappropriated, or if any such information was independently developed by a competitor, it could have a material adverse effect on our competitive position, business, financial condition, results of operations and prospects.
68
If our trademarks and trade names are not adequately protected, we may not be able to build name recognition in our markets of interest and our competitive position may be harmed.
We rely on our trademarks, trade names and brand names to distinguish our systems from the products of our competitors and have registered or applied to register many of these trademarks. We cannot guarantee that our trademark applications will be approved. Third parties may also oppose our trademark applications or otherwise challenge our use of the trademarks. Our registered and unregistered trademarks, trade names, and brand names may be challenged, infringed, circumvented, declared generic or determined to be violating or infringing on other marks. We may not be able to protect our rights to these trademarks, trade names, and brand names which we rely upon to build name recognition among potential partners and customers in our markets of interest. In the event that our trademarks are successfully challenged, we could be forced to rebrand our systems, which could result in loss of brand recognition, and could require us to devote resources to advertising and marketing new brands. Further, there can be no assurance that competitors will not infringe our trademarks or that we will have adequate resources to enforce our trademarks.
Patent terms may not be sufficient to effectively protect our systems and business for an adequate period of time.
Patents have a limited lifespan. In the United States, the natural expiration of a utility patent is generally 20 years after its first effective non-provisional filing date and the natural expiration of a design patent is generally 14 years after its issue date, unless the filing date occurred on or after May 13, 2015, in which case the natural expiration of a design patent is generally 15 years after its issue date. However, the actual protection afforded by a patent varies from country to country, and depends on many factors, including the type of patent, the scope of its coverage, the availability of regulatory-related extensions, the availability of legal remedies in a particular country and the validity and enforceability of the patent. Although various extensions may be available, the term of a patent, and the protection it affords, is limited. We may also be required to disclaim a portion of a patent term in order to overcome double patenting rejections from the patent office, thus potentially shortening our exclusivity period. Even if patents covering our proprietary technologies and their uses are obtained, once the patent has expired, we may be open to competition, which may harm our business prospects. In addition, although upon issuance in the United States a patent’s term can be extended based on certain delays caused by the USPTO, this extension can be reduced or eliminated based on certain delays caused by the patent applicant during patent prosecution. A patent term extension based on regulatory delay may be available in the United States. However, only a single patent can be extended for each marketing approval, and any patent can be extended only once, for a single product. Moreover, the scope of protection during the period of the patent term extension does not extend to the full scope of the claim, but instead only to the scope of the product as approved. Laws governing analogous patent term extensions in foreign jurisdictions vary widely, as do laws governing the ability to obtain multiple patents from a single patent family. Additionally, we may not receive an extension if we fail to exercise due diligence during the testing phase or regulatory review process, apply within applicable deadlines, fail to apply prior to expiration of relevant patents or otherwise fail to satisfy applicable requirements. Given the amount of time required for the development, testing and regulatory review of new products, patents protecting such products might expire before or shortly after such products are commercialized. If we do not have sufficient patent terms to protect our products, proprietary technologies and their uses, our business would be seriously harmed. As our patents expire, the scope of our patent protection will be reduced, which may reduce or eliminate any competitive advantage afforded by our patent portfolio. As a result, our reduced patent portfolio may not provide us with sufficient rights to exclude others from commercializing products similar or identical to ours.
Changes in U.S. or foreign patent laws or their interpretation may limit our ability to obtain, maintain, defend and/or enforce our patents.
Patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents. The Leahy-Smith America Invents Act (“Leahy-Smith Act”) includes a number of significant changes to U.S. patent law. These include provisions that affect the way patent applications are prosecuted and also affect patent litigation. The USPTO has developed regulations and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act, and in particular, the first to file provisions, which became effective on March 16, 2013. The first to file provisions limit the rights of an inventor to patent an invention if not the first to file an application for patenting that invention, even if such invention was the first invention. Accordingly, it is not clear what, if any, impact the Leahy-Smith Act will have on the operation of our business.
However, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the enforcement and defense of our issued patents. For example, the Leahy-Smith Act provides that an administrative tribunal known as the Patent Trial and Appeals Board (“PTAB”), provides a venue for challenging the validity of patents at a cost that is much lower than district court litigation and on timelines that are much faster. Although it is not clear what, if any,
69
long-term impact the PTAB proceedings will have on the operation of our business, the initial results of patent challenge proceedings before the PTAB since its inception in 2013 have resulted in the invalidation of many U.S. patent claims. The availability of the PTAB as a lower-cost, faster and potentially more potent tribunal for challenging patents could increase the likelihood that our own patents will be challenged, thereby increasing the uncertainties and costs of maintaining and enforcing them.
The Leahy-Smith Act also includes a number of significant changes that affect the way U.S. patent applications will be prosecuted and also may affect patent litigation. These include allowing third-party submission of prior art to the USPTO during patent prosecution and additional procedures to attack the validity of a patent by the USPTO administered post-grant proceedings, including post-grant review, inter partes review and derivation proceedings.
Because of a lower evidentiary standard in USPTO proceedings compared to the evidentiary standard in U.S. federal courts necessary to invalidate a patent claim, a third party could potentially provide evidence in a USPTO proceeding sufficient for the USPTO to hold a claim invalid even though the same evidence would be insufficient to invalidate the claim if first presented in a district court action. Therefore, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents. In addition, future actions by the U.S. Congress, the federal courts and the USPTO could cause the laws and regulations governing patents to change in unpredictable ways. Any of the foregoing could harm our business, financial condition and results of operations.
In addition, on June 1, 2023, the European Union Patent Package, or the EU Patent Package, regulations were implemented with the goal of providing a single pan-European Unitary Patent and a new European Unified Patent Court (the “UPC”), for litigation involving European patents. As a result, all European patents, including those issued prior to ratification of the EU Patent Package, now by default automatically fall under the jurisdiction of the UPC, unless otherwise opted out. It is uncertain how the UPC will impact granted European patents in the biotechnology and pharmaceutical industries. Our European patent applications, if issued, could be challenged in the UPC. During the first seven years of the UPC’s existence, the UPC legislation allows a patent owner to opt its European patents out of the jurisdiction of the UPC. We have opted out certain of our European patents and may decide to opt out our future European patents from the UPC, but doing so may preclude us from realizing the benefits of the UPC. Moreover, if we do not meet all of the formalities and requirements for opt-out under the UPC, our current or future European patents could remain under the jurisdiction of the UPC. The UPC will provide our competitors with a new forum to centrally revoke our European patents, and allow for the possibility of a competitor to obtain pan-European injunction. Such a loss of patent protection could have a material adverse impact on our business and our ability to commercialize our technology and our systems due to increased competition and, resultantly, on our business, financial condition, results of operations and prospects. The UPC and Unitary Patent are significant changes in European patent practice. As the UPC is a new court system, there is no precedent for the court, increasing the uncertainty of any litigation in the UPC.
We have foreign intellectual property rights and may be unable to enforce our intellectual property rights throughout the world.
We have intellectual property rights outside the United States. Filing, prosecuting and defending patents or trademarks on our systems and any future products in all countries throughout the world would be prohibitively expensive, and the laws of foreign countries may not protect our rights to the same extent as the laws of the United States. Consequently, we may not be able to prevent third parties from practicing our inventions or utilizing our trademarks in all countries outside the United States, or from selling or importing products made using our inventions in and into the United States or other jurisdictions. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and, further, may export otherwise infringing products to territories where we have patent protection but enforcement is not as strong as that in the United States. These products may compete with our products and any future products, and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing in these jurisdictions.
The laws of some foreign countries do not protect intellectual property rights to the same extent as the laws of the United States. Many companies have encountered significant problems in protecting and defending intellectual property rights in certain foreign jurisdictions. This could make it difficult for us to stop infringement of our foreign patents, if obtained, or the misappropriation of our other intellectual property rights. For example, some foreign countries have compulsory licensing laws under which a patent owner must grant licenses to third parties. In addition, some countries limit the enforceability of patents against third parties, including government agencies or government contractors. In these countries, patents may provide limited or no benefit. Patent protection must ultimately be sought on a country-by-country basis, which is an expensive and time-consuming process with uncertain outcomes. Accordingly, we may choose not to seek patent protection in certain countries, and we will not have the benefit of patent protection in such countries.
70
Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business. Accordingly, our efforts to protect our intellectual property rights in such countries may be inadequate. In addition, changes in the law and legal decisions by courts in the United States and foreign countries may affect our ability to obtain adequate protection for our technology and the enforcement of our intellectual property.
The legal systems in certain countries may also favor state-sponsored or companies headquartered in particular jurisdictions over our first-in-time patents and other intellectual property protection. We are aware of incidents where such entities have stolen the intellectual property of domestic companies in order to create competing products and we believe we may face such circumstances ourselves in the future. For example, through its “Annual Special 301 Report on Intellectual Property,” the Office of the United States Trade Representative has been reporting on the adequacy and effectiveness of intellectual property protection in a number of foreign countries that are U.S. trading partners and their protection and enforcement of intellectual property rights. Placement of a country on the Priority Watch List indicates that particular problems exist in that country with respect to intellectual property protection, enforcement, or market access for persons relying on intellectual property rights. Countries placed on the Priority Watch List are the focus of increased bilateral attention concerning the specific problem areas. It is possible that we will not be able to enforce our intellectual property rights against third parties that misappropriate our proprietary technology in those countries.
Moreover, geopolitical actions in the U.S. and in foreign countries could increase the uncertainties and costs surrounding the prosecution or maintenance of our patent applications or those of any current or future licensors and the maintenance, enforcement or defense of our issued patents or those of any current or future licensors. Government actions may also prevent maintenance of issued patents in Russia. These actions could result in abandonment or lapse of our future Russian patents or patent applications, resulting in partial or complete loss of patent rights in Russia. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and may export otherwise infringing products to territories where we have patent protection, but enforcement rights are not as strong as those in the United States. These products may compete with our products and our patents or other IP rights may not be effective or sufficient to prevent them from competing.
We may in the future be subject to claims that we or our employees have misappropriated the intellectual property of a third party, including trade secrets or know-how, or are in breach of non-competition or non-solicitation agreements with our competitors and third parties may claim an ownership interest in intellectual property we regard as our own.
We could in the future be subject to claims that we or our employees have inadvertently or otherwise used or disclosed alleged trade secrets or other confidential information of former employers or competitors. Many of our employees and consultants were previously employed at or engaged by other medical device companies, including our competitors or potential competitors. Some of these employees, consultants and contractors may have executed proprietary rights, non-disclosure and non-competition agreements in connection with such previous employment. Although we try to ensure that our employees and consultants do not use the intellectual property, proprietary information, know-how or trade secrets of others in their work for us, we may be subject to claims that we or these individuals have, inadvertently or otherwise, misappropriated the intellectual property or disclosed the alleged trade secrets or other proprietary information, of these former employers, competitors or other third parties. Additionally, we may be subject to claims from third parties challenging our ownership interest in or inventorship of intellectual property we regard as our own, for example, based on claims that our agreements with employees or consultants obligating them to assign intellectual property to us are ineffective or in conflict with prior or competing contractual obligations to assign inventions to another employer, to a former employer, or to another person or entity. Litigation may be necessary to defend against claims, and it may be necessary or we may desire to enter into a license to settle any such claim; however, there can be no assurance that we would be able to obtain a license on commercially reasonable terms, if at all. If our defense to those claims fails, in addition to paying monetary damages or a settlement payment, a court could prohibit us from using technologies, features or other intellectual property that are essential to our products, if such technologies or features are found to incorporate or be derived from the trade secrets or other proprietary information of the former employers. An inability to incorporate technologies, features or other intellectual property that are important or essential to our products could have a material adverse effect on our business and competitive position and may prevent us from selling our products. In addition, we may lose valuable intellectual property rights or personnel. Even if we are successful in defending against these claims, litigation could result in substantial costs and could be a distraction to management. Any litigation or the threat thereof may adversely affect our ability to hire employees or contract with independent sales representatives. A loss of key personnel or their work product could hamper or prevent our ability to commercialize our products, which could materially and adversely affect our business, financial condition, operating results, cash flows and prospects.
71
If our third-party manufacturing partners do not respect our intellectual property and trade secrets and produce competitive products using our designs or intellectual property, our business, financial condition and results of operations would be harmed.
Although our agreements with third-party manufacturing partners generally seek to prevent them from misusing our intellectual property and trade secrets, or using our designs to manufacture products for our competitors, we may be unsuccessful in monitoring and enforcing our intellectual property rights against these manufacturing partners and may find counterfeit goods in the market being sold as our systems or future products similar to ours produced for our competitors using our intellectual property. Although we take steps to stop counterfeits, we may not be successful and network operators who purchase these counterfeit goods may experience product defects or failures, harming our reputation and brand and causing us to lose future sales. Any of the foregoing could harm our business, financial condition and results of operations.
Our use of open source software could impose limitations on our ability to commercialize our product.
We may use open source software in the future that contains modules licensed for use from third-party authors under open source licenses. Some of the software may be provided under license arrangements that allow use of the software for research or other non-commercial purposes. As a result, in the future, as we seek to use our platform in connection with commercially available products, we may be required to license that software under different license terms, which may not be possible on commercially reasonable terms, if at all. If we are unable to license software components on terms that permit its use for commercial purposes, we may be required to replace those software components, which could result in delays, additional cost and/or additional regulatory approvals.
Use and distribution of open source software may entail greater risks than use of third-party commercial software, as open source licensors generally do not provide warranties or other contractual protections regarding infringement claims or the quality of the software code. Some open source licenses contain requirements that we make available source code for modifications or derivative works we create based upon the type of open source software we use. If we combine our proprietary software with open source software in a certain manner, we could, under certain of the open source licenses, be required to release the source code of our proprietary software to the public. This could allow our competitors to create similar products with lower development effort and time, and ultimately could result in a loss of product sales for us. Although we monitor our use of open source software, the terms of many open source licenses have not been interpreted by U.S. courts, and there is a risk that those licenses could be construed in a manner that could impose unanticipated conditions or restrictions on our ability to commercialize our product candidates. We could be required to seek licenses from third parties in order to continue offering our product candidates, to re-engineer our product candidates or to discontinue the sale of our product candidates in the event re-engineering cannot be accomplished on a timely basis, any of which could materially and adversely affect our business, financial condition, results of operations and prospects.
Intellectual property rights do not necessarily address all potential threats to our competitive advantage.
The degree of future protection afforded by our intellectual property rights is uncertain because intellectual property rights have limitations and may not adequately protect our business or permit us to maintain our competitive advantage. For example:
•others may be able to make products that are similar to ours but that are not covered by the claims of the patents that we own or have exclusively licensed;
•we or our licensors or future collaborators might not have been the first to make the inventions covered by the issued patent or pending patent application that we own or have exclusively licensed;
•we or our licensors or future collaborators might not have been the first to file patent applications covering certain of our inventions;
•others may independently develop similar or alternative technologies or duplicate any of our technologies without infringing our intellectual property rights;
•it is possible that our pending patent applications will not lead to issued patents;
•issued patents that we own or have exclusively licensed may be held invalid or unenforceable, as a result of legal challenges by our competitors;
72
•our competitors might conduct research and development activities in countries where we do not have patent rights and then use the information learned from such activities to develop competitive products for sale in our major commercial markets;
•we may not develop additional proprietary technologies that are patentable;
•we cannot predict the scope of protection of any patent issuing based on our patent applications, including whether the patent applications that we own or in-license will result in issued patents with claims that cover our products or uses thereof in the United States or in other foreign countries;
•the claims of any patent issuing based on our patent applications may not provide protection against competitors or any competitive advantages, or may be challenged by third parties;
•if enforced, a court may not hold that our patents are valid, enforceable and infringed;
•we may need to initiate litigation or administrative proceedings to enforce and/or defend our patent rights which will be costly whether we win or lose;
•we may choose not to file a patent in order to maintain certain trade secrets or know-how, and a third party may subsequently file a patent covering such intellectual property;
•the laws of foreign countries may not protect our or our licensors’, as the case may be, proprietary rights to the same extent as the laws of the United States;
•the inventors of our owned or in-licensed patents or patent applications may become involved with competitors, develop products or processes which design around our patents or become hostile to us or the patents or patent applications on which they are named as inventors;
•it is possible that our owned or in-licensed patents or patent applications omit individual(s) that should be listed as inventor(s) or include individual(s) that should not be listed as inventor(s), which may cause these patents or patents issuing from these patent applications to be held invalid or unenforceable;
•we have engaged in scientific collaborations in the past, and will continue to do so in the future. Such collaborators may develop adjacent or competing products to ours that are outside the scope of our patents;
•we may fail to adequately protect and police our trademarks and trade secrets; and
•the patents of others may have an adverse effect on our business, including if others obtain patents claiming subject matter similar to or improving that covered by our patents and patent applications.
Should any of these events occur, they could significantly harm our business, results of operations and prospects.
Risks Related to Regulatory Matters
Our devices and operations are subject to extensive government regulation and oversight in the United States, and our failure to comply with applicable requirements could harm our business.
Our systems are regulated as medical devices in the United States. Medical devices and their manufacturers and product developers are subject to extensive regulation in the United States, including by the FDA. The FDA regulates, among other things, with respect to medical devices: design, development, and manufacturing; testing, labeling, content, and language of instructions for use and storage; packaging; clinical trials; product safety; establishment registration and device listing; marketing/promotion, sales, and distribution; premarket clearance, classification, and approval or certification; recordkeeping procedures; recalls and field safety corrective actions; post-market surveillance, including reporting of deaths or serious injuries and malfunctions that, if they were to recur, could lead to death or serious injury; post-market studies; and product import and export.
The regulations to which we are subject are complex, burdensome to understand and apply, and have tended to become more stringent over time. Regulatory changes could result in restrictions on our ability to carry on or expand our operations, higher than anticipated costs, or lower than anticipated sales. The FDA enforces its regulatory requirements through, among other means, periodic unannounced inspections. We do not know whether we or any of our contract manufacturers or commercial partners will be found compliant in connection with any future FDA or foreign inspections.
73
Failure to comply with applicable regulations could jeopardize our ability to sell our systems and result in enforcement actions such as: warning letters or untitled letters; fines; injunctions; civil penalties; termination of distribution; import alerts; recalls or seizures of products; delays in the introduction of products into the market; total or partial suspension of production; refusal to grant future clearances or approvals; withdrawals or suspensions of clearances or approvals, resulting in prohibitions on sales of our systems; and in the most serious cases, criminal penalties.
Our relationships with customers, physicians and third-party payors are subject to federal and state health care fraud and abuse laws, false claims laws, physician payment transparency laws and other health care laws and regulations. If we or our employees, independent contractors, consultants, commercial partners, or vendors violate these laws we could face substantial penalties.
Our relationships with customers, physicians and third-party payors are subject to federal and state health care fraud and abuse laws, false claims laws, physician payment transparency laws and other health care laws and regulations. In particular, the promotion, sales and marketing of health care items and services is subject to extensive laws and regulations designed to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive and other business arrangements. The U.S. health care laws and regulations that may affect our ability to operate include, but are not limited to:
•the federal Anti-Kickback Statute, which prohibits, among other things, any person or entity from knowingly and willfully, offering, paying, soliciting or receiving any remuneration, directly or indirectly, overtly or covertly, in cash or in kind, to induce or reward, or in return for, the purchasing, leasing, ordering, arranging for or recommending the purchase, lease, or order of any item or service reimbursable under Medicare, Medicaid or other federal health care programs. The term “remuneration” has been broadly interpreted to include anything of value. Although there are a number of statutory exceptions and regulatory safe harbors protecting some common activities from prosecution, the exceptions and safe harbors are drawn narrowly. Practices that may be alleged to be intended to induce the purchases or recommendations, include any payments of more than fair market value, may be subject to scrutiny if they do not qualify for an exception or safe harbor. In addition, a person or entity does not need to have actual knowledge of this statute or specific intent to violate it in order to have committed a violation;
•federal civil and criminal false claims laws, including the federal civil False Claims Act, and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment or approval from Medicare, Medicaid or other federal government programs that are false or fraudulent; knowingly making, using, or causing to be made or used, a false record or statement material to a false or fraudulent claim or obligation to pay or transmit money or property to the federal government; or knowingly making a false statement or representation of a material fact to improperly avoid, decrease or conceal an obligation to pay money to the federal government, including federal health care programs. In addition, the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal civil False Claims Act;
•the federal Health Insurance Portability and Accountability Act of 1996 (“HIPAA”) which created new federal civil and criminal statutes that prohibit knowingly and willfully executing, or attempting to execute, a scheme to defraud any health care benefit program or obtain, by means of false or fraudulent pretenses, representations, or promises, any of the money or property owned by, or under the custody or control of, any health care benefit program, including private third-party payors, and knowingly and willfully falsifying, concealing or covering up by any trick, scheme or device, a material fact or making any materially false, fictitious or fraudulent statements in connection with the delivery of, or payment for, health care benefits, items or services. Similar to the federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation;
•federal consumer protection and unfair competition laws, which broadly regulate marketplace activities and activities that potentially harm consumers;
•the federal Physician Payments Sunshine Act, which requires certain manufacturers of drugs, devices, biologicals and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program (with certain exceptions) to report annually to the government information related to payments or other transfers of value made to physicians (defined to include doctors, dentists, optometrists, podiatrists and
74
chiropractors), certain other health care professionals (including physician assistants, nurse practitioners, clinical nurse specialists, certified nurse anesthetists, anesthesiologist assistants and certified nurse midwives) and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members; and
•state and foreign equivalents of each of the health care laws described above, among others, some of which may be broader in scope including, without limitation, state anti-kickback and false claims laws that may apply to sales or marketing arrangements and claims involving health care items or services reimbursed by non-governmental third party payors, including private insurers, or that apply regardless of payor (including cash payments); state laws that require device companies to comply with the industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government; state laws that require device manufacturers to report information related to payments and other transfers of value to physicians and other health care providers, and marketing expenditures; state laws that prohibit fee-splitting arrangements between companies and physicians and other health care professionals; and state and local laws requiring the registration of device sales and medical representatives. Greater scrutiny of marketing practices in the medical device industry has resulted in numerous government investigations by various government authorities, and this industry-wide enforcement activity is expected to continue. The shifting regulatory environment, along with the requirement to comply with multiple jurisdictions with different and difficult compliance and reporting requirements, increases the possibility that we may run afoul of one or more laws. The costs to comply with these regulatory requirements are becoming more expensive and will also impact our profitability.
Because of the breadth of these laws and the narrowness of the statutory exceptions and regulatory safe harbors available, it is possible that some of our business activities, including our arrangements with physicians, independent distributors and customers, could be subject to challenge under one or more of such laws. We have entered into advisory board and consulting agreements with physicians, including some who have ownership interests in us and/or influence the ordering of or use our systems in procedures they perform. Compensation under some of these arrangements includes the provision of stock or stock options. Efforts to ensure that our business arrangements will comply with applicable health care laws may involve substantial costs. It is possible that governmental and enforcement authorities will conclude that our business practices may not comply with current or future statutes, regulations or case law interpreting applicable fraud and abuse or other health care laws and regulations.
If we or our employees, agents, independent contractors, consultants, commercial partners and vendors violate these laws, we may be subject to investigations, enforcement actions and/or significant penalties, including the imposition of significant civil, criminal and administrative penalties, damages, disgorgement, monetary fines, imprisonment, possible exclusion from participation in Medicare, Medicaid and other federal health care programs, contractual damages, reputational harm, diminished profits and future earnings, additional reporting requirements and/or oversight if we become subject to a corporate integrity agreement or similar agreement to resolve allegations of non-compliance with these laws, and curtailment of our operations, any of which could adversely affect our ability to operate our business and our results of operations.
Failure to maintain marketing authorizations for our systems, or to timely obtain necessary marketing authorizations for our future products, may have a material adverse effect on our business, financial condition, results of operations, and prospects.
In the United States, before we can market a new medical device, or a new use of, or other significant modification to an existing, marketed medical device, we must first receive either clearance under Section 510(k) of the FDCA, approval of a premarket approval application (“PMA”), or grant of a de novo classification request from the FDA, unless an exemption applies. In the 510(k) clearance process, before a device may be marketed, the FDA must determine that a proposed device is “substantially equivalent” to a legally-marketed “predicate” device, which includes a device that has been previously cleared through the 510(k) process, a device that was legally marketed prior to May 28, 1976 (pre-amendments device), a device that was originally on the U.S. market pursuant to an approved PMA and later down-classified, or a 510(k)-exempt device. To be “substantially equivalent,” the proposed device must have the same intended use as the predicate device, and either have the same technological characteristics as the predicate device or have different technological characteristics and not raise different questions of safety or effectiveness than the predicate device. Clinical data are sometimes required to support substantial equivalence. In the process of obtaining PMA approval, the FDA must determine that a proposed device is safe and effective for its intended use based, in part, on extensive data, including, but not limited to, technical, pre-clinical, clinical trial, manufacturing, and labeling data. The PMA process is typically required for devices that are deemed to pose the greatest risk, such as life-sustaining, life-supporting, or implantable devices.
75
In the de novo classification process, a manufacturer whose novel device under the FDCA would otherwise be automatically classified as Class III and require the submission and approval of a PMA prior to marketing is able to request down-classification of the device to Class I or Class II on the basis that the device presents a low or moderate risk. If the FDA grants the de novo classification request, the applicant will receive authorization to market the device. This device type may be used subsequently as a predicate device for future 510(k) submissions.
The PMA approval, 510(k) clearance and de novo classification processes can be expensive, lengthy, and uncertain. The FDA’s 510(k) clearance process usually takes from three to eight months, but can take longer. The process of obtaining a PMA is much more costly and uncertain and generally takes from one to three years, or even longer, from the time the application is submitted to the FDA. In addition, a PMA generally requires the performance of one or more clinical trials. Clinical data is also required to support a de novo request. In some cases, clinical data may be required in connection with an application for 510(k) clearance. Despite the time, effort and cost, a device may not obtain marketing authorization by the FDA. We have obtained 510(k) clearances for our commercialized medical devices and we must obtain marketing authorization for any future devices we develop, unless they are exempt. Marketing authorizations for any of our future products, if granted, may include significant limitations on the indicated uses for the device, which may limit the potential commercial market for the device.
In the United States, any modification to a medical device for which we have obtained marketing authorization may require us to submit a new 510(k) premarket notification and obtain clearance, to submit a PMA and obtain FDA approval, or to submit a de novo request prior to implementing the change. For example, any modification to a 510(k)-cleared device that could significantly affect its safety or effectiveness, or that would constitute a major change in its intended use, design or manufacture, generally requires a new 510(k) clearance or other marketing authorization. The FDA requires every manufacturer to make such determinations in the first instance, but the FDA may review any manufacturer’s decision. The FDA may not agree with a manufacturer’s decisions regarding whether new clearances or approvals are necessary. We have made modifications to 510(k)-cleared products in the past and have determined based on our review of the applicable FDA regulations and guidance that in certain instances new marketing submissions were not required and the modified devices could be appropriately sold pursuant to internal documentation and, where appropriate, validation testing. We may make modifications or add additional features in the future to our medical devices that we believe do not require a new premarket submission. If the FDA disagrees with our determination and requires us to seek new marketing authorizations for such modifications, we may be required to cease marketing or to recall the modified product until we obtain such marketing authorization, and we may be subject to significant regulatory fines or penalties. If the FDA requires us to go through a lengthier, more rigorous examination for future products or modifications to existing products than we had expected, product introductions or modifications could be delayed or canceled, which could adversely affect our business.
The FDA can delay, limit or deny marketing authorization of a device for many reasons, including:
•our inability to demonstrate to the satisfaction of the FDA that our products are substantially equivalent to a predicate device or are safe and effective for their intended uses;
•the disagreement of the FDA with the design or implementation of clinical trials or the interpretation of data from preclinical studies or clinical trials;
•serious and unexpected adverse device effects experienced by participants in clinical trials;
•insufficiency of the data from preclinical studies and clinical trials to support clearance, de novo classification, or approval;
•our inability to demonstrate that the clinical and other benefits of the device outweigh the risks; and
•the manufacturing process or facilities we use may not meet applicable requirements.
We are subject to ongoing regulatory review and scrutiny. Failure to comply with post-marketing regulatory requirements could subject us to enforcement actions, including substantial penalties, and might require us to recall or withdraw a product from the market.
We are subject to ongoing and extensive regulatory requirements governing, among other things, the manufacture, marketing, promotion, advertising, medical device reporting, sale, import, export, registration, and listing of devices. For example, medical device manufacturers must submit certain reports to the FDA and keep required records as a condition of obtaining and maintaining marketing authorization. These reports include information about failures and certain adverse events potentially associated with the device after its marketing authorization. Failure to submit such reports, or failure to
76
submit the reports in a timely manner, could result in enforcement action by the FDA. Following its review of the periodic reports, the FDA might ask for additional information or initiate further investigation.
Regulatory changes could result in restrictions on our ability to continue or expand our operations, higher than anticipated costs, or lower than anticipated sales. We have ongoing responsibilities under FDA regulations, and the FDA and state regulatory authorities have broad enforcement powers. Our failure to comply with applicable regulatory requirements could result in enforcement action by the FDA or state regulatory authorities, which may include any of the following or other sanctions:
•untitled letters or warning letters;
•fines, injunctions, consent decrees, and civil penalties;
•recalls, termination of distribution, administrative detention, or seizure of our products;
•customer notifications or repair, replacement, or refunds;
•operating restrictions or partial suspension or total shutdown of production;
•delays in or refusal to grant our requests for future clearances, de novo classifications, or PMA approvals, or comparable foreign marketing authorizations of new products, new intended uses, or modifications to existing products;
•withdrawals or suspensions of any granted marketing authorizations, resulting in prohibitions on sales of our products;
•FDA refusal to issue certificates to foreign governments needed to export products for sale in other countries; and
•criminal prosecution.
Any of these sanctions could result in negative publicity, higher than anticipated costs, or lower than anticipated sales, and have a material adverse effect on our reputation, business, financial condition, results of operations, and prospects.
In addition, the FDA may change its policies which may affect future products by rendering our clinical data or regulatory filings insufficient for marketing authorization. The FDA may adopt additional regulations or revise existing regulations, or take other actions, which may prevent or delay marketing authorization of any products under development or impact our ability to modify any products authorized for market on a timely basis. Such changes may also occur in foreign jurisdictions where we may market our products in the future. Such changes could impose additional requirements upon us that could delay our ability to obtain future marketing authorizations, increase the costs of compliance, or restrict our ability to maintain any marketing authorizations we have obtained.
Our products must be manufactured in accordance with applicable laws and regulations, and we could be forced to recall our devices or terminate production if we fail to comply with these regulations.
In the United States, the methods used in, and the facilities used for, the manufacture of medical devices must comply with the FDA’s cGMPs for medical devices, known as the QMSR, which is a complex regulatory scheme that covers the procedures and documentation of the design, testing, production, process controls, quality assurance, labeling, packaging, handling, storage, distribution, installation, servicing, and shipping of medical devices. The QMSR, which went into effect on February 2, 2026, replaces the QSR, and, among other things, incorporates by reference the quality management system requirements of ISO 13485:2016. Furthermore, we are required to verify that our suppliers maintain facilities, procedures, and operations that comply with our quality standards and applicable regulatory requirements. The FDA enforces the QMSR through periodic announced or unannounced inspections of medical device manufacturing facilities, which may include the facilities of subcontractors. Our products are also subject to similar state regulations governing manufacturing.
Despite our efforts to ensure compliance, our third-party manufacturers may not take the necessary steps to comply with applicable regulations, which could cause delays in the delivery of our medical devices. In addition, failure to comply with applicable FDA requirements or later discovery of previously unknown problems with our products or manufacturing processes could result in, among other things: warning letters or untitled letters; fines, injunctions, or civil penalties; suspension or withdrawal of marketing authorizations; seizures or recalls of our products; total or partial suspension of production or distribution; administrative or judicially imposed sanctions; the FDA’s refusal to grant pending or future clearances or approvals for our products or similar decisions by foreign regulatory authorities or notified bodies; clinical
77
holds; refusal to permit the import or export of our products; and criminal prosecution of us, our suppliers, or our employees. Any of these actions could significantly and negatively affect supply of our products. If any of these events occurs, our reputation could be harmed, we could be exposed to product liability claims, and we could lose customers and experience reduced sales and increased costs.
Legislative or regulatory reforms may have a material adverse effect on us.
From time to time, legislation is drafted and introduced in Congress that could significantly change the statutory provisions governing the regulation of medical devices. In addition, the FDA may change its clearance and approval policies, adopt additional regulations, or revise existing regulations, or take other actions, which may prevent or delay marketing authorization of any future products under development or impact our ability to modify any products for which we have already obtained marketing authorizations on a timely basis. For example, on January 31, 2024, the FDA issued a final rule to amend the QSR, which establishes cGMP requirements for medical device manufacturers, to align more closely with the ISO standards. Specifically, this final rule, which went into effect on February 2, 2026, replaces the QSR with the QMSR, and among other things, incorporates by reference the quality management system requirements of ISO 13485:2016. It is unclear the extent to which any other legislative or regulatory proposal, if adopted, could impose additional or different regulatory requirements on us that could increase the costs of compliance or otherwise create competition that may negatively affect our business.
In addition, FDA regulations and guidance are often revised or reinterpreted by the FDA in ways that may significantly affect our business and our products. Any new statutes, regulations or revisions or reinterpretations of existing regulations may make it more difficult and costly to manufacture, market, or distribute our commercialized products, or may impose additional costs, lengthen marketing authorization review times, or make it more difficult to obtain marketing authorizations for any future products we develop. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may be subject to enforcement action and we may not achieve or sustain profitability.
In response to perceived increases in health care costs in recent years, there have been and continue to be proposals by the federal government, state governments, regulators and third-party payors to control these costs and, more generally, to reform the U.S. health care system. Certain of these proposals could limit the prices we will be able to charge for our products or the amount of reimbursement available for our products and could limit the acceptance and availability of our products.
In March 2010, the federal government enacted the ACA. Among other provisions, the ACA established new value-based payment programs, increased funding of comparative effectiveness research, reduced hospital payments for avoidable readmissions and hospital acquired conditions, and pilot programs to evaluate alternative payment methodologies that promote care coordination (such as bundled physician and hospital payments). Since its enactment, there have been judicial, executive and Congressional challenges to certain aspects of the ACA. On June 17, 2021, the U.S. Supreme Court dismissed the most recent judicial challenge to the ACA brought by several states without specifically ruling on the constitutionality of the ACA.
Other legislative changes have been proposed and adopted since the ACA was enacted. The Budget Control Act of 2011, among other things, reduced Medicare payments to providers, effective on April 1, 2013 and, due to subsequent legislative amendments to the statute, will remain in effect through 2032, with the exception of a temporary suspension from May 1, 2020 through March 31, 2022, unless additional Congressional action is taken.
There are additional state and federal health care reform measures under consideration that may be adopted in the future which could have a material adverse effect on our industry generally and on our customers. We cannot predict what health care programs and regulations will be ultimately implemented at the federal or state level, particularly given the recent change in administration, or the effect of any future legislation or regulation. However, any regulatory and legal changes that lower reimbursement for our products, increase taxes on our medical devices, increase cost containment pressures on us or others in the health care sector, or reduce medical procedure volumes could adversely affect our business, financial condition, results of operations or cash flows.
The misuse or off-label use of our systems may result in injuries that harm patients and lead to product liability suits, harm our reputation in the marketplace, or result in costly investigations, fines, or sanctions by regulatory bodies if we are deemed to have engaged in the promotion of these uses, any of which could be costly to our business.
Our systems that we currently commercialize, and any marketing authorization we may receive for future products, are, and will be, limited to specified indications for use. Our dedicated commercial leadership team and marketing
78
personnel are trained to not promote our devices for uses outside of the FDA-authorized indications for use, known as “off-label uses.” We cannot, however, prevent a healthcare professional from using our devices off-label, when in the healthcare professional’s independent professional judgment he or she deems it appropriate. There may be increased risk of injury to patients if healthcare professionals attempt to use our devices off-label, which could harm our reputation in the marketplace among healthcare professionals and patients.
If the FDA determines that our promotional materials or training constitute promotion of an off-label use, it could request that we modify our training or promotional materials or subject us to regulatory or enforcement actions, including the issuance or imposition of an untitled letter (used for violators that do not necessitate a warning letter), a warning letter, injunction, seizure, civil fine, or criminal penalties. It is also possible that other federal or state enforcement authorities might take action under other regulatory authority, such as false advertising and consumer protection laws, or false claims laws, if they consider our business activities to constitute promotion of an off-label use, which could result in significant penalties, including, but not limited to, criminal, civil, and administrative penalties, damages, fines, disgorgement, exclusion from participation in government healthcare programs, and the curtailment of our operations.
In addition, healthcare professionals may misuse our products or use improper techniques if they are not adequately trained, potentially leading to injury and an increased risk of product liability. If our devices are misused or used with improper technique, we may become subject to costly litigation by our customers or their patients. As described above, product liability claims could divert management’s attention from our core business, be expensive to defend, and result in sizeable damage awards against us that may not be covered by insurance, all of which would have a material adverse effect on our business, financial condition, results of operations, and prospects.
Our implant systems and other future products may cause or contribute to adverse medical events or be subject to failures or malfunctions which we may be required to report to the FDA, and if we fail to do so, we would be subject to sanctions that could harm our reputation, business, financial condition, results of operations, and prospects. In addition, the discovery of serious safety issues, or a voluntary or required recall, could have a negative impact on us.
It is possible that there may be side effects and adverse events associated with the use of our implant systems or any future products we develop. We have experienced a modest increase in the number of direct patient complaints relative to the number of procedures performed using our systems. The FDA’s medical device reporting regulations require us to assess reportability of adverse events that come to our attention and report to the FDA when we receive or become aware of information that reasonably suggests that one or more of our products may have caused or contributed to a death or serious injury or malfunctioned in a way that, if the malfunction were to recur, it could cause or contribute to a death or serious injury. The timing of our obligation to report is triggered by the date we become aware of the adverse event as well as the nature of the event. We may fail to report adverse events of which we become aware within the prescribed timeframe. We may also fail to recognize that we have become aware of a reportable adverse event, especially if it is not reported to us as an adverse event or if it is an adverse event that is unexpected or removed in time from the use or implant of the system. The FDA may also disagree with our determinations that an event was not reportable. If we fail to comply with our reporting obligations, the FDA could take action, including warning letters, untitled letters, administrative actions, criminal prosecution, imposition of civil monetary penalties, revocation of our marketing authorizations, seizure of our implant systems, or delay in obtaining marketing authorizations for our future products.
The FDA has the authority to require the recall of commercialized products based on a finding that there is reasonable probability that the device could cause serious, adverse health consequences or death. We may also choose to voluntarily recall a product if any material deficiency is found. A government-mandated or voluntary recall by us could occur as a result of an unacceptable risk to health, component failures, malfunctions, manufacturing defects, labeling or design deficiencies, packaging defects, or other deficiencies or failures to comply with applicable regulations. Product defects or other errors may occur in the future.
Depending on the corrective action we take to redress a product’s deficiencies or defects, the FDA may require, or we may decide, that we will need to obtain new marketing authorizations for the device before we may market or distribute the corrected device. Seeking such clearances or approvals may delay our ability to replace the recalled devices in a timely manner. Moreover, if we do not adequately address problems associated with our devices, we may face additional regulatory enforcement action, including FDA warning letters, product seizure, injunctions, administrative penalties, or civil or criminal fines.
Companies are required to maintain certain records of recalls and corrections, even if they are not reportable to the FDA. We have and may in the future initiate voluntary withdrawals or corrections for our products that we determine do not require notification of the FDA. If the FDA disagrees with our determinations, it could require us to report those
79
actions as recalls and we may be subject to enforcement action. A future recall announcement could harm our reputation with customers, potentially lead to product liability claims against us, and negatively affect our sales. Any corrective action, whether voluntary or involuntary, as well as defending ourselves in a lawsuit, will require the dedication of our time and capital, distract management from operating our business, and may harm our reputation, business, financial condition, results of operations, and prospects.
Disruptions at the FDA and other government agencies caused by funding shortages, staffing limitations, or global health concerns could hinder their ability to hire, retain or deploy key leadership and other personnel, and prevent new or modified products from being developed, reviewed, approved or commercialized in a timely manner or at all, which could negatively impact our business.
The ability of the FDA and foreign regulatory authorities to review and approve new products can be affected by a variety of factors, including government budget and funding levels, statutory, regulatory, and policy changes, the FDA’s or foreign regulatory authorities’ ability to hire and retain key personnel and accept the payment of user fees, and other events that may otherwise affect the FDA’s or foreign regulatory authorities’ ability to perform routine functions. Average review times at the FDA and foreign regulatory authorities have fluctuated in recent years as a result. In addition, government funding of other government agencies that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable. Disruptions at the FDA and other agencies may also slow the time necessary for new devices or modifications to authorized devices to be reviewed and/or authorized by necessary government agencies, which would adversely affect our business. For example, in recent years, the U.S. government has shut down several times and certain regulatory agencies, such as the FDA, have had to furlough critical employees and stop critical activities. In addition, the current U.S. Presidential administration has issued certain policies and Executive Orders directed towards reducing the employee headcount and costs associated with U.S. administrative agencies, including the FDA, and it remains unclear the degree to which these efforts may limit or otherwise adversely affect the FDA’s ability to conduct routine activities.
Separately, in response to the COVID-19 pandemic, the FDA postponed most inspections at domestic and foreign manufacturing facilities at various points. If a prolonged government shutdown occurs, or if renewed global health concerns, funding shortages or staffing limitations hinder or prevent the FDA or other regulatory authorities from conducting their regular inspections, reviews, or other regulatory activities, it could significantly impact the ability of the FDA or other such regulatory authorities to timely review and process our regulatory submissions, which could have a material adverse effect on our business.
We and the third parties with whom we work are subject to stringent and evolving U.S. and foreign laws, regulations, and rules, contractual obligations, industry standards, policies and other obligations related to data privacy and security. Our (or the third parties with whom we work) actual or perceived failures to comply with applicable data protection, privacy and security laws, regulations, standards and other requirements could adversely affect our business, results of operations and financial condition and lead to regulatory investigations or actions; litigation (including class action claims) and mass arbitration demands; fines and penalties; disruptions of our business operations; reputational harm; loss of revenue or profits; loss of customers or sales; and other adverse business consequences.
In the course of our operations, we may collect, use, store, disclose, transfer and otherwise process an increasing volume of personal information, including from our employees and third parties with whom we conduct business. We and our partners are or may become subject to federal, state and foreign data protection laws and regulations (i.e., laws and regulations that address data privacy and security). In the United States, numerous federal and state laws and regulations, including data breach notification laws, health information privacy laws and consumer protection laws and regulations (e.g., Section 5 of the FTC Act), that govern the collection, use, disclosure and protection of health-related and other personal information could apply to our operations or the operations of our partners. We may also be subject to U.S. federal rules, regulations and guidance concerning data security for medical devices, including guidance from the FDA.
For example, HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, imposes specific requirements relating to the privacy, security, and transmission of individually identifiable protected health information. Furthermore, the Federal Trade Commission (“FTC”) and many state attorneys general continue to enforce federal and state consumer protection laws against companies for online collection, use, dissemination and security practices that appear to be unfair or deceptive. The FTC expects a company’s data security measures to be reasonable and appropriate in light of the sensitivity and volume of consumer information it holds, the size and complexity of its business, and the cost of available tools to improve security and reduce vulnerabilities.
80
The regulatory framework for data privacy and security worldwide is continuously evolving and developing and, as a result, interpretation and implementation standards and enforcement practices are likely to remain uncertain for the foreseeable future. Certain states have also adopted comparable privacy and security laws and regulations that govern the privacy, processing and protection of health-related and other personal information. Such laws and regulations will be subject to interpretation by various courts and other governmental authorities, thus creating potentially complex compliance issues for us and our future customers and strategic partners. For example, California enacted the California Consumer Privacy Act, as amended by the California Privacy Rights Act (collectively, the “CCPA”), which requires covered businesses that process the personal information of California residents to, among other things, (i) provide certain disclosures to California residents regarding the business’s collection, use, and disclosure of their personal information; (ii) receive and respond to requests from California residents to access, delete, and correct their personal information, or to opt out of certain disclosures of their personal information; and (iii) enter into specific contractual provisions with service providers that process California resident personal information on the business’s behalf. Washington State enacted the “My Health My Data Act,” which broadly defines consumer health data, creates a private right of action to allow individuals to sue for violations of the law, imposes stringent consent requirements, and grants consumers certain rights with respect to their health data, including to request deletion of their information. Additional compliance investment and potential business process changes may be required. Similar laws have been passed in other states, and are continuing to be proposed at the state and federal level, reflecting a trend toward more stringent privacy legislation in the United States. The enactment of such laws could have potentially conflicting requirements that would make compliance challenging. In the event that we are subject to or affected by HIPAA, the CCPA or other domestic privacy and data protection laws, any liability from failure to comply with the requirements of these laws could adversely affect our financial condition.
As our operations and business grow, we may become subject to or affected by new or additional data protection laws and regulations and face increased scrutiny or attention from regulatory authorities. For example, in the EU, the European Union General Data Protection Regulation (“GDPR”) imposes strict requirements for processing the personal data of individuals within the European Economic Area (“EEA”). Companies that must comply with the GDPR face increased compliance obligations and risk, including more robust regulatory enforcement of data protection requirements and potential fines for noncompliance of up to €20,000 or 4% of the annual global revenues of the noncompliant company, whichever is greater.
Further, Europe and other jurisdictions have enacted laws requiring data to be localized or limiting the transfer of personal data to other countries. In particular, the EEA and the United Kingdom (“UK”) have significantly restricted the transfer of personal data to the United States and other countries whose privacy laws it generally believes are inadequate. Other jurisdictions may adopt or have already adopted similarly stringent data localization and cross-border data transfer laws.
Although there are currently various mechanisms that may be used to transfer personal data from the EEA and UK to the United States in compliance with law, such as the EEA standard contractual clauses, the UK’s International Data Transfer Agreement / Addendum, and the EU-U.S. Data Privacy Framework and the UK extension thereto (which allows for transfers to relevant U.S.-based organizations who self-certify compliance and participate in the EU-U.S. Data Privacy Framework), these mechanisms are subject to legal challenges, and there is no assurance that we can satisfy or rely on these measures to lawfully transfer personal data to the United States.
To the extent our business and operations expand to the EEA, the UK or other jurisdictions and there is no lawful manner for us to transfer personal data from such jurisdictions to the United States, or if the requirements for a legally-compliant transfer are too onerous, we could face significant adverse consequences, including the interruption or degradation of our operations, the need to relocate part of or all of our business or data processing activities to other jurisdictions (such as Europe) at significant expense, increased exposure to regulatory actions, substantial fines and penalties, the inability to transfer data and work with partners, vendors and other third parties, and injunctions against our processing or transferring of personal data necessary to operate our business. Additionally, companies that transfer personal data out of the EEA and the UK to other jurisdictions, particularly to the United States, are subject to increased scrutiny from regulators, individual litigants, and activist groups.
In addition to government regulation, privacy advocates and industry groups have and may in the future propose self-regulatory standards from time to time. These and other industry standards may legally or contractually apply to us, or we may elect to comply with such standards. We expect that there will continue to be new proposed laws and regulations concerning data privacy and security, and we cannot yet determine the impact such future laws, regulations and standards may have on our business. We make public statements about our use and disclosure of personal information through our privacy policies, information provided on our website and press statements. Although we endeavor to comply with our public statements and documentation, we may at times fail to do so or be alleged to have failed to do so. The publication of
81
our privacy policies and other statements that provide promises and assurances about data privacy and security can subject us to potential government or legal action if they are found to be deceptive, unfair or misrepresentative of our actual practices. Any concerns about our data privacy and security practices, even if unfounded, could damage the reputation of our business and harm our business, financial condition and results of operations.
Although we work to comply with applicable laws, regulations and standards, our contractual obligations and other legal obligations, these requirements are evolving and may be modified, interpreted and applied in an inconsistent manner from one jurisdiction to another, and may conflict with one another or other legal obligations with which we must comply. This evolution creates uncertainty in our business, affects our ability to operate in certain jurisdictions or to collect, store, transfer use and share personal information, necessitates the acceptance of more onerous obligations in our contracts, results in liability or imposes additional costs on us. The cost of compliance with these laws, regulations and standards is high and is likely to increase in the future. Any failure or perceived failure by us or our employees, representatives, contractors, consultants, collaborators or other third parties to comply with such requirements or adequately address privacy and security concerns, even if unfounded, could result in additional cost and liability to us, damage our reputation and adversely affect our business and results of operations. In addition, if our practices are not consistent, or viewed as not consistent, with legal and regulatory requirements, including changes in laws, regulations and standards or new interpretations or applications of existing laws, regulations and standards, we may also become subject to audits, inquiries, whistleblower complaints, adverse media coverage, investigations, criminal or civil sanctions, all of which may harm our business, financial condition and results of operations.
Risks Related to Ownership of Our Common Stock
We are an “emerging growth company” and a “smaller reporting company,” and we cannot be certain if the reduced reporting requirements applicable to emerging growth companies and smaller reporting companies will make our common stock less attractive to investors.
We are an “emerging growth company,” as defined in the JOBS Act. We will remain an “emerging growth company” until the earliest to occur of:
•the last day of the fiscal year during which our total annual revenue equals or exceeds $1.235 billion (subject to adjustment for inflation);
•the last day of the fiscal year following the fifth anniversary of our IPO;
•the date on which we have, during the previous three-year period, issued more than $1 billion in non-convertible debt; or
•the date on which we are deemed to be a “large accelerated filer” under the Exchange Act.
As a result of our “emerging growth company” status, we may take advantage of exemptions from various reporting requirements that would otherwise be applicable to public companies including, but not limited to, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved.
We also are a “smaller reporting company,” meaning that (i) the market value of our stock held by non-affiliates is less than $250,000 thousand or (ii) our annual revenue is less than $100,000 thousand during the most recently completed fiscal year and the market value of our stock held by non-affiliates is less than $700,000 thousand. If we are a smaller reporting company at the time we cease to be an emerging growth company, we may continue to rely on exemptions from certain disclosure requirements that are available to smaller reporting companies. Specifically, as a smaller reporting company we may choose to present only the two most recent fiscal years of audited financial statements in our annual report on Form 10-K and, similar to emerging growth companies, smaller reporting companies have reduced disclosure obligations regarding executive compensation.
Investors may find our common stock less attractive because we may rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and the market price of our common stock may be adversely affected and more volatile.
82
We continue to incur increased costs and are subject to additional regulations and requirements as a public company, which could lower our profits or make it more difficult to run our business.
As a public company, we continue to incur significant legal, accounting, and other expenses, including costs associated with public company reporting requirements. We have also incurred and will continue to incur costs associated with the Sarbanes-Oxley Act and related rules implemented by the SEC and the exchange on which our securities are listed. The expenses generally incurred by public companies for reporting and corporate governance purposes have been increasing. We expect these rules and regulations to increase our legal and financial compliance costs and to make some activities more time-consuming and costly, although we are currently unable to estimate these costs with any degree of certainty. These laws and regulations also could make it more difficult or costly for us to obtain certain types of insurance, including director and officer liability insurance, and we may be forced to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. These laws and regulations could also make it more difficult for us to attract and retain qualified persons to serve on our board of directors, on our board committees or as our executive officers. Furthermore, if we are unable to satisfy our obligations as a public company, we could be subject to delisting of our common stock, fines, sanctions, other regulatory action, and potentially civil litigation.
If we are unable to design, implement, and maintain effective internal control over financial reporting in the future, investors may lose confidence in the accuracy and completeness of our financial reports and the market price of our common stock may decline.
As a public company, we are required to maintain internal control over financial reporting and to report any material weaknesses in such internal controls. In addition, beginning with our second annual report on Form 10-K, we will be required to furnish a report by management on the effectiveness of our internal control over financial reporting, pursuant to the rules and regulations of the SEC regarding compliance with Section 404 of the Sarbanes-Oxley Act. The process of designing, implementing and testing the internal control over financial reporting required to comply with this obligation is time consuming, costly and complicated. While we have not identified material weaknesses in the past, we may identify material weaknesses in our internal control over financial reporting in the future. Any failure to maintain internal control over financial reporting could severely inhibit our ability to accurately report our financial condition, results of operations, or cash flows. Further, if we identify one or more material weaknesses in our internal control over financial reporting, if we are unable to comply with the requirements of Section 404 of the Sarbanes-Oxley Act in a timely manner, or if we or, if required, our auditors, are unable to assert that our internal control over financial reporting is effective, investors may lose confidence in the accuracy and completeness of our financial reports and the market price of our common stock could decline, and we could also become subject to investigations by the stock exchange on which our common stock is listed, the SEC or other regulatory authorities, which could require additional financial and management resources. Failure to remedy any material weakness in our internal control over financial reporting, or to implement or maintain other effective control systems required of public companies, could also restrict our future access to the capital markets.
We do not intend to pay dividends in the foreseeable future. As a result, your ability to achieve a return on your investment will depend on appreciation in the market price of our common stock.
We have never declared or paid cash dividends on our capital stock, and we do not currently intend to pay any cash dividends on our capital stock in the foreseeable future. We currently intend to retain all available funds and any future earnings to fund the development and expansion of our business. Any future determination related to dividend policy will be made at the discretion of our board of directors, subject to applicable laws, and will depend upon, among other factors, our results of operations, prospects, financial condition, contractual restrictions and capital requirements. In addition, our ability to pay cash dividends on our capital stock is limited by the terms of the Trinity Loan Agreement with Trinity Capital, and may be limited by the terms of any future debt or preferred securities we issue or any future credit facilities we enter into. Accordingly, investors must for the foreseeable future rely on sales of their common stock after price appreciation, which may never occur, as the only way to realize any future gains on their investments.
The market price of our common stock may be volatile, which could result in substantial losses for owners of our common stock.
The market price of our common stock may be highly volatile and could be subject to wide fluctuations. Some of the factors that may cause the market price of our common stock to fluctuate include:
•delays or setbacks in the ongoing commercialization of our products and procedures;
•the success of existing or new competitive products or technologies;
83
•regulatory or legal developments in the United States and other countries that we pursue expansion in;
•developments or disputes concerning patent applications, issued patents or other proprietary rights;
•the recruitment or departure of key personnel;
•the commencement of litigation;
•actual or anticipated changes in estimates as to financial condition and results of operations;
•announcement or expectation of additional financing efforts;
•announcements by us or our competitors of significant business developments, acquisitions, new offerings, licenses, strategic partnerships, joint ventures or capital commitments;
•the impact of pandemics epidemics, endemics and other public health emergencies on the performance of procedures;
•the impact of political instability, natural disasters, events of terrorism and or war, such as the ongoing conflict between Ukraine and Russia and in Israel, Gaza and surrounding areas, and the corresponding tensions created from such conflict between Russia, the United States and countries in Europe as well as other countries such as China, and broader conflicts in the Middle East;
•sales of our common stock by us, our insiders or other stockholders, or the anticipation of such sales;
•variations between our actual operating results, or those of companies that are perceived to be similar to us, and the expectations of securities analysts, investors and the financial community;
•any forward-looking financial or operating information we may provide to the public or securities analysts, any changes in this information or our failure to meet expectations based on this information;
•changes in estimates or recommendations by securities analysts, if any, that cover our stock;
•various macroeconomic events, including changes in inflation, interest rates and overall economic conditions and uncertainties;
•changes in the structure of health care payment systems;
•market conditions in the medical device sector;
•changes in the anticipated future size and growth rate of our market;
•the seasonality of our business;
•an increase in the rate of returns of our products;
•general economic, industry and market conditions, including economic recessions or slowdowns; and
•the other factors described in this “Risk Factors” section.
In recent years, the stock market in general, and the market for medical device companies in particular, has experienced significant price and volume fluctuations that have often been unrelated or disproportionate to changes in the operating performance of the companies whose stock is experiencing those price and volume fluctuations. Further, the stock market in general has been highly volatile due to various macroeconomic events. Broad market and industry factors may seriously affect the market price of our common stock, regardless of our actual operating performance. Following periods of such volatility in the market price of a company’s securities, securities class action litigation has often been brought against that company. Because of the potential volatility of our stock price, we may become the target of securities litigation in the future. Securities litigation could result in substantial costs and divert management’s attention and resources from our business.
84
If our operating and financial performance in any given period does not meet any guidance that we provide to the public, the market price of our common stock may decline.
We may, but are not obligated to, provide public guidance on our expected operating and financial results for future periods. Any such guidance will be comprised of forward-looking statements subject to the risks and uncertainties described in this Annual Report and in our other public filings and public statements. Our actual results may not always be in line with or exceed any guidance we have provided, especially in times of economic uncertainty. If actual circumstances differ from those in our assumptions, our operating and financial results could fall below our publicly announced guidance or the expectations of investors. If, in the future, our operating or financial results for a particular period do not meet any guidance we provide or the expectations of investment analysts or investors generally, or if we reduce our guidance for future periods, the market price of our common stock may decline. Even if we do issue public guidance, there can be no assurance that we will continue to do so in the future.
Our principal stockholders and management own a significant percentage of our stock and will be able to exert significant control over matters subject to stockholder approval.
Our directors, officers, pre-IPO holders of our outstanding stock and their respective affiliates beneficially own a significant number of our outstanding shares. As a result, these stockholders, if they act together, will be able to influence our management and affairs and all matters requiring stockholder approval, including the election of directors and approval of significant corporate transactions. This concentration of ownership may have the effect of delaying or preventing a change in control of our company and might affect the market price of our common stock.
Sales of a substantial number of shares of our common stock in the public market could cause our stock price to fall.
Sales of a substantial number of shares of our common stock in the public market could occur at any time. These sales, or the perception of these sales, could reduce the market price of our common stock.
As of December 31, 2025, we had 20,623,457 shares of common stock outstanding. Certain of our stockholders have rights, subject to some conditions, to require us to file registration statements covering their shares to include their shares in registration statements that we may file for ourselves or our stockholders, subject to market standoff and lockup agreements. We have registered and plan to continue to register shares of common stock that we have issued and may issue under our employee equity incentive plans. As we register such shares, they will be able to be sold freely in the public market upon issuance.
The market price of the shares of our common stock could decline as a result of the sale of a substantial number of our shares of common stock in the public market or the perception in the market that the holders of a large number of shares intend to sell their shares.
Provisions in our charter documents and under Delaware law could discourage a takeover that stockholders may consider favorable and may lead to entrenchment of management.
Our amended and restated certificate of incorporation and amended and restated bylaws contain provisions that could delay or prevent changes in control or changes in our management without the consent of our board of directors. These provisions include the following:
•a classified board of directors with three-year staggered terms, which may delay the ability of stockholders to change the membership of a majority of our board of directors;
•no cumulative voting in the election of directors, which limits the ability of minority stockholders to elect director candidates;
•the exclusive right of our board of directors, unless the board of directors grants such right to the stockholders, to elect a director to fill a vacancy created by the expansion of the board of directors or the resignation, death or removal of a director, which prevents stockholders from being able to fill vacancies on our board of directors;
•the required approval of at least 66-2/3% of the shares entitled to vote to remove a director for cause, and the prohibition on removal of directors without cause;
•the ability of our board of directors to authorize the issuance of shares of preferred stock and to determine the price and other terms of those shares, including preferences and voting rights, without stockholder approval, which could be used to significantly dilute the ownership of a hostile acquiror;
85
•the ability of our board of directors to alter our amended and restated bylaws without obtaining stockholder approval;
•the required approval of at least 66-2/3% of the shares entitled to vote to adopt, amend or repeal our amended and restated bylaws or repeal the provisions of our amended and restated certificate of incorporation regarding the election and removal of directors;
•a prohibition on stockholder action by written consent, which forces stockholder action to be taken at an annual or special meeting of our stockholders;
•an exclusive forum provision providing that the Court of Chancery of the State of Delaware is the exclusive forum for certain actions and proceedings;
•the requirement that a special meeting of stockholders may be called only by the board of directors, which may delay the ability of our stockholders to force consideration of a proposal or to take action, including the removal of directors; and
•advance notice procedures that stockholders must comply with in order to nominate candidates to our board of directors or to propose matters to be acted upon at a stockholders’ meeting, which may discourage or deter a potential acquiror from conducting a solicitation of proxies to elect the acquiror’s own slate of directors or otherwise attempting to obtain control of us.
We are also subject to the anti-takeover provisions contained in Section 203 of the General Corporation Law of the State of Delaware (the “Delaware General Corporation Law”). Under Section 203, a corporation may not, in general, engage in a business combination with any holder of 15% or more of its capital stock unless the holder has held the stock for three years or, among other exceptions, the board of directors has approved the transaction.
Claims for indemnification by our directors, officers, and other employees or agents may reduce our available funds to satisfy successful third-party claims against us and may reduce the amount of money available to us.
Our amended and restated certificate of incorporation and amended and restated bylaws provide that we will indemnify our directors and officers, in each case to the fullest extent permitted by Delaware law. In addition, as permitted by Section 145 of the Delaware General Corporation Law, our amended and restated bylaws and our indemnification agreements that we have entered into with our directors, officers and certain other employees provide that:
•We will indemnify our directors and officers for serving us in those capacities or for serving other business enterprises at our request, to the fullest extent permitted by Delaware law. Delaware law provides that a corporation may indemnify such person if such person acted in good faith and in a manner such person reasonably believed to be in or not opposed to the best interests of the registrant and, with respect to any criminal proceeding, had no reasonable cause to believe such person’s conduct was unlawful.
•We may, in our discretion, indemnify employees and agents in those circumstances where indemnification is permitted by applicable law.
•We are required to advance expenses, as incurred, to our directors and officers in connection with defending a proceeding, except that such directors or officers shall undertake to repay such advances if it is ultimately determined that such person is not entitled to indemnification.
•We will not be obligated pursuant to our amended and restated bylaws to indemnify a person with respect to proceedings initiated by that person against us or our other indemnitees, except with respect to proceedings authorized by our board of directors or brought to enforce a right to indemnification.
•The rights conferred in our amended and restated bylaws are not exclusive, and we are authorized to enter into indemnification agreements with our directors, officers, employees, and agents and to obtain insurance to indemnify such persons.
•We may not retroactively amend our amended and restated bylaws provisions to reduce our indemnification obligations to directors, officers, employees, and agents.
Our current amended and restated certificate of incorporation provides that the Court of Chancery of the State of Delaware is the exclusive forum for substantially all disputes between us and our stockholders and that the federal
86
district courts shall be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities Act, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers or employees or the underwriters or any offering giving rise to such claim.
Our amended and restated certificate of incorporation provides that the Court of Chancery of the State of Delaware is the exclusive forum for any derivative action or proceeding brought on our behalf, any action asserting a breach of fiduciary duty, any action asserting a claim against us arising pursuant to the Delaware General Corporation Law, our amended and restated certificate of incorporation or our amended and restated bylaws, or any action asserting a claim against us that is governed by the internal affairs doctrine; provided, that, this provision would not apply to suits brought to enforce a duty or liability created by the Exchange Act or any other claim for which the federal courts have exclusive jurisdiction. Furthermore, our amended and restated certificate of incorporation also provides that unless we consent in writing to the selection of an alternative forum, the federal district courts of the United States shall be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities Act. These choice of forum provisions may result in increased costs to stockholders to bring a claim, limit a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers or other employees, and may generally have the effect of discouraging lawsuits against us and our directors, officers and other employees. By agreeing to this provision, however, stockholders will not be deemed to have waived our compliance with the federal securities laws and the rules and regulations thereunder. Furthermore, the enforceability of similar choice of forum provisions in other companies’ certificates of incorporation has been challenged in legal proceedings, and it is possible that a court could find these types of provisions to be inapplicable or unenforceable. If a court were to find the choice of forum provisions in our amended and restated certificate of incorporation to be inapplicable or unenforceable in an action, we may incur additional costs associated with resolving such action in other jurisdictions, which could adversely affect our business and financial condition.
General Risk Factors
If we engage in acquisitions or strategic partnerships, it may increase our capital requirements, dilute our stockholders, cause us to incur debt or assume contingent liabilities, and subject us to other risks.
From time to time, we may evaluate various acquisitions and strategic partnerships, including licensing or acquiring complementary offerings, intellectual property rights, technologies, or businesses. Any acquisition or strategic partnership may entail numerous risks, including:
•increased operating expenses and cash requirements;
•the assumption of additional indebtedness or contingent liabilities;
•assimilation of operations, intellectual property, and products of an acquired company, including difficulties associated with integrating new personnel;
•the diversion of our management’s attention from our existing operations in pursuing such a strategic merger or acquisition;
•loss of key personnel and uncertainties in our ability to maintain key business relationships;
•uncertainties associated with the other party to such a transaction, including the prospects of that party and their existing products or future products and regulatory approvals; and
•our inability to generate revenue from acquired technology and/or products sufficient to meet our objectives in undertaking the acquisition or even to offset the associated acquisition and maintenance costs.
In addition, if we undertake acquisitions or strategic partnerships, we may issue dilutive securities, assume or incur debt obligations, incur large one-time expenses, and acquire intangible assets that could result in significant future amortization expense. Moreover, we may not be able to locate suitable acquisition or partnership opportunities, and even if we do locate such opportunities, we may not be able to successfully bid for or obtain them due to competitive factors or lack of sufficient resources. This inability could impair our ability to grow or obtain access to technology or products that may be important to the development of our business.
87
Unfavorable global and regional economic, political and health conditions could adversely affect our business, financial condition or results of operations.
Our results of operations could be adversely affected by global or regional economic, political and health conditions. A global financial crisis or global or regional political and economic instability (including changes in inflation, interest rates and overall economic conditions and uncertainties), tariffs, wars, terrorism, civil unrest, pandemics, epidemics, endemics and other public health emergencies, and other unexpected events, such as supply chain constraints or disruptions, could cause extreme volatility, increase our costs and disrupt our business. Business disruptions could include, among others, disruptions to our commercial activities, including due to supply chain or distribution constraints or challenges, clinical enrollment, clinical site availability, patient accessibility and conduct of our clinical trials, as well as temporary closures of our facilities and the facilities of suppliers or contract manufacturers in our supply chain. For example, these macroeconomic factors could affect the ability of our current or potential future manufacturers, sole source or single source suppliers, licensors or licensees to remain in business, or otherwise manufacture or supply components, materials or services relevant to our systems. Any failure by any of them to remain in business could affect the manufacture of our systems or our ability to meet demand for our systems. In addition, if inflation or other factors were to significantly increase our business costs, we may be unable to pass through price increases to our customers. Interest rates and the ability to access credit markets could also adversely affect the ability of our customers to purchase our systems.
The imposition of tariffs and other orders or restrictions impacting trade could adversely impact our business, including by increasing or otherwise impacting the costs and expenses we incur in connection with our operations and supply chain, and by potentially increasing the price of our systems to purchasers. For example, certain of our suppliers import raw materials, including certain metals, from Canada. The actual impacts of any tariffs and other orders or restrictions are subject to a number of factors including the effective date and duration of such tariffs, orders and restrictions, the amount, scope and nature of such inputs, any countermeasures that the target countries may take and any mitigating actions that may become available.
In addition, during certain crises and events, patients may prioritize other items over certain or all of their treatments or delay their requisite shoulder care, which could have a negative impact on our commercial sales.
A severe or prolonged economic downturn, political disruption or adverse health conditions could result in a variety of risks to our business, including our ability to raise capital when needed on acceptable terms, if at all. Any of the foregoing could harm our business and we cannot anticipate all of the ways in which the political or economic climate and financial market conditions could adversely impact our business.
We or the third parties we depend on may be adversely affected by natural disasters and other catastrophic events, and our business continuity and disaster recovery plans may not adequately protect us from a serious natural disaster or other catastrophic event. Any interruption in our operations or the operations of third parties who supply components or other materials for our products may have a material adverse effect on our business, financial condition, results of operations, and prospects.
Severe weather, natural disasters and other catastrophic events, including pandemics or other public health crises, earthquakes, tsunamis, hurricanes, floods, fires, explosions, accidents, power outages, cyberattacks, telecommunications failures, mechanical failures, unscheduled downtimes, civil unrest, strikes, transportation interruptions, unpermitted discharges or releases of toxic or hazardous substances, other environmental risks, wars or other conflicts (including wars in Russia and Ukraine as well as the Middle East), sabotage, terrorist attacks, or other intentional acts of vandalism or misconduct could severely disrupt our operations, or the operations of third parties who manufacture or supply components or other materials for our products, and have a material adverse effect on our business, financial condition, results of operations, and prospects.
If a natural disaster or other catastrophic event occurs that prevents us or third-party suppliers or manufacturers from using all or a significant portion of our or their headquarters or other facilities, that damages critical infrastructure or that otherwise disrupts operations, it may be difficult or, in certain cases, impossible, for us to continue our business for a substantial period of time. For example, we are highly dependent on our corporate headquarters in Grand Rapids, Michigan which has approximately 7,000 square feet of warehouse, distribution and office space as well as an approximately 3,000 square foot distribution facility in Modesto California that we leverage to distribute our implant systems in the Western United States. Any such natural disaster or catastrophic event that impacts either of these facilities could have a material adverse impact on our business, financial condition and results of operations. Moreover, the disaster recovery and business continuity plans we have in place currently are limited and are unlikely to prove adequate in the event of a serious disaster or similar catastrophic event. The potential impact of any disruption would depend on the nature and extent of the damage
88
caused by a disaster. We may incur substantial expenses as a result of the limited nature of our disaster recovery and business continuity plans, which, particularly when taken together with our lack of earthquake insurance, could have a material adverse effect on our business, financial condition, results of operations, and prospects.
In addition, integral parties in our supply chain are similarly vulnerable to natural disasters or other sudden, unforeseen, and severe adverse events. If such an event were to affect our supply chain, it could have a material adverse effect on our business, financial condition, results of operations, and prospects.
We are subject to risks from legal and arbitration proceedings that may prevent us from pursuing our business activities or require us to incur additional costs in defending against claims or paying damages.
We may become subject to legal disputes and regulatory proceedings in connection with our business activities involving, among other things, product liability, product defects, intellectual property infringement, employment matters, and/or alleged violations of other applicable laws in various jurisdictions. We may not be insured against all potential damages that may arise out of any claims to which we may be party in the ordinary course of our business. A negative outcome of these proceedings may prevent us from pursuing certain activities and/or require us to incur additional costs in order to do so and pay damages. In addition, securities class action litigation has often been instituted against companies following periods of volatility in the market price of a company’s securities. This type of litigation, if instituted, could result in substantial costs and a diversion of management’s attention and resources, which would harm our business, financial condition, results of operations and prospects. Additionally, the significant increase in the cost of directors’ and officers’ liability insurance may cause us to opt for lower overall policy limits or to forgo insurance that we may otherwise rely on to cover significant defense costs, settlements, and damages awarded to plaintiffs.
The outcome of pending or potential future legal and arbitration proceedings is difficult to predict with certainty. In the event of a negative outcome of any material legal or arbitration proceeding, whether based on a judgment or a settlement agreement, we could be obligated to make substantial payments, which could have a material adverse effect on our business, financial condition, results of operations, and prospects. In addition, the costs related to litigation and arbitration proceedings may be significant, and any legal or arbitration proceedings could have a material adverse effect on our business, financial condition, results of operations, and prospects.
The requirements of being a public company may divert our management’s attention from our growth strategies and other business concerns.
As a public company, we are subject to the reporting requirements of the Exchange Act and will be required to comply with the applicable requirements of the Sarbanes-Oxley Act and the Dodd- Frank Wall Street Reform and Consumer Protection Act of 2010, the listing requirements of the New York Stock Exchange and other applicable securities rules and regulations. Compliance with these rules and regulations increases our legal and financial compliance costs, makes some activities more difficult, time consuming or costly and increases demand on our systems and resources. Among other things, the Exchange Act requires that we file annual, quarterly and current reports with respect to our business and results of operations and maintain effective disclosure controls and procedures and internal controls over financial reporting. In order to maintain and, if required, improve our disclosure controls and procedures and internal controls over financial reporting to meet this standard, significant resources and management oversight may be required. As a result, management’s attention may be diverted from executing our growth strategies and managing other business concerns and, which could have a material adverse effect on our business, financial condition and results of operations. Although we intend to hire additional employees to comply with these requirements, we may need to hire even more employees in the future, which will increase our costs and expenses. Additionally, as a public company, it is more expensive for us to obtain director and officer liability insurance, and we may be required to accept reduced coverage or incur substantially higher costs to obtain coverage. These factors could make it more difficult for us to attract and retain qualified members of our board of directors, particularly to serve on our audit committee and compensation committee, and qualified executive officers.
We continue to incur increased costs and are subject to additional regulations and requirements as a public company, which could lower our profits or make it more difficult to run our business.
As a public company, we continue to incur significant legal, accounting, and other expenses, including costs associated with public company reporting requirements. We have also incurred and will continue to incur costs associated with the Sarbanes-Oxley Act and related rules implemented by the SEC and the NYSE. The expenses generally incurred by public companies for reporting and corporate governance purposes have been increasing. We expect these rules and regulations to increase our legal and financial compliance costs and to make some activities more time-consuming and costly, although we are currently unable to estimate these costs with any degree of certainty. These laws and regulations also could make it
89
more difficult or costly for us to obtain certain types of insurance, including director and officer liability insurance, and we may be forced to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. These laws and regulations could also make it more difficult for us to attract and retain qualified persons to serve on our board of directors, on our board committees or as our executive officers. Furthermore, if we are unable to satisfy our obligations as a public company, we could be subject to delisting of our common stock, fines, sanctions, other regulatory action, and potentially civil litigation.
If securities or industry analysts issue an adverse or misleading opinion regarding our stock, our stock price and trading volume could decline.
The trading market for our common stock is influenced by the research and reports that industry or securities analysts publish about us or our business. If any of the analysts who cover us issue an adverse or misleading opinion regarding us, our business model, our intellectual property or our stock performance, or if our results of operations fail to meet the expectations of analysts, our stock price would likely decline. If one or more of these analysts cease coverage of us or fail to publish reports on us regularly, we could lose visibility in the financial markets, which in turn could cause our stock price or trading volume to decline.
Our disclosure controls and procedures may not prevent or detect all errors or acts of fraud.
We are subject to the periodic reporting requirements of the Exchange Act. We must design our disclosure controls and procedures to reasonably assure that information we must disclose in reports we file or submit under the Exchange Act is accumulated and communicated to management, and recorded, processed, summarized, and reported within the time periods specified in the rules and forms of the SEC. We believe that any disclosure controls and procedures or internal controls and procedures, no matter how well-conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met.
These inherent limitations include the realities that judgments in decision-making can be faulty, and that breakdowns can occur because of simple error or mistake. For example, our directors or executive officers could inadvertently fail to disclose a new relationship or arrangement, causing us to fail to make a required related party transaction disclosure. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people, or by an unauthorized override of the controls. Accordingly, because of the inherent limitations in our control system, misstatements due to error or fraud may occur and not be detected.
Our insurance may not cover all potential losses or liabilities that may arise.
We are not insured against all potential losses or liabilities that may arise, as insurance coverage may be unavailable, not cost-effective, or subject to significant limitations. For example, we are not insured against business interruptions suffered by third parties that we depend on, environmental liabilities or patent infringement, among other types of risks. Furthermore, no assurance can be given that an insurance carrier will not seek to cancel or deny coverage after a claim has occurred. If a loss or liability occurs that is not or not fully covered by insurance, we may be required to pay substantial amounts, which could adversely affect its cash position and results of operations.
We are subject to U.S. anti-bribery, anti-corruption, and anti-money laundering laws, including the FCPA, as well as export control, customs laws, economic sanctions and other trade laws and regulations (collectively, the “Trade Laws”). We can face serious consequences for violations.
As we grow our international presence and global operations, we will be increasingly exposed to trade and economic sanctions and other restrictions imposed by the governments of the United States and other applicable jurisdictions. Compliance with applicable regulatory requirements regarding the export of our products may create delays in the introduction of our products in international markets or, in some cases, prevent the export of our products to some countries altogether. Furthermore, U.S. export control laws and economic sanctions prohibit the provision of certain products and services to countries, governments, and persons targeted by U.S. sanctions.
We are also subject to anti-corruption laws, including the FCPA, the U.S. domestic bribery statute contained in 18 U.S.C. 201, the U.S. Travel Act, and other state and national anti-bribery laws in the countries in which we may conduct activities in the future. Anti-corruption laws are interpreted broadly and generally prohibit companies and their employees, agents and intermediaries from authorizing, promising, offering or providing, directly or indirectly, corrupt or improper payments or anything else of value to recipients in the public or private sector. The FCPA also requires public companies to make and keep books and records that accurately and fairly reflect the transactions of the corporation and to devise and
90
maintain an adequate system of internal accounting controls. We can be held liable for the corrupt or illegal activities of our agents and intermediaries, even if we do not explicitly authorize or have actual knowledge of such activities.
Violations of Trade Laws can result in substantial criminal fines and civil penalties, imprisonment, the loss of trade privileges, debarment, tax reassessments, breach of contract and fraud litigation, reputational harm and other consequences. Likewise, any investigation of potential violations of Trade Laws could also have an adverse impact on our reputation, our business, results of operations and financial condition.
We cannot assure you that our policies and procedures are or will be sufficient or that directors, officers, employees, representatives, consultants and agents have not engaged and will not engage in conduct for which we may be held responsible, nor can we assure you that our business partners have not engaged and will not engage in conduct that could materially affect their ability to perform their contractual obligations to us or even result in our being held liable for such conduct. Violations of the FCPA, Trade Laws, or other export control, anti-corruption, anti-money laundering and anti-terrorism laws or regulations may result in severe criminal or civil sanctions, and we may be subject to other liabilities, which could negatively affect our business, financial condition and results of operations.
We, along with our suppliers, are dependent on various information technology systems. If our information technology systems or those of third parties with whom we work or our data, are or were compromised, we could experience adverse consequences resulting from such compromise, including but not limited to regulatory investigations or actions; litigation; fines and penalties; disruptions of our business operations; reputational harm; loss of revenue or profits; loss of customers or sales; and other adverse consequences.
We and our suppliers collect and maintain information in digital form that is necessary to conduct our business, and rely extensively on information technology systems, networks and services, including internet sites, data hosting and processing facilities and tools, physical security system and other hardware, software and technical applications and platforms, some of which are managed, hosted, provided or used by third-parties or their vendors, and some of which may be stored outside of the United States, to the extent we expand our operations internationally. These systems include, but are not limited to, ordering and managing materials from suppliers, converting materials to finished products (suppliers), shipping products to customers, processing transactions, summarizing and reporting results of operations, complying with regulatory, legal or tax requirements, providing data security and other processes necessary to manage our business.
A significant breakdown, invasion, corruption, destruction or interruption of critical information technology systems or infrastructure, by our workforce, others with authorized access to our systems or unauthorized persons could negatively impact operations. The use of cloud-based computing creates opportunities for the unintentional dissemination or intentional destruction of confidential information stored in our or our third-party providers’ systems, portable media or storage devices. Our internal computer systems and those of our contractors, consultants and collaborators have been and are vulnerable to damage from cyberattacks, “phishing” attacks, intentional or accidental actions or omissions to act that cause vulnerabilities, computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures.
Attacks upon information technology systems are increasing in their frequency, levels of persistence, sophistication and intensity, and are being conducted by sophisticated and organized groups and individuals with a wide range of motives and expertise. Cyber-attacks, malicious internet-based activity, online and offline fraud, and other similar activities threaten the confidentiality, integrity, and availability of our sensitive information and information technology systems, and those of the third parties with whom we work. Our information technology systems and those of our third-party service providers, strategic partners and other contractors or consultants are vulnerable to attack, damage and interruption from computer viruses and malware (e.g. ransomware), misconfigurations, “bugs” or other vulnerabilities, malicious code, natural disasters, terrorism, war, telecommunication and electrical failures, hacking, cyberattacks, phishing attacks and other social engineering schemes, employee theft or misuse, human error, fraud, denial or degradation of service attacks and sophisticated nation-state and nation-state-supported actors.
We have also outsourced elements of our information technology infrastructure, and as a result a number of third-party vendors may or could have access to our confidential information. If we or our third-party vendors were to experience a significant cybersecurity breach of our or their information systems or data, the costs associated with the investigation, remediation and potential notification of the breach to counterparties and data subjects could be material. In addition, our remediation efforts may not be successful. If we do not allocate and effectively manage the resources necessary to build and sustain the proper technology and cybersecurity infrastructure, we could suffer significant business disruption, including transaction errors, supply chain or manufacturing interruptions, processing inefficiencies, data loss or the loss of or damage to intellectual property or other proprietary information. There can also be no assurance that our and our third-
91
party service providers’, strategic partners’, contractors’, consultants’, Contract Research Organizations’ and collaborators’ cybersecurity risk management program and processes, including policies, controls or procedures, will be fully implemented, complied with or effective in protecting our systems, networks and confidential information. For example, a third-party software provider which we have engaged as part of our advanced implant systems was recently inspected by the FDA, which identified a single observation regarding inadequate validation of the software to address active vulnerability threats. While such a finding may not be material, if not properly remediated, it could affect our brand, business, financial condition, results of operations and prospects.
We and certain of our service providers are from time to time subject to cyberattacks and security incidents. While we do not believe that we have experienced any significant system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our development programs and our business operations, whether due to a loss, corruption or unauthorized disclosure of our trade secrets, personal information or other proprietary or sensitive information or other similar disruptions. We may also experience security breaches that may remain undetected for an extended period. If our systems are damaged or cease to function properly due to any number of causes, ranging from catastrophic events to power outages to security breaches, and our business continuity plans do not effectively compensate timely, we may suffer interruptions in our ability to manage operations, and would also be exposed to a risk of loss, including financial assets or litigation and potential liability, which could materially adversely affect our business, financial condition, results of operations and prospects.
We cannot assure you that any limitations of liability provisions in our contracts would be enforceable or adequate or would otherwise protect us from any liabilities or damages with respect to any particular claim relating to a security lapse or breach. While we maintain certain insurance coverage, including cyber insurance, our insurance may be insufficient or may not cover all liabilities incurred by such attacks. We also cannot be certain that our insurance coverage will be adequate for data handling or data security liabilities actually incurred, that insurance will continue to be available to us on economically reasonable terms, or at all, or that any insurer will not deny coverage as to any future claim. The successful assertion of one or more large claims against us that exceeds available insurance coverage, or the occurrence of changes in our insurance policies, including premium increases or the imposition of large deductible or co-insurance requirements, could have a material adverse effect on our business, including our financial condition, operating results and reputation.
Changes in tax laws or regulations or in their implementation or interpretation that are applied adversely to us or our customers may seriously harm our business.
We are or may become subject to income and non-income taxes in the United States under federal, state and local jurisdictions and in certain foreign jurisdictions in which we may operate in the future. New income, sales, use or other tax laws, statutes, rules, regulations or ordinances could be enacted at any time, which could affect the tax treatment of any of our future domestic and foreign earnings. Any new taxes could adversely affect our domestic and international business operations, and our business and financial performance. Further, existing tax laws, statutes, rules, regulations or ordinances could be interpreted, changed, modified or applied adversely to us, possibly on a retroactive basis.
Our ability to use our net operating loss carryforwards and certain other tax attributes may be subject to limitation.
As of December 31, 2025, we had U.S. federal net operating loss carryforwards of approximately $[ ] thousand, which may be available to offset future taxable income for U.S. income tax purposes. Under the Internal Revenue Code of 1986, as amended (the “Code”), our federal net operating losses generated in taxable years beginning after December 31, 2017 will not expire and may be carried forward indefinitely, but the deductibility of such federal net operating loss carryforwards in a taxable year is limited to 80% of current year taxable income (with certain adjustments). Our ability to utilize our federal net operating loss carryforwards and certain other tax attributes may be further limited under Section 382 and 383 of the Code. This limitation applies if we experience an “ownership change,” which is generally defined as a greater than 50 percentage point change (by value) in the ownership of our equity by certain stockholders over a rolling three-year period. The amount of the annual limitation is generally equal to the product of the applicable long-term tax exempt-rate (as published by the U.S. Internal Revenue Service for the month in which the “ownership change” occurred) and the value of our outstanding stock immediately prior to the “ownership change.” If we have a net unrealized built-in gain in our assets immediately prior to the “ownership change,” the annual limitation may be increased as certain gains are, or are treated as, recognized during the five-year period beginning on the date of the “ownership change.”
Similar provisions of state tax law may also apply to limit the use of any state net operating loss carryforwards. For example, California imposed limits on the usability of California state net operating losses to offset taxable income in tax years beginning on or after January 1, 2024 and before January 1, 2027. We have undergone and may in the future undergo an “ownership change” due to transactions in our stock, which may be outside of our control. As a result, if we earn net taxable income, our ability to use our net operating loss carryforwards and other tax attributes to offset taxable income may be subject to limitations, which could potentially result in increased future tax liability to us. We cannot predict whether
92
any future “ownership change” would result in a significant limitation on our ability to use our net operating loss carryforwards and other tax attributes to offset our taxable income and adversely affect our future cash flows. If not utilized, a portion of our net operating loss carryforwards may expire.
The increasing use of social media platforms presents new risks and challenges.
Social media is increasingly being used to communicate about our systems and technologies, and we intend to utilize appropriate social media in connection with the commercialization of our advanced implant systems. Social media practices in the medical device industry continue to evolve and regulations and regulatory guidance relating to such use are evolving and not always clear. This evolution creates uncertainty and risk of noncompliance with regulations applicable to our business, resulting in potential regulatory actions against us, along with the potential for litigation. In addition, there is a risk of inappropriate disclosure of sensitive information or negative or inaccurate posts or comments about us or our systems on any social networking website. If any of these events were to occur or we otherwise fail to comply with any applicable regulations, we could incur liability, face restrictive regulatory actions, or incur other harm to our business such as reputational damage.
We could be subject to securities class action litigation.
In the past, securities class action litigation has often been brought against a company following a decline in the market price of its securities. This risk is especially relevant for us because biotechnology and biopharmaceutical companies have experienced significant stock price volatility in recent years. If we face such litigation, even if ultimately decided in our favor, it could result in substantial costs and a diversion of our management’s attention and resources, which could harm our business.
Item 1B. Unresolved Staff Comments
Not applicable.
Item 1C. Cybersecurity.
Our cybersecurity management program includes the following elements:
Policies and Procedures: The Company maintains policies that detail acceptable use of IT related assets, safeguarding physical and digital access and how the organization would respond to a data breach or related incident. Processes are documented to formalize the implementation of the related policies.
Continuous Monitoring: Use of automated tools and third-party services for real-time threat detection, vulnerability scanning and incident alerting. The Company has established additional detection and protection systems including email and web filtering, dark web monitoring and firewalls.
Security Incident Response Plan: A formal plan that includes containment, eradication, recovery, root cause analysis and communication protocols.
93
Training and Awareness: All of the Company’s team members, including senior management, receive mandatory quarterly cybersecurity training and periodic phishing simulations. Individuals who are determined to have a higher associated risk are provided additional training. Further, upon beginning employment and then annually employees must acknowledge policies related to technology in our employee handbook.
Periodic Risk Assessment: Company policies, including those related to cybersecurity and data breach response, are assessed periodically for emerging risks and subject to continuous improvement initiatives.
In the event of a cybersecurity incident the incident response team, led by our CEO and comprised of a cross-functional team including representatives from IT, Finance, Quality, Legal, and Human Resources , and further augmented by additional individuals as deemed necessary, follows the Company’s Security Incident Response Plan. Pursuant to this process, designated personnel are responsible for assessing the severity of the incident, types of data involved, and any associated threats. The incident response team is responsible for identifying any exposure containing protected or sensitive data, containing potential threats by removing access or quarantining effected devices and systems and performing analyses to understand root cause with the goal of minimizing impact to the Company’s stakeholders. As appropriate, a communication plan to satisfy internal and other reporting obligations will be drafted and executed. Post-incident analysis and program enhancements would be implemented as appropriate.
We describe risks faced by us from identified cybersecurity threats in Item 1A, “Risk Factors—Risks Related to Our Business—We, along with our suppliers, are dependent on various information technology systems. If our information technology systems or those of third parties with whom we work or our data, are or were compromised, we could experience adverse consequences resulting from such compromise, including but not limited to regulatory investigations or actions; litigation; fines and penalties; disruptions of our business operations; reputational harm; loss of revenue or profits; loss of customers or sales; and other adverse consequences.”
Item 2. Properties
Our corporate headquarters is located in Grand Rapids, Michigan where we lease a 7,000 square foot facility pursuant to a lease agreement that expires in June 2026. This facility includes warehouse, distribution and office space. We also lease a third-party approximately 3,000 square foot distribution facility in Modesto, California that distributes our implant systems in the western United States. We believe that this facility is sufficient to meet our current and anticipated needs in the near term and that suitable additional space is available as needed to accommodate expansion of our operations and distribution activities.
Item 3. Legal Proceedings
From time to time, we may be party to legal proceedings that arise in the ordinary course of our business, some of which may be covered by insurance. Except for the description of legal proceedings disclosed in Note 13 to our audited financial statements, which is incorporated herein by reference, management believes that we do not have any pending legal proceedings that, separately or in the aggregate, would have a material adverse effect on our results of operations, financial condition or cash flows. No material legal proceedings were terminated, settled or otherwise resolved during the year ended December 31, 2025.
Item 4. Mine Safety Disclosures
Not applicable.
94
Part II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market Information
On July 31, 2025 our common shares began trading on the New York Stock Exchange under the symbol “SI.” Prior to that time, there was no public market for our common shares.
Holders of Record
As of March 3, 2026, there were 293 registered holders of record of our common shares. The actual number of holders is greater than this number and includes shareholders who are beneficial owners but whose shares are held in “street name” by banks, brokers, and other financial institutions. This number of record holders also does not include shareholders whose shares may be held in trust by other entities.
Dividend Policy
We have never declared or paid cash dividends on our capital stock. We do not expect to pay dividends on our common share for the foreseeable future. Instead, we anticipate that all of our earnings, if any, will be used for the operation and growth of our business. Any future determination to declare cash dividends would be subject to the discretion of our Board of Directors and would depend upon various factors, including our results of operations, financial condition and capital requirements, restrictions that may be imposed by applicable law and our contracts and other factors deemed relevant by our Board of Directors. We are not obligated to pay dividends on our common shares.
Use of Proceeds
On August 1, 2025, we completed our Initial Public Offering (“IPO”) in which we issued and sold 5,000,000 shares of common stock at a public offering price of $15.00 per share. Morgan Stanley & Co. LLC, Goldman Sachs & Co. LLC and Piper Sandler & Co. acted as lead underwriters for the IPO. We raised net proceeds of $64,212 thousand after deducting underwriter discounts and commissions and fees and expenses payable by us. No payments for such expenses were made directly or indirectly to (i) any of our officers or directors or their associates, (ii) any persons owning 10% or more of any class of our equity securities or (iii) any of our affiliates.
All shares issued and sold in the IPO were registered under the Securities Act pursuant to the Registration Statement on Form S-1 (File No. 333-288549), as amended, declared effective by the SEC on July 30, 2025 (the “Registration Statement”).
There has been no material change in the expected use of the net proceeds from our IPO as described in our Registration Statement. Certain of the net proceeds from our IPO have been invested primarily in savings and money market accounts.
Recent Sales of Unregistered Securities
None.
Item 6. [Reserved]
95
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with other sections of this Annual Report, including “Item 1. Business,” “Item 1A. Risk Factors” and our audited consolidated financial statements and related notes included elsewhere in this Annual Report.
Overview
We are a commercial-stage medical technology company exclusively focused on transforming the shoulder surgical care market. We currently offer advanced implant systems for shoulder arthroplasty. These systems are a core element of our ecosystem, which we designed to improve core components of shoulder surgical care – preoperative planning, implant design and procedural efficiency – to benefit each stakeholder in the care chain. Our ecosystem is also comprised of enabling technologies, efficient instrument systems, specialized support and surgeon-to-surgeon collaboration. Together, these elements seek to address the long-standing clinical and operational challenges in the shoulder surgical care market by delivering predictable outcomes, procedural simplicity, and efficiency across all sites of care. We believe our exclusive focus on shoulder surgical care, combined with a highly specialized commercial organization and strong clinical data, positions us well to capture significant share in this large, growing market.
We believe the shoulder surgical care market today presents a significant market opportunity. Our initial focus within this broader market is on shoulder arthroplasty. Shoulder arthroplasty is an established surgical procedure involving the reconstruction of the shoulder joint with prosthetic implants through one of two main approaches: aTSA and rTSA. Both approaches can be performed in inpatient hospital settings and in outpatient settings, including ASCs. A key competitive advantage of ours has been the emergence of ASCs as a cost-efficient site of care with positive outcomes relative to hospital-based care. We expect that future growth in the shoulder surgical care market will be significantly driven by ASCs as hospitals face capacity constraints and are more limited in their ability to meet increasing demand.
We view ourselves as specialists serving specialists, having purposefully built our product ecosystem around the unique needs of shoulder surgeons. Our commercial organization is comprised of three key components: (i) a dedicated commercial leadership team, (ii) a CEME team and (iii) a network of independent distributors. These key components work in tandem to form a flywheel that is designed to build and provide key product support to surgeons and other stakeholders in the shoulder surgical care market, accelerate adoption, and enhance long-term retention. Our commercial organization is strategically focused on surgeons in hospital and ASC settings, with a particular focus on the high-volume surgeons who perform the vast majority of shoulder arthroplasty procedures each year.
We utilize third-party manufacturing and supply providers to manufacture our implants. We believe this outsourcing strategy provides the expertise and capacity required to effectively and efficiently scale production based on demand, and helps to ensure low-cost production and a capital efficient business model.
We have experienced significant growth in recent years, primarily driven by growth in our net revenue from the sale of our advanced implant systems sold.
Key Business Metrics
We regularly review a number of operating and financial metrics to evaluate our business, measure our performance, identify trends affecting our business, formulate our business plan and make strategic decisions. We believe that the number of implant systems sold is a useful indicator of our ability to drive demand for our implant systems, generate net revenue and expand our business. The following table sets forth the number of implant systems sold in each of the three-month periods indicated:
| Three Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||
| Mar. 31, 2024 | June 30, 2024 | Sept. 30, 2024 | Dec. 31, 2024 | Mar. 31, 2025 | June 30, 2025 | Sept. 30, 2025 | Dec. 31, 2025 | ||||||||||||||||||||||||||||||||||||||||
| Implant systems sold | 971 | 1,121 | 1,037 | 1,220 | 1,443 | 1,503 | 1,584 | 1,976 | |||||||||||||||||||||||||||||||||||||||
While we believe that the number of implant systems sold is a useful indicator and is helpful in tracking the progress of our current business, we anticipate this metric may be substituted for additional or different metrics as our business continues to grow and scale.
96
Key Factors Affecting Our Results of Operations
We believe the following important factors have impacted and will continue to impact our results of operations for the foreseeable future. While these factors may present significant opportunities for us, they also pose risks and challenges that we must address, as well as those described in the section titled “Risk Factors.”
•Market awareness and adoption. The growth of our business depends on our ability to generate broader awareness of our ecosystem in an effort to drive adoption by new surgeons and to increase utilization by existing surgeons. To drive adoption, our commercial organization is strategically focused on surgeons in hospital and ASC settings, and leverages our internal business intelligence platform to appropriately target surgeons in the shoulder surgical care market. The organization uses key touchpoints, surgeon support and surgeon education initiatives to deliver high quality services and information to surgeons. We are also focused on supporting surgeons that already use our implant systems in order to further increase utilization. We intend to continue scaling our commercial organization to further drive awareness, adoption and demand. Over time, we expect to further expand and utilize our external network of independent distributors. In the future, we may increase our international presence and any such expansion may adversely affect our gross margin and results of operations. Our financial performance will be significantly impacted by the extent to which we can increase awareness of our ecosystem, as well as the timing and rate of adoption of our implant systems by key stakeholders in the shoulder surgical care market.
•Increasing importance of outpatient and ASC settings. While our ecosystem provides advantages across all shoulder surgical care settings, we believe we are particularly well positioned to address the increasing importance of outpatient and ASC settings. The number of procedures performed in outpatient and ASC settings has increased over time due to both the transition of such procedures from the hospital setting, as well as from a general increase in the number of total procedures performed, due in part to the access and availability of these settings. We generally derive a similar amount of net revenue from procedures whether they are performed in a hospital or an ASC. As a result, we believe outpatient and ASC settings represent an important and growing opportunity to drive demand and net revenue.
•Continued investments in product development, innovation and growth. We expect to continue to focus on long-term revenue growth through investments in our ecosystem and expansion of our operations. In research and development, we continually invest in improving our technologies, developing new products and further expanding our cleared indications. For example, we began using ProVoyance in 2021 and have developed a fracture-specific system, a revision solution and implants tailored for patients with metal-hypersensitivity. We also believe our ecosystem can be further expanded to address adjacent markets, such as sports medicine and trauma. We anticipate we will continue to invest significantly in product development, including with respect to our supporting technologies, in order to further bolster our ecosystem. While research and development are time consuming and costly and therefore negatively impact our results of operations in the near term, we believe expanding into new areas, implementing product improvements and continuing to demonstrate the efficacy, safety and cost effectiveness of our products through clinical data and surgeon education are all critical to increasing the adoption of our implant systems and to the success of our business over the long term. As we expand our operations in line with our anticipated growth, we will be required to maintain sufficient levels of inventory and instrumentation to meet our estimated demand, which we expect will increase expenses.
•Reimbursement and coverage. Healthcare providers generally rely on third-party payors, including federal Medicare, state Medicaid and private health insurance plans, to cover and reimburse all or part of the cost of our implant systems. As a result, demand for our implant systems depends in large part on the availability of reimbursement from such payors and the rates that such payors reimburse for procedures using our implant systems, which can vary due to geographic location, nature of facility in which the procedure is performed and other factors. While we benefit from established reimbursement practice and codes applicable to partial and total shoulder arthroplasty, we also work with payors to ensure positive coverage decisions and payment rates in
97
outpatient settings. Effective as of January 1, 2024, Centers for Medicare and Medicaid Services (“CMS”) added total shoulder arthroplasty to the ASC covered procedures list, which allows procedures that use our implant systems to be performed at ASCs and be reimbursed by Medicare. We believe this decision helped to improve demand from ASCs and supported improved payment rates in outpatient settings for the year ended December 31, 2025, which had a positive impact on net revenue during the period. We expect this trend to continue and further support our growth in outpatient settings, such as ASCs.
•Seasonality. We have experienced and expect to continue to experience seasonality in our business. While we have experienced significant growth across quarters, we expect that future demand for our advanced implant systems will typically be lower in the months in and surrounding the third calendar quarter, as is common across our industry, as a result of summer seasonality associated with warmer weather and its corresponding impact on individual lifestyles.
Non-GAAP Financial Measures
In addition to our results and measures of performance determined in accordance with U.S. GAAP, we believe that non-GAAP financial measures can be useful in evaluating and comparing our financial and operational performance over multiple periods, identifying trends affecting our business, formulating business plans and making strategic decisions. We use and present Adjusted EBITDA for these purposes. We define Adjusted EBITDA as net loss before interest expense, net, income tax expense, depreciation and amortization, and stock-based compensation expense.
We believe that Adjusted EBITDA, together with a reconciliation to net loss, provides meaningful supplemental information regarding our performance by excluding certain items that may not be indicative of our business, results of operations, or outlook. However, Adjusted EBITDA has limitations as an analytical tool, and you should not consider this measure in isolation or as a substitute for analysis of our financial results as reported under U.S. GAAP. Some of these potential limitations include:
•other companies, including companies in our industry which have similar business arrangements, may report Adjusted EBITDA, or similarly titled measures but calculate them differently, which reduces their usefulness as comparative measures;
•although depreciation and amortization expenses are non-cash charges, the assets being depreciated and amortized may have to be replaced in the future, and Adjusted EBITDA does not reflect cash capital expenditures for such replacements or for new capital expenditure requirements;
•Adjusted EBITDA also does not reflect changes in, or cash requirements for, our working capital needs or the potentially dilutive impact of stock-based compensation; and
•Adjusted EBITDA does not reflect the interest expense, or the cash requirements necessary to service interest or principal payments, on existing or future debt that we may incur.
Because of these and other limitations, you should consider Adjusted EBITDA only as supplemental to other GAAP-based financial measures.
The following table presents a reconciliation of Adjusted EBITDA to the most comparable GAAP financial measure, net loss, for each of the periods indicated:
98
| Year Ended December 31, | |||||||||||
| 2025 | 2024 | ||||||||||
| Net loss | $ | (40,359) | $ | (15,619) | |||||||
| Interest expense, net | 70 | 1,316 | |||||||||
| Income tax expense | — | — | |||||||||
| Depreciation and amortization expense | 3,208 | 2,196 | |||||||||
| Stock-based compensation expense | 996 | 754 | |||||||||
| Adjusted EBITDA | $ | (36,085) | $ | (11,353) | |||||||
Components of Our Results of Operations
Net Revenue
We currently derive our net revenue from the sale of our Anatomic Total Shoulder Arthroplasty (“aTSA”) and Reverse Total Shoulder Arthroplasty (“rTSA”) implant systems, which generally consist of our InSet Glenoid and humeral stem products. We sell our implants to hospitals, outpatient centers and ASCs in the United States through a dedicated commercial leadership team and a network of external independent distributors. Net revenue is recognized when the performance obligation to deliver these implant systems to our customers is satisfied and we transfer control of the implants to our customers, which is generally when we have received a purchase order and appropriate notification that the procedure has been used or implanted. Revenue is recognized in the amount of the consideration received net of any sales taxes that we expect to collect from customers. We also record shipping and handling costs as revenue. Our average sales price for our implant systems was $7,273 and $7,271 for the years ended December 31, 2025 and 2024, respectively. No single customer accounted for more than 10% of our net revenue during the years ended December 31, 2025 and 2024. We expect our net revenue to increase for the foreseeable future as we expand our commercial organization, add new customers, expand our sales territories, introduce new products, as existing customers perform more procedures using our systems and as we generally expand awareness of our systems with new and existing customers. While industry trends have resulted in increased downward pricing pressure on medical services and products, we have not experienced a material impact on our net revenue to date; however, we cannot assure you that our net revenue will not be impacted in the future by these industry trends. Our net revenue may fluctuate from quarter to quarter due to a variety of factors, such as the size and success of our dedicated commercial leadership team, the number of hospitals and physicians who are aware of and use our systems and seasonality.
Cost of Goods Sold, Gross Profit and Gross Margin
Cost of Goods Sold
Cost of goods sold consists primarily of the cost of components, packaging and sterilization, and obsolete inventory adjustments. Our systems are manufactured to our specifications primarily by third-party suppliers in the United States and are generally ordered on a purchase order basis. Cost of goods sold is recognized at the time the related revenue is recognized. Prior to use in surgery, the cost of our products is recorded as inventories, net of obsolescence reserve on our Balance Sheets. Cost of goods sold does not include depreciation expense for instruments, which is included in selling, general and administrative expenses. Depreciation expense for instruments was $2,908 thousand and $1,896 thousand for the years ended December 31, 2025 and 2024, respectively. See Note 4 to our audited financial statements included elsewhere in this Annual Report for additional information. We expect cost of goods sold to increase as our net revenue increases and more of our implant systems are sold.
Gross Profit and Gross Margin
Gross profit is calculated as net revenue less cost of goods sold. We calculate gross margin as gross profit divided by net revenue. Our gross margin has been and will continue to be affected by a variety of factors, including average selling prices, sales mix for our implant systems, costs associated with third-party manufacturing, seasonality of our business and costs of other services. We expect our gross margin to remain consistent for the foreseeable future as our net revenue grows and our related costs of goods sold increases.
99
Operating Expenses
Our operating expenses consist of (i) selling, general and administrative expenses and (ii) research and development expenses.
Selling, General and Administrative Expenses
Selling, general and administrative expenses consist primarily of personnel costs, including commissions, salaries, bonuses, benefits and stock-based compensation related to personnel performing selling, marketing and general and administrative functions, including the costs associated with marketing initiatives and medical education programs. All of our stock-based compensation charges are included in selling, general and administrative expenses. In addition, selling, general and administrative expenses include depreciation expense for instruments, royalty payments made to product design surgeons, royalty payments made pursuant to our License Agreement (as defined below), travel expenses, professional services fees (including consulting, legal, finance, audit and tax fees), insurance costs, allocated facility expenses and other general corporate expenses.
We expect our selling, general and administrative expenses to continue to increase for the foreseeable future as we continue to grow our business and increase our utilization of internal and external resources within our commercial organization. As we continue to invest in growth, we will be required to maintain significant levels of instrumentation, which we expect to increase our selling, general and administrative expenses. Furthermore, the royalty payments made pursuant to our License Agreement will increase as our net revenue increases. Additionally, we anticipate increased expenses related to audit, legal, regulatory and tax-related services associated with being a public company, compliance with exchange listing and SEC requirements, director and officer insurance premiums and investor relations costs. We also expect to see an increase in our stock-based compensation expense with the establishment of a new publicly-traded company equity plan and to the extent of grants in the form of restricted stock units or options.
Research and Development Expenses
Research and development expenses consist of costs incurred in performing or for the outsourcing of various research and development activities, including consulting fees and other expenses paid related to such activities, costs associated with our registry, any future clinical trial costs and costs related to prototypes and related supplies related to our research and development efforts. We maintain a procedurally focused approach to product development and have projects underway to add new systems and implants across multiple shoulder indications and to add additional functionality or versatility to our existing systems. We expect our research and development expenses to increase as we pursue development of new products and product enhancements.
Other Expense
Our other expense consists of (i) interest expense, net, (ii) change in fair value of Series E purchase option, (iii) change in fair value of convertible notes, net, and (iv) other expense (income), net.
Interest Expense, Net
Interest expense, net consists of interest expense related to our term loan facility (the “Trinity Loan Agreement”) with Trinity Capital Inc. (“Trinity Capital”), interest expense related to our convertible notes, and non-cash interest related to the amortization of debt discount, issuance costs and deferred interest associated with our indebtedness, as well as interest income earned on our cash, cash equivalents and marketable securities.
Other Expense (Income), Net
Other expense (income), net consists primarily of adjustment in the fair market value of marketable securities and change in fair value of warrant liabilities.
Change in Fair Value of Warrant Liabilities
Change in fair value of warrant liabilities consists of gains and losses resulting from the remeasurement of the fair value of our warrant liabilities at each balance sheet date. During the third quarter of 2025 and upon completion of the Company’s IPO, a final remeasurement of the fair value of the warrant liability was made and the warrants were reclassified to equity within the additional paid-in capital line item.
100
Results of Operations
Comparison of Years Ended December 31, 2025 and 2024
The following table sets forth the components of our statements of operations for the periods presented below:
| Year Ended December 31, | Change | ||||||||||||||||||||||
| 2025 | 2024 | $ | % | ||||||||||||||||||||
| ($ in thousands) | |||||||||||||||||||||||
| Net revenue | $ | 47,317 | $ | 31,623 | $ | 15,694 | 49.6 | % | |||||||||||||||
| Cost of goods sold | 11,115 | 7,282 | 3,833 | 52.6 | % | ||||||||||||||||||
| Gross profit | 36,202 | 24,341 | 11,861 | 48.7 | % | ||||||||||||||||||
| Operating expenses: | |||||||||||||||||||||||
Selling, general and administrative expenses(1) | 54,768 | 34,505 | 20,263 | 58.7 | % | ||||||||||||||||||
| Research and development expenses | 7,731 | 4,489 | 3,242 | 72.2 | % | ||||||||||||||||||
| Total operating expenses | 62,499 | 38,994 | 23,505 | 60.3 | % | ||||||||||||||||||
| Operating loss | (26,297) | (14,653) | (11,644) | 79.5 | % | ||||||||||||||||||
| Other expense | |||||||||||||||||||||||
| Interest expense, net | 70 | 1,316 | (1,246) | (94.7) | % | ||||||||||||||||||
Change in fair value of convertible notes, net | 2,217 | — | 2,217 | * | |||||||||||||||||||
| Change in fair value of Series E purchase option | 11,719 | — | 11,719 | * | |||||||||||||||||||
Other (income) expense, net | 56 | (350) | 406 | (116.0) | % | ||||||||||||||||||
| Total other expense | 14,062 | 966 | 13,096 | * | |||||||||||||||||||
| Loss before income tax expense | (40,359) | (15,619) | (24,740) | 158.4 | % | ||||||||||||||||||
| Income tax expense | — | — | — | — | |||||||||||||||||||
| Net loss | $ | (40,359) | $ | (15,619) | $ | (24,740) | 158.4 | % | |||||||||||||||
__________________
(1)Includes stock-based compensation expense of $996 thousand and $754 thousand for the years ended December 31, 2025 and 2024, respectively.
*Not meaningful
Net Revenue. Net revenue increased $15,694 thousand, or 49.6%, to $47,317 thousand for the year ended December 31, 2025, compared to $31,623 thousand for the year ended December 31, 2024. The increase in net revenue was due to an increase in the number of implant systems sold, as well as an increase in the number of customers.
Cost of Goods Sold and Gross Margin. Cost of goods sold increased $3,833 thousand, or 52.6%, to $11,115 thousand for the year ended December 31, 2025, compared to $7,282 thousand for the year ended December 31, 2024. This increase in cost of goods sold was primarily due to the increase in the number of our systems sold. Gross margin for the year ended December 31, 2025 decreased to 76.5%, compared to 77.0% for the year ended December 31, 2024.
Selling, General and Administrative Expenses. Selling, general and administrative expenses increased $20,263 thousand, or 58.7%, to $54,768 thousand for the year ended December 31, 2025, compared to $34,505 thousand for the year ended December 31, 2024. This increase in selling, general and administrative expenses was primarily due to a $7,365 thousand increase in personnel-related expenses as a result of increased headcount of our commercial organization, a $5,074 thousand increase in commissions and sales related costs due to higher sales of our systems, a $2,447 thousand increase in general corporate costs such as information technology, business development and insurance costs, a $4,365 thousand increase in legal, accounting, and professional services fees related and a $1,012 thousand increase in depreciation of surgical instruments.
Research and Development Expenses. Research and development expenses increased $3,242 thousand, or 72.2%, to $7,731 thousand for the year ended December 31, 2025, compared to $4,489 thousand for the year ended December 31, 2024. The increase in research and development expenses was due to our investment in new product development efforts, including an increase in external consulting fees of $1,888 thousand related to such efforts.
101
Interest Expense, Net. Interest expense, net decreased $1,246 thousand, or 94.7%, to $70 thousand for the year ended December 31, 2025, compared to $1,316 thousand for the year ended December 31, 2024. This decrease in interest expense, net was due to higher interest earned on marketable securities.
Change in fair value of convertible notes. Change in fair value of convertible notes was $2,217 thousand for the year ended December 31, 2025, compared to $0 for the year ended December 31, 2024. The convertible notes were entered into during the third quarter of 2025 and converted into shares of the Company’s common stock upon completion of the Company’s IPO. The increase resulted from recognition of the fair value change between issuance and the completion of the IPO. There were no convertible notes in 2024.
Change in fair value of Series E purchase option. Change in fair value of Series E purchase option was $11,719 thousand for the year ended December 31, 2025, compared to $0 for the year ended December 31, 2024. The Series E purchase option was issued in connection with the Company’s Series E preferred stock financing entered into during the first quarter of 2025. The increase resulted from recognition of the fair value change between issuance and the exercise of the Series E preferred stock purchase option in June 2025. There was no purchase option in 2024.
Other Expense (income), Net. Other expense, net increased by $406 thousand to $56 thousand for the year ended December 31, 2025, compared to other income, net of $350 thousand for the year ended December 31, 2024. This increase in other expense, net was due to a decrease of $269 thousand in gain on investment and an increase of $137 thousand in expense related to changes in the fair value of our preferred stock warrant liability.
Liquidity and Capital Resources
To date, our primary sources of capital have been from net revenue received from the sale of our implant systems, the sale of common stock in our IPO, proceeds from private placements of our convertible preferred stock and debt financing arrangements. On August 1, 2025, we completed our IPO, selling 5,000,000 shares of our common stock at $15.00 per share. Upon completion of our IPO, we received net proceeds of approximately $64,212 thousand, after deducting underwriting discounts and commissions and offering expenses. Since inception, we have raised a total of $114,600 thousand in net proceeds from private placements of our convertible preferred stock. As of December 31, 2025, we had cash, cash equivalents and marketable securities of $124,305 thousand and $15,000 thousand of principal outstanding under our Trinity Loan Agreement.
We have generated losses from our operations since our inception as reflected in our accumulated deficit of $97,400 thousand as of December 31, 2025. Our losses primarily resulted from the costs incurred in the development, sales, and marketing of our systems and providing support for our operations. We expect to continue to incur losses for the foreseeable future and to expend significant amounts of cash for the foreseeable future as we continue to scale our business, increase selling, general and administrative expenses to support the expansion of our commercial organization and efforts, increase general and administrative expenses to support being a publicly-traded company and invest in research and development activities.
Indebtedness
On August 7, 2023, we entered into the Trinity Loan Agreement, as amended on July 21, 2025, with Trinity Capital, as administrative agent and collateral agent (in such capacities, the “Agent”) and as a lender, and the other lenders from time to time party thereto, providing for term loans of up to an aggregate principal amount of $45,000 thousand, available in three tranches: (i) a $15,000 thousand tranche that was fully funded on the August 7, 2023, (ii) a $15,000 thousand tranche that expired on December 31, 2025 and (iii) a $15,000 thousand tranche available through December 31, 2026. The availability of the third tranche is subject to, among other things, our achievement of at least $45,000 thousand of annualized tailing six-month revenue by December 31, 2026. In connection with the Trinity Loan Agreement, as amended on July 21, 2025, we issued a warrant to purchase 87,157 shares of our Series D convertible preferred stock to Trinity Capital. Upon completion of the Company’s IPO the warrants converted to warrants to purchase shares of the Company’s common stock. The warrant has an exercise price of $10.33 per share and expires ten years from the date of its issuance. As of December 31, 2025, the aggregate outstanding principal balance under the Trinity Loan Agreement was $15,000 thousand. Additionally, the second tranche expired on December 31, 2025, prior to Company drawing on the tranche.
The term loans under the Trinity Loan Agreement bear interest at an annual rate equal to the greater of the prime rate plus 3.50% and 11.00%. Under the terms of the Trinity Loan Agreement, the prime rate is equal to the greater of 8.0% per year and the prime rate as reported in The Wall Street Journal. We are required to make monthly payments of interest only through maturity of the term loans on September 1, 2028 (“Maturity Date”). The unpaid balance of principal and accrued interest is due on the Maturity Date. The Trinity Loan Agreement provides that we can at any time prepay the term loans,
102
in whole or in part, subject to a prepayment premium equal to: (i) 2.50% of the then-outstanding principal amount of the term loans, if such prepayment occurs on or prior to the first anniversary of the Trinity Loan Agreement; (ii) 1.50% of the then-outstanding principal amount of the advance, if such prepayment occurs after the first anniversary of the Trinity Loan Agreement and on or prior to the second anniversary of the Trinity Loan Agreement; and (iii) 1.00% of the then-outstanding principal amount of the advance, if such prepayment occurs after the second anniversary of the Trinity Loan Agreement and prior to the Maturity Date. We are required to make an end of term payment equal to 3.00% of the aggregate principal amount of the term loans funded on the earlier of (i) the Maturity Date, (ii) the date that we prepay all of the outstanding principal in full or (iii) the date of acceleration of the balance of the outstanding term loans by the Agent. The term loans are secured by substantially all our assets, including intellectual property.
The Trinity Loan Agreement also includes customary affirmative and negative covenants and events of default. Upon the occurrence and continuance of an event of default the Agent may demand immediate repayment of all principal and unpaid interest under the Trinity Loan Agreement, and exercise remedies against us and the collateral securing the Trinity Loan Agreement. Events of default under the Trinity Loan Agreement include, among other things: (i) insolvency, bankruptcy or similar proceedings subject to a certain grace period in respect of any involuntary insolvency, bankruptcy or similar proceedings; (ii) failure to pay any debts due under the Trinity Loan Agreement or other loan documents on a timely basis; (iii) failure to observe any covenant or secured obligation under the Trinity Loan Agreement, subject to a certain cure period; (iv) occurrence of a material adverse change; (v) material misrepresentations; (vi) occurrence of any default under any material agreement (or termination thereof) or any other agreement resulting in a right by the applicable third party to accelerate debt in excess of $500 thousand; (vii) entry of certain final, non-appealable judgments against us in excess of $500 thousand not paid or bonded within 10 days of such entry; (viii) a change of control unless as a condition to the closing of such change of control all outstanding term loans will be paid in full; and (ix) certain changes in the composition of board of directors.
As of December 31, 2025, we were in compliance with all covenants contained in the Trinity Loan Agreement.
Future Funding Requirements
Based on our current operating plan, we believe that the expected cash generated from the sale of our systems, our existing cash, cash equivalents and marketable securities and amounts under our Trinity Loan Agreement, will be sufficient to fund our planned operating expenses and capital expenditure requirements for at least 12 months from the date hereof. We have based this estimate on assumptions that may prove to be wrong, and we could deplete our capital resources sooner than we expect. We may experience lower than expected cash generated from operating activities or greater than expected capital expenditures, cost of goods sold, or operating expenses, and may need to raise additional capital to fund operations, increase our commercial organization and efforts, further research and development activities, or acquire, invest in, or in-license other businesses, assets, or technologies.
Our future capital needs will depend upon many factors, including:
•the market awareness and adoption of our systems, including our InSet Glenoid and InSet humeral stem products;
•the scope, timing and costs of supporting the growth and expansion of our commercial organization and efforts;
•the cost and pace of our research and development activities;
•the costs associated with any product recall that may occur;
•the costs associated with the manufacture and supply of our products at increased production levels;
•the costs associated with securing additional suppliers and service providers;
•the scope, rate of progress and costs of our current or future clinical and registries as well as costs associated with complying with regulatory requirements;
•the cost and timing of additional regulatory clearances or approvals;
•the costs of attaining, defending, and enforcing our intellectual property rights;
•whether we acquire third-party products or technologies;
•the terms and timing of any other distribution, collaborative, licensing, and other arrangements that we may establish;
103
•the emergence of competing technologies or other adverse market developments;
•our ability to raise additional funds to finance our operations;
•debt service requirements;
•the rate at which we expand internationally; and
•the cost associated with being a public company.
We have based this estimate on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we expect. If we are unable to satisfy our liquidity requirements, including because of the risks described in this Annual Report, we may seek to raise any necessary additional capital through public or private equity offerings or debt financings, credit or loan facilities or a combination of one or more of these or other funding sources. Additional funds may not be available to us on acceptable terms or at all. If we fail to obtain necessary capital when needed on acceptable terms, or at all, we could be forced to delay, limit, reduce or terminate our product development programs, commercialization efforts or other operations. If we raise additional funds by issuing equity securities or convertible debt, our stockholders will suffer dilution and the terms of any financing may adversely affect the rights of our stockholders. In addition, as a condition to providing additional funds to us, future investors may demand, and may be granted, rights superior to those of existing stockholders. If we raise additional capital through collaborations agreements, licensing arrangements or marketing and distribution arrangements, we may have to relinquish valuable rights, future revenue streams, research programs or product or grant licenses that may not be favorable to us. Debt financing, if available, is likely to involve restrictive covenants limiting our flexibility in conducting future business activities, and, in the event of insolvency, debt holders would be repaid before holders of our equity securities received any distribution of our corporate assets.
Cash Flows
The following table shows a summary of our cash flows for each of the periods presented:
| Year Ended December 31, | Change | ||||||||||||||||||||||
| 2025 | 2024 | $ | % | ||||||||||||||||||||
| Net cash (used in) provided by: | |||||||||||||||||||||||
| Operating activities | $ | (28,612) | $ | (14,143) | $ | (14,469) | 102.3 | % | |||||||||||||||
| Investing activities | (95,597) | 13,964 | (109,561) | * | |||||||||||||||||||
| Financing activities | 144,957 | 66 | 144,891 | * | |||||||||||||||||||
Net increase (decrease) in cash and cash equivalents | $ | 20,748 | $ | (113) | $ | 20,861 | * | ||||||||||||||||
__________________
*Not meaningful
Operating Activities
For the year ended December 31, 2025, net cash used in operating activities was $28,612 thousand, consisting primarily of a net loss of $40,359 thousand and net cash used by changes in our operating assets and liabilities of $6,751 thousand, partially offset by non-cash charges of $18,498 thousand. The non-cash charges primarily consisted of changes in the fair value of our preferred stock warrant liability and Series E purchase option of $11,975 thousand, depreciation and amortization expense of $3,208 thousand, change in fair value of convertible notes, net of $2,217 thousand, and stock-based compensation expense of $996 thousand. Net cash used by changes in our operating assets and liabilities primarily consisted of an increase of $7,636 thousand to inventory, an increase of $3,299 thousand in trade accounts receivable and an increase of $1,087 thousand in prepaid expenses, partially offset by an increase of $1,902 thousand in other current liabilities and an increase of $3,369 thousand in accounts payable.
For the year ended December 31, 2024, net cash used in operating activities was $14,143 thousand, consisting primarily of a net loss of $15,619 thousand, partially offset by non-cash charges of $2,513 thousand and net cash provided by changes in our operating assets and liabilities of $1,037 thousand. The non-cash charges primarily consisted of depreciation and amortization expense of $2,196 thousand and stock-based compensation expense of $754 thousand, partially offset by realized gain on marketable securities of $482 thousand. Net cash used by changes in operating assets and liabilities primarily consisted of an increase of $4,032 thousand in inventory, partially offset by an increase of $3,124 thousand in accounts payable.
104
Investing Activities
For the year ended December 31, 2025, net cash used in investing activities was $95,597 thousand, consisting primarily of purchases of $119,491 thousand in marketable securities and $7,408 thousand in fixed assets purchases, partially offset by proceeds of $31,302 thousand from sales of our marketable securities.
For the year ended December 31, 2024, net cash provided by investing activities was $13,964 thousand, consisting primarily of the cash proceeds of $23,260 thousand from sales of our marketable securities, partially offset by purchases of $5,281 thousand in marketable securities and $4,015 thousand in fixed assets purchases.
Financing Activities
For the year ended December 31, 2025, net cash provided by financing activities was $144,957 thousand, consisting of proceeds of $64,212 thousand from the issuance of common stock, net of commissions and issuance costs from the Company’s IPO, proceeds of $39,863 thousand from the issuance convertible notes net of issuance costs, proceeds of $39,508 thousand from the issuance and sale of shares of our Series E convertible preferred stock, proceeds of $919 thousand from the exercise of common stock options and $455 thousand proceeds from the exercise of warrants for our Series Seed and Series B preferred stock.
For the year ended December 31, 2024, net cash provided by financings activities was $66 thousand, consisting of $66 thousand from proceeds from the exercise of common stock options.
Contractual Obligations and Commitments
Our contractual commitments will have an impact on our future liquidity. These commitments include future payments on our Trinity Loan Agreement, future payments on facility leases and certain royalty obligations. Where applicable, we calculate our obligation based on termination fees that can be paid to exit the contract.
Debt
The principal outstanding under our Trinity Loan Agreement was $15,000 thousand as of December 31, 2025, however, we are required to make monthly payments of interest only through September 2027. Following the interest-only period, we are required to make payments of interest and principal in monthly installments through maturity of the term loans on September 1, 2028.
Leases
We have entered into an operating lease for office space in Michigan. The lease has a five-year term, which commenced in July 2021 and is renewable for one additional five-year term upon expiration. We have entered into an operating lease for warehouse space in California. The lease has a three-year term, which commenced in March 2025 and is renewable for one additional one-year term upon expiration. As of December 31, 2025, the operating lease obligations under these operating leases were $125 thousand.
Royalties
On October 22, 2020, we entered into a software license agreement with Genesis Software Innovations, LLC (“Genesis Software”), which was amended and restated on June 10, 2025 and subsequently amended and restated on June 10, 2025 (as amended and restated, the “License Agreement”), pursuant to which we are required to pay Genesis Software certain payments, including royalty payments, until such time we have paid Genesis Software an aggregate of $7,000 thousand under the License Agreement. As of December 31, 2025, we have paid an aggregate of $5,687 thousand of the total $7,000 thousand, including royalties of $1,641 thousand and $1,169 thousand in the years ended December 31, 2025 and 2024, respectively.
Off-Balance Sheet Arrangements
We did not have during the periods presented, and we do not currently have, any off-balance sheet arrangements or any relationships with unconsolidated entities or financial partnerships, such as structured finance, special purpose entities, or variable interest entities, that were established for the purpose of facilitating off-balance sheet arrangements or other contractually narrow or limited purposes.
105
Critical Accounting Policies and Estimates
Management’s discussion and analysis of our financial condition and results of operations is based on our financial statements, which have been prepared in accordance with U.S. generally accepted accounting principles. The preparation of these financial statements requires us to make estimates and assumptions for the reported amounts of assets, liabilities, revenue, expenses and related disclosures. Our estimates are based on our historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions and any such differences may be material.
While our significant accounting policies are more fully described in Note 1 of our financial statements included elsewhere in this Annual Report, we believe the following discussion addresses our most critical accounting policies and estimates, which are those that are most important to our financial condition and results of operations and require our most difficult, subjective and complex judgments.
Revenue Recognition
Revenue is recognized as the performance obligations to deliver products are satisfied and are recorded based on the amount of consideration we expect to receive in exchange for satisfying the performance obligations. Our sales are recognized primarily when we transfer control to the customer, which is generally when we have received a purchase order and appropriate notification the product has been used or implanted. Products are primarily transferred to customers at a point in time.
Revenue represents the amount of consideration we expect to receive from customers in exchange for transferring products. Net revenue excludes sales taxes we collect from customers. Other costs to obtain and fulfill contracts are generally expensed as incurred due to the short-term nature of most of our sales. We extend terms of payment to our customers based on commercially reasonable terms for the markets of our customers, while also considering their credit quality. Shipping and handling costs charged to customers are included in net sales.
Our payment terms with customers are customary and vary by customer but typically range from 30 to 60 days. We have evaluated the terms of our arrangements and determined that they do not contain significant financing components.
Inventories
Inventories consist of finished goods purchased from third-party suppliers. Inventories are stated at the lower of average cost or net realizable value. We have applied these inventory cost valuation methods consistently from year to year. Two suppliers provide substantially all of our finished goods.
We record inventory reserves for estimated obsolescence or unmarketable inventory equal to the difference between the cost of inventory and the estimated net realizable value based upon the age of specific inventory on hand and assumptions about future demand and market conditions. Our inventory has an initial five year usable life and we maintain the ability to extend the usable life of inventory for an additional five years. As a result, our analysis focuses on to what extent inventory on hand is in excess of market demand, which requires assessment of various factors that require the use of judgment including forecasted sales, continued growth, impact of competitors’ products and ensuring we maintain adequate inventory on hand. If actual conditions differ from our assumptions, adjustments to the reserve may be required.
Inventories presented on the balance sheets are net of reserves for estimated obsolescence or unmarketable inventory which were $845 thousand and $287 thousand as of December 31, 2025 and 2024, respectively. Our inventories as of December 31, 2025 included $465 thousand of inventories that, as of December 31, 2025, did not have material sales in the preceding twelve months. These inventories were primarily comprised of implants of less commonly used sizes that are within various implant families made available to surgeons at the time of surgery. These inventories are included in our analysis of our reserve for estimated obsolescence or unmarketable inventory. A portion of these inventories have been reserved for, and the remaining portion of these inventories are expected to be sold through our existing distribution network or in future markets.
Stock-Based Compensation
We measure all stock options based on their fair value on the date of the grant. Those awards typically have a graded vesting schedule and compensation expense for awards with only service conditions are recognized on a straight-line basis over the requisite service period, which is generally the vesting period of the respective award. We have not issued any stock-based awards with performance-based or market-based vesting conditions.
106
We use the Black-Scholes option pricing model, which incorporates assumptions and estimates, to measure the fair value of its option awards on the date of grant of each stock option award. We determined the assumptions for the Black-Scholes option pricing model as discussed below. Each of these inputs described below is subjective and generally requires significant judgment to determine. Forfeitures were accounted for as they occurred.
•Expected Term. The expected term represents the period that the stock-based awards are expected to be outstanding. As we do not have sufficient historical exercise data to provide a reasonable basis upon which to estimate the expected term, the expected term of stock options granted has been determined using the simplified method, which is the average of the midpoints between the vesting date and the contractual term for all vesting tranches.
•Risk-Free Interest Rate. The risk-free interest rate is based on the rate of the U.S. Treasury yield curve in effect at the time of grant of the award for time periods approximately equal to the expected term of the award.
•Expected Volatility. Because we do not have a trading history of our common stock, the expected volatility was derived from the average historical stock volatilities of several public companies within our industry that we consider to be comparable to our business over a period equivalent to the expected term of the stock-based award.
•Expected Dividend Yield. The expected dividend yield is zero as we have not paid and do not anticipate paying any dividends for the foreseeable future.
If any of the assumptions used in the Black-Scholes option pricing model change significantly, stock-based compensation for future awards may differ materially compared with the awards granted previously. See Note 10 to our financial statements included elsewhere in this Annual Report for further details.
Determination of Fair Value of Warrant Liabilities
Given the absence of a public trading market to date, the fair value of our common stock has been determined by our board of directors at the time of issuance of warrants, with input from management, considering contemporaneous independent third-party valuations of common and preferred stock, and our board of directors’ assessment of additional objective and subjective factors that it believed were appropriate.
Our common stock and preferred stock warrants require liability classification and accounting. The warrants are recorded at fair value upon issuance and are subject to remeasurement to fair value at each balance sheet date, with any changes in fair value recognized in other (income) expense, net. The fair value was estimated using a Black-Scholes option pricing model. The valuation model used incorporates significant assumptions and estimates, which include, but are not limited to, the fair value per share of the underlying shares, the remaining contractual term of the warrants, risk-free interest rate and expected volatility of the price of the underlying shares.
Recently Issued Accounting Pronouncements
See Note 1 to our financial statements included elsewhere in this Annual Report for a description of recent accounting pronouncements applicable to our financial statements.
Emerging Growth Company and Smaller Reporting Company Status
We are an emerging growth company, as defined in the JOBS Act. The JOBS Act permits an “emerging growth company” such as us to take advantage of an extended transition period for complying with new or revised accounting standards. This provision allows an emerging growth company to delay the adoption of some accounting standards until those standards would otherwise apply to private companies. We have elected to use the extended transition period for any other new or revised accounting standards during the period in which we remain an emerging growth company; however, we may adopt certain new or revised accounting standards early. As a result, we will not be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies and our financial statements may not be comparable to other public companies that comply with new or revised accounting pronouncements as of public company effective dates. The JOBS Act also exempts us from having to provide an attestation and report from our independent registered public accounting firm on the assessment of our internal control over financial reporting pursuant to the Sarbanes-Oxley Act.
We will remain an emerging growth company until the earliest of: (i) the last day of the fiscal year following the fifth anniversary of the consummation of this offering; (ii) the last day of the fiscal year in which we have total annual gross revenue of at least $1.235 billion; (iii) the last day of the fiscal year in which we are deemed to be a “large accelerated
107
filer” as defined in Rule 12b-2 under the Exchange Act, which would occur if the market value of our common stock held by non-affiliates exceeded $700.0 million as of the last business day of the second fiscal quarter of such year; or (iv) the date on which we have issued more than $1.0 billion in non-convertible debt securities during the prior three-year period.
We are also a “smaller reporting company” as defined in the Exchange Act. We may continue to be a smaller reporting company even after we are no longer an emerging growth company. We may take advantage of certain of the scaled disclosures available to smaller reporting companies and will be able to take advantage of these scaled disclosures for so long as our voting and non-voting common stock held by non-affiliates is less than $250.0 million measured on the last business day of our second fiscal quarter, or our annual revenue is less than $100.0 million during the most recently completed fiscal year and our voting and non-voting common stock held by non-affiliates is less than $700.0 million measured on the last business day of our second fiscal quarter. We cannot predict if investors will find our shares of common stock less attractive because we may rely on these exemptions. If some investors find our shares of common stock less attractive as a result, there may be a less active trading market for shares of our common stock and our share price may be more volatile.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
Interest Rate Risk
The primary risk associated with fluctuating interest rates is related to our debt. The term loans under the Trinity Loan Agreement bear interest at an annual rate equal to the greater of the prime rate plus 3.50% and 11.00%. Under the terms of the Trinity Loan Agreement, the prime rate is equal to the greater of 8.0% per year and the prime rate as reported in The Wall Street Journal. In addition, we hold cash and cash equivalents as well as marketable securities, all of which may generate interest income. The primary objectives of our investment activities are to preserve principal and provide liquidity. Since our results of operations are not dependent on investments, we believe the risk associated with fluctuating interest rates is limited. We do not believe that a hypothetical 10% increase or decrease in interest rates during any of the periods presented would have had a material negative effect on our financial statements included elsewhere in this Annual Report considering the balance of outstanding debt compared to the balance of marketable securities on the Company’s Balance Sheets. We do not currently use or plan to use financial derivatives in our investment portfolio and we do not currently engage in hedging transactions to manage our exposure to interest rate risk.
Financial Institution Risk
Substantially all of our cash, cash equivalents and marketable securities are held with two financial institutions. Cash amounts held at financial institutions are insured by the Federal Deposit Insurance Corporation up to $250,000.
Inflation Risk
Inflation generally affects us by increasing our cost of labor. Inflationary and supply chain pressures may adversely impact our future financial results. Our operating costs have increased and may continue to increase because of these pressures, and we may not be able to fully offset these cost increases by raising prices for products or other mitigation efforts, which could result in downward pressure on margins.
108
Item 8. Financial Statements and Supplementary Data
Table of Contents
Consolidated Financial Statements
Independent Auditor’s Report (PCAOB ID: | |||||
| Audited Financial Statements | |||||
109
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Stockholders and Board of Directors of Shoulder Innovations, Inc.
Opinion on the Financial Statements
We have audited the accompanying balance sheets of Shoulder Innovations, Inc. (the "Company") as of December 31, 2025 and 2024, the related statements of operations and comprehensive loss, changes in convertible preferred stock and stockholders' equity (deficit), and cash flows, for each of the two years in the period ended December 31, 2025, and the related notes (collectively referred to as the "financial statements"). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2025 and 2024, and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2025, in conformity with accounting principles generally accepted in the United States of America.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company's financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Deloitte & Touche LLP
March 10, 2026
We have served as the Company’s auditor since 2023.
110
Shoulder Innovations, Inc.
Balance Sheets
(in thousands, except share and per share amounts)
| December 31, 2025 | December 31, 2024 | ||||||||||
| Assets | |||||||||||
| Current Assets | |||||||||||
| Cash and cash equivalents | $ | $ | |||||||||
| Marketable securities | |||||||||||
| Trade accounts receivable, net of allowance for credit losses | |||||||||||
| Inventories, net | |||||||||||
| Prepaid expenses | |||||||||||
| Other current assets | |||||||||||
| Total Current Assets | |||||||||||
| Property and equipment, net | |||||||||||
| Operating lease right-of-use asset | |||||||||||
| Intangible assets, net | |||||||||||
| Total Assets | |||||||||||
Liabilities, Convertible Preferred Stock, and Stockholders’ Equity (Deficit) | |||||||||||
| Current Liabilities | |||||||||||
Accounts payable ($ | |||||||||||
| Current operating lease obligations | |||||||||||
Accrued liabilities ($ | |||||||||||
| Total Current Liabilities | |||||||||||
| Long-Term Liabilities | |||||||||||
| Preferred stock warrant liability | |||||||||||
| Long-term debt | |||||||||||
| Long-term operating lease obligations | |||||||||||
| Total Long-Term Liabilities | |||||||||||
| Total Liabilities | |||||||||||
| Commitments and contingencies (note 13) | |||||||||||
| Convertible Preferred Stock | |||||||||||
Series Seed, $ | |||||||||||
Series A, $ | |||||||||||
Series B, $ | |||||||||||
Series C, $ | |||||||||||
Series D, $ | |||||||||||
| Total Convertible Preferred Stock | |||||||||||
Stockholders’ Equity (Deficit) | |||||||||||
111
Shoulder Innovations, Inc.
Balance Sheets
(in thousands, except share and per share amounts)
Common stock, $ | |||||||||||
Preferred stock, $ | |||||||||||
| Additional paid-in capital | |||||||||||
| Accumulated deficit | ( | ( | |||||||||
| Accumulated other comprehensive income | |||||||||||
Total Stockholders’ Equity (Deficit) | ( | ||||||||||
Total Liabilities, Convertible Preferred Stock, and Stockholders’ Equity (Deficit) | $ | $ | |||||||||
See accompanying notes to financial statements.
112
Shoulder Innovations, Inc.
Statements of Operations and Comprehensive Loss
(in thousands, except share and per share amounts)
Year Ended December 31, | |||||||||||
| 2025 | 2024 | ||||||||||
| Net Revenue | $ | $ | |||||||||
| Cost of Goods Sold | |||||||||||
| Gross Profit | |||||||||||
Selling, General, and Administrative Expenses (Includes $ | |||||||||||
Research and Development Expenses (Includes $ | |||||||||||
| Operating Loss | ( | ( | |||||||||
| Other Expense | |||||||||||
| Interest expense, net | |||||||||||
Change in fair value of convertible notes, net | |||||||||||
Change in fair value of Series E purchase option | |||||||||||
| Other (income) expense, net | ( | ||||||||||
| Total Other Expense | |||||||||||
| Loss before income tax expense | ( | ( | |||||||||
| Income Tax Expense | |||||||||||
| Net Loss | $ | ( | $ | ( | |||||||
Other Comprehensive loss, net | |||||||||||
Unrealized loss on marketable securities | ( | ( | |||||||||
Total Other Comprehensive loss, net | ( | ( | |||||||||
| Comprehensive loss | $ | ( | $ | ( | |||||||
| Net loss per share attributed to common stock – basic and diluted: | |||||||||||
| Net loss per share | $ | ( | $ | ( | |||||||
| Weighted average shares outstanding: | |||||||||||
| Weighted average common shares outstanding – basic and diluted | |||||||||||
See accompanying notes to financial statements.
113
Shoulder Innovations, Inc.
Statements of Changes in Convertible Preferred Stock and Stockholders’ Equity (Deficit)
(in thousands, except shares and per share amounts)
| Common Stock | ||||||||||||||||||||||||||||||||||||||||||||
| Preferred Stock | Shares | Amount | Additional Paid-in Capital | Accumulated Other Comprehensive Income | Accumulated Deficit | Total Stockholders’ Equity (Deficit) | ||||||||||||||||||||||||||||||||||||||
| Balance, December 31, 2023 | $ | $ | $ | $ | $ | ( | $ | ( | ||||||||||||||||||||||||||||||||||||
| Issuance of common stock | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||
| Stock-based compensation expense | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||
| Net Loss | — | — | — | — | — | ( | ( | |||||||||||||||||||||||||||||||||||||
Other Comprehensive loss, net | — | — | — | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||
| Balance, December 31, 2024 | $ | $ | $ | $ | $ | ( | $ | ( | ||||||||||||||||||||||||||||||||||||
Issuance of preferred stock in private placement, net of issuance cost of $ | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||
Exercise of preferred stock warrant | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||
| Conversion of preferred stock to common stock upon IPO | ( | — | — | |||||||||||||||||||||||||||||||||||||||||
Issuance of common stock upon IPO, net of offering costs of $ | — | — | — | |||||||||||||||||||||||||||||||||||||||||
| Conversion of redeemable convertible preferred stock warrants to common stock warrants on IPO | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||
| Conversion of convertible notes to common stock upon IPO | — | — | — | |||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||
Exercise of common stock warrants | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||
| Stock-based compensation expense | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||
| Net Loss | — | — | — | — | — | ( | ( | |||||||||||||||||||||||||||||||||||||
Other Comprehensive loss, net | — | — | — | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||
| Balance, December 31, 2025 | $ | $ | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||||||
See accompanying notes to financial statements.
114
Year Ended December 31, | |||||||||||
| 2025 | 2024 | ||||||||||
| Cash Flows from Operating Activities | |||||||||||
| Net loss | $ | ( | $ | ( | |||||||
| Adjustments to reconcile net loss to net cash used in operating activities | |||||||||||
| Depreciation and amortization | |||||||||||
| Amortization of debt discounts | |||||||||||
Credit losses, net of recoveries | |||||||||||
Loss on disposal of long-lived assets | |||||||||||
| Stock compensation expense | |||||||||||
| Realized gain on marketable securities | ( | ( | |||||||||
Change in fair value of warrant liabilities and Series E purchase option | |||||||||||
Change in fair value of convertible notes, net | |||||||||||
| Other | ( | ||||||||||
| Change in Operating Assets and Liabilities | |||||||||||
| Trade accounts receivable | ( | ( | |||||||||
| Inventory | ( | ( | |||||||||
| Prepaid expenses | ( | ( | |||||||||
| Accounts payable | |||||||||||
Other current assets and liabilities | |||||||||||
| Net Cash Used in Operating Activities | ( | ( | |||||||||
| Cash Flows from Investing Activities | |||||||||||
| Fixed asset purchases | ( | ( | |||||||||
| Purchases of marketable securities | ( | ( | |||||||||
| Sales of marketable securities | |||||||||||
| Net Cash Provided (Used) in Investing Activities | ( | ||||||||||
| Cash Flows from Financing Activities | |||||||||||
| Proceeds from exercise of common stock options | |||||||||||
Proceeds from exercise of preferred stock warrants | |||||||||||
Proceeds from Series E convertible preferred stock, net of issuance cost | |||||||||||
Proceeds from the issuance of convertible notes, net of issuance costs | |||||||||||
Proceeds from IPO, net of commissions and issuance costs | |||||||||||
| Net Cash Provided in Financing Activities | |||||||||||
| Net Change in Cash for Period | ( | ||||||||||
| Cash and Cash Equivalents, beginning of year | |||||||||||
| Cash and Cash Equivalents, end of year | $ | $ | |||||||||
| Supplemental Cash Flows information | |||||||||||
| Cash paid for interest | $ | $ | |||||||||
| Non-Cash Investing and Financing Activities | |||||||||||
Series E second tranche right equity conversion | $ | $ | |||||||||
Preferred stock warrant liability equity conversion | $ | $ | |||||||||
Preferred stock warrant liability equity conversion on completion of IPO | $ | $ | |||||||||
Conversion of redeemable convertible preferred stock into common stock on completion of IPO | $ | $ | |||||||||
Conversion of convertible notes into common stock on completion of IPO | $ | $ | |||||||||
Capital expenditures included in accounts payable | $ | $ | |||||||||
See accompanying notes to financial statements.
115
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
1. Summary of Significant Accounting Policies
Business Activity
Shoulder Innovations, Inc. (the Company) is principally involved in developing next generation shoulder replacement implants, utilizing contract manufacturing partners, and distributing them nationwide for surgeries through a network of employed and contracted sales representatives. The Company is headquartered in Grand Rapids, Michigan and markets and sells its products throughout the United States.
Reverse Stock Split
On July 23, 2025, the Company amended its amended and restated certificate of incorporation to effect a reverse stock split of shares of the Company’s common stock on a 1-for-19.08 basis (the “Reverse Stock Split”). The common stock warrants and options to purchase common stock were subsequently adjusted as a result of the Reverse Stock Split. All impacted share and per-share information included in these financial statements and notes thereto have been retroactively adjusted to give effect to the Reverse Stock Split.
Initial Public Offering
On August 1, 2025, the Company closed its initial public offering (“IPO”), and issued 5,000,000 shares of common stock at public offering price of $15.00 per share. The Company received net proceeds of approximately $64,212 , after deducting underwriting discounts and commissions and offering expenses. Immediately prior to the closing of the IPO, all outstanding shares of convertible preferred stock and $40,000 in aggregate principal amount of convertible notes converted into 15,176,862 shares of common stock. In connection with the closing of the IPO, on August 1, 2025, the Company amended and restated its certificate of incorporation to authorize the issuance of up to 730,000,000 shares of common stock, par value $0.001 per share and 20,000,000 shares of preferred stock, par value $0.001 per share.
Emerging Growth Company
The Company is an “emerging growth company,” as defined in Section 2(a) of the Securities Act, as modified by the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”), and it may take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies including, but not limited to, not being required to comply with the independent registered public accounting firm attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in its periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved.
Further, Section 102(b)(1) of the JOBS Act exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies (that is, those that have not had a Securities Act registration statement declared effective or do not have a class of securities registered under the Exchange Act) are required to comply with the new or revised financial accounting standards. The JOBS Act provides that an emerging growth company can elect to opt out of the extended transition period and comply with the requirements that apply to non-emerging growth companies but any such an election to opt out is irrevocable. The Company has elected not to opt out of such extended transition period, which means that when a standard is issued or revised and it has different application dates for public or private companies, the Company, as an emerging growth company, can adopt the new or revised standard at the time private companies adopt the new or revised standard. This may make comparison of the Company’s financial statements with another public company that is neither an emerging growth company nor an emerging growth company that has opted out of using the extended transition period difficult or impossible because of the potential differences in accounting standards used.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America (“GAAP”) requires management to make estimates and assumptions that affect the reported amounts of
116
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates.
Concentrations of Credit Risk
Financial instruments, which potentially subject us to concentrations of credit risk, are primarily marketable securities and trade accounts receivable. Concentrations of credit risk with respect to accounts receivable are limited due to the number of entities comprising our customer base. We perform ongoing credit evaluations of our customers and generally do not require collateral.
There was no customer that accounted for 10% or more of revenue, or accounts receivable for the years ended December 31, 2025 and 2024, respectively.
Cash and Cash Equivalents
The Company considers all highly liquid funds with an original contractual maturity on the date of purchase of three months or less to be classified and presented as cash equivalents. The Company maintains cash balances in bank checking accounts. From time to time during the year, cash deposited in these accounts may be in excess of the federally insured limit.
Marketable Securities
The Company’s marketable securities consist predominately of investment-grade, U.S. dollar-denominated fixed and floating-rate debt, which are classified as available-for-sale and are valued in accordance with the fair value measurement guidance. Available-for-sale securities are carried at fair value with unrealized gains and losses reported in the statements of operations and comprehensive loss as other comprehensive loss, net of tax. Realized gains and losses, if any, are calculated on the specific identification method and are included in the statements of operations and comprehensive loss.
Available-for-sale debt securities are recorded at fair value. When the fair value of the securities declines below the amortized cost basis, impairment is indicated and it must be determined whether it is other than temporary. Impairment is considered to be other than temporary if the Company: (i) intends to sell the security, (ii) will more likely than not be forced to sell the security before recovering its cost, or (iii) does not expect to recover the security’s amortized cost basis. If the decline in fair value is considered other than temporary, the cost basis of the security is adjusted to its fair market value and the realized loss is reported in other (income) expense, net. Subsequent increases or decreases in fair value are reported as a component of stockholders’ equity in accumulated other comprehensive income.
Interest income from the Company’s marketable securities for the years ended December 31, 2025 and 2024 totaled $1,948 and $770 , respectively, and is included in Interest expense, net on the statements of operations and comprehensive loss.
Trade Accounts Receivable
December 31 | |||||||||||
| 2025 | 2024 | ||||||||||
| Beginning Balance | $ | $ | |||||||||
| Provision | |||||||||||
| Write-offs | ( | ||||||||||
| Ending Balance | $ | $ | |||||||||
117
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
Inventories
December 31, | |||||||||||
| 2025 | 2024 | ||||||||||
| Finished Goods | $ | $ | |||||||||
| Inventory reserve | ( | ( | |||||||||
| Inventories, net | $ | $ | |||||||||
Property and Equipment and Depreciation
Property and equipment are stated at cost, less accumulated depreciation. Property and equipment are depreciated using straight-line methods over the estimated useful lives of the related assets. Leasehold improvements are amortized over the shorter of the remaining lease term or the estimated useful lives of the assets. Maintenance and repairs are charged to expense as incurred.
When items of property or equipment are sold or retired, the related costs and accumulated depreciation are removed from the accounts and any gain or loss is included in the results of operations.
Surgical instruments are provided to surgeons and hospitals to facilitate the use of the Company’s products. The Company does not receive additional or separate consideration for the use of its surgical instruments by surgeons and hospital staff. Surgical instruments are owned and controlled by the Company and are used to generate long-term economic benefits. The Company has concluded that surgical instruments meet the criteria under ASC 360 - Property, Plant and Equipment and that appropriate expense recognition is through depreciation over the useful life of the surgical instruments.
Estimated useful lives for financial reporting purposes are as follows:
| Asset Category | Useful Life (Years) | |||||||
| Machinery and equipment | ||||||||
| Instruments | ||||||||
| Computer equipment | ||||||||
| Furniture and Fixtures | ||||||||
| Leasehold Improvements | ||||||||
Long-Lived Assets
The Company reviews long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. Impairment is determined by comparing the carrying value of the assets to their estimated future undiscounted cash flows. If the sum of the estimated future undiscounted cash flows expected to result from the use and eventual disposition of an asset is less than the carrying amount of the asset, an impairment loss is recognized. If it is determined that an impairment has occurred, the asset is written down to its estimated fair value and a charge to income is recognized. The Company has not recorded any impairment losses on long-lived assets during the years ended December 31, 2025 and 2024. As of December 31, 2025 and 2024, all long-lived assets were located in the U.S.
118
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
Intangible Assets
| December 31, 2025 | Useful Life | Gross Carrying Amount | Accumulated Amortization | Net Carrying Value | ||||||||||||||||||||||
| Software License | $ | $ | $ | |||||||||||||||||||||||
| December 31, 2024 | Useful Life | Gross Carrying Amount | Accumulated Amortization | Net Carrying Value | ||||||||||||||||||||||
| Software License | $ | $ | $ | |||||||||||||||||||||||
Amortization expense in each of the next three years is expected to be the following:
Year ending December 31, | ||||||||
| 2026 | $ | |||||||
| Total | $ | |||||||
Advertising Costs
Research and Development Expenses
Research and development expenses are expensed in the period in which they are incurred and totaled $7,731 and $4,489 for the years ended December 31, 2025 and 2024, respectively. Research and development expenses may include costs incurred in performing research and development activities, including clinical trial costs, manufacturing costs for both clinical and pre-clinical materials as well as other contracted services, and other external costs. Non-refundable advance payments for goods and services that will be used in future research and development activities are expensed when the activity is performed or when the goods have been received, rather than when payment is made, in accordance with ASC 730, Research and Development.
Presentation of Sales Taxes
Income Taxes
Provision for income taxes is based on amounts reported in the statements of operations and comprehensive loss and include deferred taxes on temporary differences for tax and financial statement purposes. Temporary differences arise from differences between methods used for tax purposes and book purposes. Deferred taxes are computed using the liability method as prescribed by ASC 740, Income Taxes. The Company recorded a valuation allowance to reduce deferred tax assets to the amount that is believed more likely than not to be realized.
The Company follows the provisions of ASC 740-10-25, Accounting for Uncertainty in Income Taxes, which seeks to reduce the diversity in practice associated with certain aspects of the recognition and measurement related to accounting for
119
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
income taxes. Under ASC 740-10-25, an organization must recognize the tax benefit associated with tax positions taken for tax return purposes when it is more likely than not that the position will be sustained. The Company recognizes any corresponding interest and penalties associated with its income tax positions in income tax expense. The Company records liabilities in which it believes the position is not more likely than not sustainable.
There are no
Convertible Preferred Stock Warrants
The Company accounts for its freestanding warrants on its convertible preferred stock as liabilities, as the instruments underlying the warrants are classified outside of permanent equity. The underlying shares of convertible preferred stock are classified as mezzanine equity due to contingent redemption provisions outside of the Company’s control. Such warrants are measured and recognized at fair value and subject to re-measurement at each balance sheet date. At the end of each reporting period, changes in fair value during the period are recognized as a component of other expense in the accompanying statements of operations and comprehensive loss until the warrants are exercised or expire. Upon completion of the Company’s IPO all outstanding preferred stock warrants converted to common stock warrants.
Revenue Recognition
Revenue is recognized as the performance obligation to deliver products is satisfied and are recorded based on the amount of consideration the Company expects to receive in exchange for satisfying the performance obligations. Our sales are recognized primarily when the Company transfers control to the customer, which is generally when the Company has received a purchase order and appropriate notification that the product has been used or implanted. Products are primarily transferred to customers at a point in time.
Revenue represents the amount of consideration the Company expects to receive from customers in exchange for transferring products. Net revenue exclude sales taxes the Company collects from customers. Other costs to obtain and fulfill contracts are generally expensed as incurred due to the short-term nature of most of our sales. The Company extends terms of payment to our customers based on commercially reasonable terms for the markets of our customers, while also considering their credit quality. Shipping and handling costs charged to customers are included in net sales. Total shipping and handling costs for the years ended December 31, 2025 and 2024 were $901 and $456 , respectively.
The Company’s payment terms with customers are customary and vary by customer but typically range from 30 to 60 days. The Company has evaluated the terms of its arrangements and determined that they do not contain significant financing components.
The Company does not provide any warranty on their products other than for implied use. The Company has not experienced any warranty claims and does not carry a reserve.
Employee Benefit Plan
New Accounting Pronouncements
In August 2020, the FASB issued ASU 2020-06, Debt — Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging — Contracts in Entity’s Own Equity (Subtopic 815-40), which simplifies the accounting for certain financial instruments with characteristics of liabilities and equity, including convertible instruments and contracts on an entity’s own equity. ASU 2020-06 permits adoption on a retrospective basis to financial instruments outstanding as of the beginning of the first comparative reporting period presented. The Company adopted the new standard on January 1, 2024. There was not a significant impact to the financial statements as a result of this pronouncement.
120
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
In November 2023, the FASB issued ASU 2023-07—Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosures was designed to improve reportable segment disclosure requirements, primarily through enhanced disclosures about significant segment expenses. The Company adopted the new standard on January 1, 2024. There was not a significant impact to the Company’s balance sheets, statements of operations and comprehensive loss or statement of cash flows. The adoption impacted the Company’s financial statement disclosure in Note 12.
In December 2023, the FASB issued ASU 2023-09, Income Taxes (Topic 740) (“ASC 740”). The update requires all public business entities on an annual basis to (1) disclose specific categories in the rate reconciliation and (2) provide additional information for reconciling items that meet a quantitative threshold and an explanation, if not otherwise evident, of the individual reconciling items disclosed, such as the nature, effect, and underlying causes of the reconciling items and the judgment used in categorizing the reconciling items. In addition, the update requires certain new disclosures of the amount of income taxes paid (net of refunds received) disaggregated by federal, state, and foreign taxes and the amount of income taxes paid (net of refunds received) disaggregated by individual jurisdictions in which income taxes paid is equal to or greater than five percent of total income taxes paid (net of refunds received). Other new disclosures required include income (or loss) from continuing operations before income tax expense (or benefit) disaggregated between domestic and foreign and income tax expense (or benefit) from continuing operations disaggregated by federal, state, and foreign. The new guidance is effective for annual periods beginning after December 15, 2024. Early adoption is permitted. The amendments are to be applied on a prospective basis, with retrospective application permitted. As an emerging growth company that has not opted out of the extended transition period for complying with new or revised financial accounting standards, the amendments in ASU 2023-09 are effective for annual periods beginning after December 15, 2025, with early adoption permitted. The Company is currently evaluating the potential impact of adopting this new guidance on the Company’s financial statements and related disclosures.
In November 2024, the FASB issued ASU 2024-03, Income Statement - Reporting Comprehensive Income - Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses requires additional, disaggregated disclosure around certain income statement expense line items. This ASU mandates that entities, at each interim and annual period, disclose the amounts of (a) inventory purchases, (b) employee compensation, (c) depreciation, (d) intangible asset amortization, and (e) depletion, depreciation, and amortization for oil and gas activities included within each relevant expense caption presented on the income statement within continuing operations. Entities are also required to (1) combine certain disclosures already mandated under GAAP with these new requirements, (2) provide qualitative descriptions of expenses that are not disaggregated quantitatively, and (3) disclose total selling expenses and, annually, the definition of selling expenses. The guidance is effective for annual periods beginning after December 15, 2026, and interim periods beginning after December 15, 2027. Early adoption is permitted. The Company is currently evaluating the potential impact of adopting this new guidance on the Company’s financial statements and related disclosures.
2. Fair Value Measurements
The Company utilizes fair value measurement guidance prescribed by accounting standards to value its financial instruments. The guidance establishes a fair value hierarchy for financial instruments measured at fair value that distinguishes between assumptions based on market data (observable inputs) and the Company’s own assumptions (unobservable inputs). Observable inputs are inputs that market participants would use in pricing the asset or liability based on market data obtained from sources independent of the Company. Unobservable inputs are inputs that reflect the Company’s assumptions about the inputs that market participants would use in pricing the asset or liability and are developed based on the best information available in the circumstances.
Fair value is defined as the exchange price, or exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants on the measurement date. The fair value hierarchy contains three levels of inputs that may be used to measure fair value, in accordance with ASC 820, Fair Value Measurement, of which the first two are considered observable and the last is considered unobservable. These levels are as follows:
Level 1 – Inputs at this level include unadjusted quoted prices in active markets for identical assets or liabilities that the Company has the ability to access.
Level 2 – Inputs at this level include observable inputs other than Level 1 inputs, such as quoted prices for similar assets or liabilities in active markets, quoted prices for identical or similar assets or liabilities in markets that are not active,
121
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the asset or liability.
Level 3 – Inputs at this level include unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the underlying asset or liability. Level 3 assets and liabilities include those whose fair value measurements are determined using pricing models, discounted cash flow methodologies, or similar valuation techniques, as well as significant management judgment or estimation.
To the extent the valuation is based on models or inputs that are less observable or unobservable in the market, the determination of fair value requires more judgment. Accordingly, the degree of judgment exercised by the Company in determining fair value is greatest for instruments categorized in Level 3. A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement.
The recorded amounts of certain financial instruments, including cash, cash equivalents, accounts receivable, accounts payable, and accrued liabilities approximate fair value due to their relatively short maturities.
The fair value of marketable securities as of December 31, 2025 and 2024 are summarized below:
| December 31, 2025 | ||||||||||||||||||||
| Level 1 | Level 2 | Level 3 | ||||||||||||||||||
| Assets: | ||||||||||||||||||||
| Cash Equivalents | ||||||||||||||||||||
| Money market funds | $ | $ | $ | |||||||||||||||||
| Short-term marketable securities at fair value | ||||||||||||||||||||
| U.S. treasury and government agencies | ||||||||||||||||||||
| Corporate and international bonds | ||||||||||||||||||||
| Total assets | $ | $ | $ | |||||||||||||||||
| December 31, 2024 | ||||||||||||||||||||
| Level 1 | Level 2 | Level 3 | ||||||||||||||||||
| Assets: | ||||||||||||||||||||
| Cash Equivalents | ||||||||||||||||||||
| Money market funds | $ | $ | $ | |||||||||||||||||
| Short-term marketable securities at fair value | ||||||||||||||||||||
| U.S. treasury and government agencies | ||||||||||||||||||||
| Corporate and international bonds | ||||||||||||||||||||
| Total assets | $ | $ | $ | |||||||||||||||||
| Liabilities: | ||||||||||||||||||||
| Preferred stock warrant liability | $ | |||||||||||||||||||
Warrants
Liabilities related to stock warrants are remeasured at fair value on a recurring basis using the Black-Scholes option pricing model. The following table presents the change in fair value of the stock warrants which are classified in Level 3 of the fair value hierarchy for the years ended December 31, 2025 and 2024:
122
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
| December 31, 2025 | |||||||||||
Preferred Stock warrant liability | Series E purchase option | ||||||||||
Balance January 1, 2025 | $ | $ | |||||||||
Additions | |||||||||||
Change in fair value | |||||||||||
Exercises | ( | ( | |||||||||
Conversion to equity upon IPO | $ | ( | $ | ||||||||
Balance December 31, 2025 | $ | $ | |||||||||
The following table presents the change in fair value of the preferred stock warrants which are classified in Level 3 of the fair value hierarchy for the year ended December 31, 2024:
| 2024 | |||||
Preferred Stock warrant liability | |||||
Balance January 1, 2024 | $ | ||||
Change in fair value | |||||
Balance December 31, 2024 | $ | ||||
The stock warrants were valued under the option pricing model, which considers the estimated volatility of the Company’s common stock at the date of measurement based on selected metrics of applicable volatility calculations from guideline public companies. The remeasurement of the convertible preferred stock warrant liability resulted in $256 and $119 recognized as other (income) expense, net for the years ended December 31, 2025 and 2024, respectively.
The fair value of the warrants were estimated as of December 31, 2025 and 2024 using an option pricing model with the following weighted average assumptions:
| December 31 | |||||
| Description | 2024 | ||||
| Weighted average volatility | % | ||||
| Weighted average risk-free rate | % | ||||
| Expected dividend yield | % | ||||
The Series E purchase option was valued under the option pricing model, which considers the estimated volatility of the Company’s equity at the date of measurement based on selected metrics of applicable volatility calculations from guideline public companies. The remeasurement of the Series E purchase option resulted in $11,719 recognized as an increase in fair value of Series E purchase option for the year ended December 31, 2025.
The convertible notes were valued using the fair value election per ASC 825 Financial Instruments. The convertible notes were valued using the expected stock price upon IPO and the probability of the IPO occurring. The Company weighted an IPO scenario at 97 % and a liquidation event at 3 % and an expected stock price within the floor and cap described in Note 9. The initial fair value measurement at issuance of the convertible notes was $49,815 and resulted in $9,952 recognized as a day one loss which is included in change in fair value of convertible notes, net for the year ended December 31, 2025 in the Company’s Statements of Operations and Comprehensive Loss. At completion of the Company’s IPO the convertible notes and accrued interest converted into 2,810,428 shares of common stock. The Company recognized a $7,739 gain upon settlement of the convertible notes which is recognized in change in fair value of convertible notes, net for the year ended December 31, 2025 in the Company’s Statements of Operations and Comprehensive Loss.
123
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
3. Accrued Liabilities and Other Current Liabilities
Accrued liabilities and other current liabilities consist of the following:
December 31, | ||||||||||||||
| 2025 | 2024 | |||||||||||||
| Commissions payable | $ | $ | ||||||||||||
Accrued legal fees | ||||||||||||||
| Accrued payroll | ||||||||||||||
| Accrued royalties | ||||||||||||||
| Other | ||||||||||||||
| Accrued liabilities and other liabilities | $ | $ | ||||||||||||
4. Property and Equipment, Net
Net property and equipment, net consist of the following:
December 31, | ||||||||||||||
| 2025 | 2024 | |||||||||||||
| Instruments | $ | $ | ||||||||||||
| Machinery and equipment | ||||||||||||||
| Computer equipment | ||||||||||||||
| Furniture and fixtures | ||||||||||||||
| Leasehold improvements | ||||||||||||||
| Total property and equipment | ||||||||||||||
| Less: Accumulated depreciation | ( | ( | ||||||||||||
| Property and Equipment, Net | $ | $ | ||||||||||||
Depreciation expense for the years ended December 31, 2025 and 2024 totaled $2,908 and $1,896 , respectively, and is included in selling, general, and administrative expense in the statements of operations and comprehensive loss.
5. Debt
On July 18, 2025, the Company issued $40,000 aggregate principal amount of convertible notes. The convertible notes were to mature on September 1, 2028 and accrue interest at a rate of 5.0 % per annum through June 30, 2026, at which time the interest rate was to increase to 10.0 % per annum. Upon the consummation of the IPO, the convertible notes automatically converted into a number of shares of common stock equal to the outstanding principal amount of the notes and any accrued interest divided by the applicable conversion price. The conversion price was the lower of (i) a 20.0 % discount to the IPO price and (ii) $280,000 divided by the number of fully diluted shares of capital stock (on an as-converted basis) outstanding immediately prior to the IPO, but excluding the convertible notes (the “IPO Capitalization”); provided that, in no event shall such conversion price be less than the quotient obtained by dividing $210,000 by the IPO Capitalization. Upon a liquidation event, the holders of the convertible notes would have received a cash payment equal to the sum of 1.3 times the outstanding principal amount of the convertible notes plus any accrued interest. The Company recognized a $9,952 loss related to the issuance of the convertible notes. Refer to Note 2, Fair Value Measurements, for discussion regarding the valuation of the convertible notes.
On August 1, 2025 the convertible notes and accrued interest converted into 2,810,428 shares of common stock. The Company recognized a $7,739 gain upon the conversion of the convertible notes.
On August 7, 2023, the Company entered into a loan and security agreement (the “Trinity Loan Agreement”) with Trinity Capital Inc. as lender, administrative agent, and collateral agent.
124
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
The Trinity Loan Agreement provides a term loan commitment of $45,000 in three potential tranches: (i) a $15,000 term loan facility funded on August 7, 2023, (ii) a $15,000 term loan facility available at the Company’s request on or before December 31, 2024, if the Company achieves certain revenue milestones, and (iii) a $15,000 term loan facility available at the Company’s request on or before December 31, 2025, if the Company achieves certain additional revenue milestones. All three of these term loans have a maturity date of September 1, 2028. Borrowings under all three term loan facilities bear interest at a variable annual rate equal to the greater of (i) 11.50 % and (ii) the Prime Rate plus 3.50 %. The second tranche expired on December 31, 2025, prior to Company drawing on the tranche.
On July 21, 2025, the Company amended the Trinity Loan Agreement to extend the commitment date of the second and third tranches as follows: (i) a $15,000 draw from before December 31, 2024 to before December 31, 2025 and to extend the commitment date of the tranche (ii) a $15,000 draw from before December 31, 2025 to before December 31, 2026. The interest rate on the instrument was amended from a floor of 11.5 % to 11.0 %, along with increasing the interest only period from an initial term of 48-months to 60-months . Additionally, warrants originally vesting upon the funding of the third tranche draw were cancelled and are no longer outstanding.
In connection with the closing of the Trinity Loan Agreement the Company issued a warrant to the Lender to purchase shares of the Company’s Series D convertible preferred stock (“Trinity Warrant”). The Trinity Warrant has an exercise price of $10.33 per share and expires 10 years from the issue date of the Trinity Loan Agreement; refer to Note 9. Upon the advance of the tranche A loan, 43,578 shares vested, and an additional 43,578 shares that would have vested upon the advance of the tranche B loan expired on December 31, 2025.
The Trinity Loan Agreement provides that the Company can at any time prepay the term loans, in whole or in part, subject to a prepayment premium equal to: (a) 2.50 % of the then-outstanding principal amount of the term loans, if such prepayment occurs on or prior to the first anniversary of the Trinity Loan Agreement; (b) 1.50 % of the then-outstanding principal amount of the advance, if such prepayment occurs after the first anniversary of the Trinity Loan Agreement and on or prior to the second anniversary of the Trinity Loan Agreement; and (c) 1.00 % of the then-outstanding principal amount of the advance, if such prepayment occurs after the second anniversary of the Trinity Loan Agreement and prior to the Maturity Date. We are required to make an end of term payment equal to 3.00 % of the aggregate principal amount of the term loans funded on the earlier of (i) the Maturity Date, (ii) the date that the Company prepays all of the outstanding principal in full or (iii) the date of acceleration of the balance of the outstanding term loans by the Agent. The term loans are secured by substantially all our assets, including intellectual property.
Proceeds related to the Trinity Loan Agreement were $14,850 , net of issuance costs of $250 and return of deposit of $100 . As of December 31, 2025, the Company recorded long-term debt related to the Trinity Loan Agreement of $14,911 , which includes a principal amount of $15,000 and unamortized discount, debt issuance costs and deferred interest of $571 , based on an imputed interest rate of 12.33 %. As of December 31, 2024, the Company recorded long-term debt related to the Trinity Loan Agreement of $14,658 , which included principal amount of $15,000 and unamortized debt discount, issuance cost and deferred interest of $623 , based on an imputed interest rate of 14.00 %. Interest expense for the years ended December 31, 2025 and 2024 totaled $2,018 and $2,086 , respectively.
6. Leases
Month-to-Month Lease
The Company leases a facility in Michigan from a related party. The lease is on a month-to-month basis. Total rent expense under this agreement for the years ended December 31, 2025 and 2024, was $10 and $33 , respectively. There are no future minimum lease payments associated with this lease. The Company has elected to apply the short-term lease exception guidance and therefore has not recorded a right-of-use asset or liability related to this lease.
Lease Obligations
The Company leases office and distribution space in Michigan under a five-year lease agreement, which commenced in July 2021. The Company also leases a distribution facility in California under a three-year lease agreement, which commenced in March 2025. The Company has determined these lease agreements qualify as operating lease arrangements. The Company’s remaining operating lease term is approximately 1.8 years at December 31, 2025.
125
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
As the lease contract does not provide an implicit discount rate, the Company used 10.25 % representing its incremental borrowing rate based on information available at the commencement date, to determine the present value of lease payments. The incremental borrowing rate is based on a borrowing with a term similar to that of the associated lease. The Company does not include renewal, termination, or purchase options that are not reasonably certain of exercise when determining the term of the borrowing.
At December 31, 2025, the right-of-use asset and lease liability relating to operating leases was $110 and $113 , respectively. Operating lease expense and variable lease expense for the year ended December 31, 2025 were $69 and $74 , respectively. Operating lease expense and variable lease expense for the year ended December 31, 2024 were $48 and $17 , respectively.
At December 31, 2025, the commitment for operation lease liabilities for future annual periods was as follows:
Year Ending December 31, | |||||
| 2026 | $ | ||||
| 2027 | |||||
2028 | |||||
| Total Lease Payments | |||||
| Less imputed interest | |||||
| Present Value of Lease Liabilities | $ | ||||
7. Income Taxes
Provision Expense for Income Taxes
The Company had no current or deferred tax expense recorded as of December 31, 2025 and 2024.
Income tax expense differs from the statutory rate due primarily to the impact of the valuation allowance against the Company’s deferred tax assets.
Deferred Income Tax Assets and Liabilities
The components of deferred income tax assets and liabilities are as follows:
December 31, | ||||||||||||||
| 2025 | 2024 | |||||||||||||
| Deferred Tax Assets | ||||||||||||||
| Net operating loss carryforwards | $ | $ | ||||||||||||
| Tax credits and other carryforwards | ||||||||||||||
| Section 174 expenditures | ||||||||||||||
Warrant and Series E liabilities | ||||||||||||||
| Accrued liabilities and other items | ||||||||||||||
| Deferred Tax Assets | ||||||||||||||
| Deferred Tax Liability | ||||||||||||||
| Depreciation and amortization | ( | ( | ||||||||||||
| Deferred Tax Liability | ( | ( | ||||||||||||
| Deferred Tax Assets, Net | ||||||||||||||
| Valuation Allowance | ( | ( | ||||||||||||
| Net Deferred Tax Asset | $ | $ | ||||||||||||
A reconciliation of the Company’s provision for income taxes at the federal statutory rate of 21% to the reported income tax provision for the years ended December 31, 2025 and 2024 is as follows:
126
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
December 31, | ||||||||||||||
| 2025 | 2024 | |||||||||||||
| Computed “expected” tax benefit | $ | ( | $ | ( | ||||||||||
| Permanent differences | ||||||||||||||
| General Business tax credits | ( | |||||||||||||
| Other | ( | |||||||||||||
| Change in valuation allowance | ||||||||||||||
| Income Tax expense (benefit) | $ | $ | ||||||||||||
The ultimate realization of deferred tax assets is dependent upon the generation of future taxable income during the periods in which those temporary differences become deductible. Management considers the scheduled reversal of deferred tax liabilities (including the impact of available carryback and carryforward periods), projected future taxable income, and tax-planning strategies in making this assessment. The Company has determined for the years ended December 31, 2025 and 2024, based on all available evidence analyzed on a more likely than not basis, a valuation allowance should be established on all of the net deferred tax asset in its U.S. jurisdiction since there is not enough positive evidence to support the more likely than not position for the future realization in the U.S. jurisdiction.
On December 22, 2017, the U.S. government enacted comprehensive tax legislation commonly referred to as the Tax Cuts and Jobs Act (the Tax Act). The Tax Act allows for the indefinite carryforward of net operating losses (“NOLs”) arising in a taxable year ending after December 31, 2017, which would be considered an indefinite lived asset. The amendments limit the future usage of such NOLs to 80% of taxable income in a single year and disallow the carryback of NOLs to prior years with taxable income.
As of December 31, 2025 and 2024, the Company has federal net operating loss carryforwards of $87,390 and $45,295 , respectively. The Company has federal tax credit carryforwards of $691 and $464 for the years ended December 31, 2025 and 2024, respectively. The NOLs and tax credit carryforwards recorded are subject to limitations under Section 382 and 383. However, the Section 382 limitation did not limit the usage of the net operating loss carryforwards in 2025 or 2024 for federal tax purposes. In addition, the Company’s ability to utilize the current NOL carryforwards might be further limited by future equity issuances. The federal net operating loss carryforwards originated after 2016 and have an indefinite life which may be used to offset 80% of a future year’s taxable income. The Company has various state net operating loss carryforwards with various carryforward periods that will begin to expire in 2034.
The Company operates in various states within the United States and files income tax returns in these various jurisdictions and has generated various net operating loss carryforwards in these jurisdictions to date. The Company does not believe a material uncertain tax position exists as of December 31, 2025 and 2024. Based on the Company’s assessment of many factors, including past experience and complex judgments about future events, the Company does not currently anticipate significant changes in its uncertain tax positions over the next 12 months. In connection with the adoption of the referenced provision, the Company recognizes interest and penalties accrued related to unrecognized tax benefits in income tax expense. As of December 31, 2025 and 2024, the Company had no
The Company’s federal and state tax returns are open for review going back to the 2022 tax year.
8. Related Party Transactions
On October 22, 2020, the Company entered into a software license agreement with Genesis Software which was amended and restated on January 1, 2023. Robert Ball, the Company’s Chief Executive Officer and Executive Chairman, is a co-founder and director of Genesis Software. Mr. Ball and Matthew Ahearn, the Company’s Chief Operating Officer and a director, are directors of Genesis Investment Holdings, LLC, which has an ownership interest in Genesis Software. In addition, cultivate(MD) Capital Accelerator Fund L.P. and Genesis Investment Holdings, LLC are investors in Genesis Software. The software licensing agreement is a 5 year term. The agreement required an upfront payment of $1,000 , an incremental $500 payment when FDA clearance was obtained, and quarterly payments of royalties equal to 4 % of the net sales price of each licensed product sold, until such time we have paid Genesis Software an aggregate of $7,000 under the software license agreement. For each of the years ended December 31, 2025 and 2024, the Company paid Genesis
127
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
Software $1,641 and $1,169 , respectively, pursuant to the license agreement. Amounts owed under the agreement of $575 and $344 are included in accrued liabilities on the Balance Sheets at December 31, 2025, and 2024, respectively.
For the years ended December 31, 2025 and 2024, the Company paid $1,997 and $1,925 , respectively, for software development to Genesis Software. Amounts payable of $863 and $276 are included in accounts payable on the Balance Sheets at December 31, 2025, and 2024, respectively.
The Company has entered into a consulting agreement with an entity under common ownership. The consulting agreement is currently on a year-to-year basis. The agreement requires compensation for services performed. If services performed are on an hourly basis, the Company shall be responsible to pay for hours actually worked by the consultant’s employees. The Company will reimburse the consultant for all reasonable expenses incurred in connection with performing services for the Company. For the years ended December 31, 2025 and 2024, the Company paid $4,821 and $3,589 , respectively. Amounts payable of $357 and $537 are included in accounts payable on the balance sheets at December 31, 2025, and 2024, respectively.
During the first quarter of 2024, Robert Ball, the Company’s Chief Executive Officer and Executive Chairman, was an investor in Revelation Medical Devices (“RMD”), which manufactures surgical instruments used during procedures involving our systems. After the first quarter of 2024, Mr. Ball is no longer an investor in RMD. For the year ended December 31, 2024, the Company paid RMD $859 .
9. Convertible Preferred Stock
On March 6, 2025, the Company entered into a Series E Preferred Stock Purchase Agreement pursuant to the Series E Preferred Stock Financing, whereby it received a total commitment amount of $40,130 for the issuance and sale of 58,774,332 shares of Series E Preferred Stock pursuant to two separate closing tranches, the first of which closed on March 6, 2025 whereby the Company issued 29,455,169 shares of Series E preferred stock resulting in total gross proceeds to the Company of $20,111 . The second tranche could have occurred any time prior to September 15, 2025, for the issuance and sale of 29,319,143 shares of Series E Preferred Stock for an aggregate purchase price of $20,019 . The Company determined that the second tranche constitutes a free standing financial instrument and allocated $218 of the Series E proceeds to the purchase option.
In June 2025 the Company called the second tranche of its Series E Preferred Stock for the issuance of 29,319,143 shares of Series E Preferred Stock resulting in gross proceeds to the Company of $20,019 . The Series E purchase option was revalued as of the date the second tranche was called. The Company recognized the change in value of the Series E purchase option in the change in fair value of Series E purchase option caption in its Statements of Operations and Comprehensive Loss. The fair value of the Series E purchase option was recognized as Series E convertible preferred stock upon the exercise of the Series E purchase option. See Note 2 for additional details.
On March 4, 2025, Genesis Investment Holdings, LLC exercised 988,999 series Seed warrants resulting in proceeds of $83 .
For the year ended December 31, 2025, 1,000,000 Series B warrants were exercised, resulting in proceeds of $372 .
Immediately prior to the closing of the Company’s IPO on August 1, 2025, all shares of the Company’s redeemable convertible preferred stock converted into shares of the Company’s common stock.
For all series of preferred stock, the initial conversion price equals their original issuance price per share, at December 31, 2024, adjusted for the Reverse Stock Split as described in Note 1.
A summary of preferred stock is as follows:
128
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
| Series Seed Convertible Preferred Stock | Series A Convertible Preferred Stock | Series B Convertible Preferred Stock | Series C Convertible Preferred Stock | Series D Convertible Preferred Stock | Series E Convertible Preferred Stock | Total | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Shares | Amount | Shares | Amount | Shares | Amount | Shares | Amount | Shares | Amount | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Balance at December 31, 2023 | $ | $ | $ | $ | $ | — | $ | — | $ | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Balance at December 31, 2024 | $ | $ | $ | $ | $ | — | $ | — | $ | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Balance at December 31, 2024 | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Exercise of preferred stock warrants | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Issuance of Series E | — | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Conversion of redeemable convertible preferred stock to common stock upon IPO | ( | ( | ( | ( | ( | ( | ( | ( | ( | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Balance at December 31, 2025 | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Issued and Outstanding Preferred Shares
As of December 31 2024, the total authorized, issued, and outstanding shares of Preferred Stock were as follows (in thousands, except par value, share and per share amounts):
| Series | Par Value | Authorized | Issued and Outstanding | Issuance Price Per Share | Liquidation Value | |||||||||||||||||||||||||||
| Seed | $ | $ | $ | |||||||||||||||||||||||||||||
| A | ||||||||||||||||||||||||||||||||
| B | ||||||||||||||||||||||||||||||||
| C | ||||||||||||||||||||||||||||||||
| D | ||||||||||||||||||||||||||||||||
| Total | $ | |||||||||||||||||||||||||||||||
Preferred Stock Provisions
Conversion of Convertible Preferred Stock to Equity
Preferred Stock contains a provision at the option of the holder, at any time and from time to time, and without the payment of additional consideration by the holder thereof, to be converted into common stock. The number of shares of common stock to which a holder of Preferred Stock shall be entitled upon conversion shall be the product obtained by multiplying the applicable conversion prices then in effect by the number of shares of Preferred Stock being converted. The conversion price of Preferred Stock shall be subject to adjustment in the event of any stock split, stock dividend, combination, subdivision, recapitalization, or the like. All Preferred Stock shall be automatically converted into common stock at their then respective conversion prices: (i) in the event of an underwritten public offering of shares of the common stock at an aggregate offering price (prior to underwriting discounts, commissions, and expenses) of at least $50,000 or (ii) the date upon which the Company obtains the vote or consent of the preferred majority and the vote or consent of the holders of at least 65 % of the Series D Preferred Stock. Convertible preferred stock is presented as temporary equity in the mezzanine section of the balance sheets, as conversion of the preferred stock is not within the control of the Company. The purpose of this classification is to convey that such a security may not be permanently a part of equity and could result in a demand for cash, securities, or other assets of the entity in the future. All series of preferred stock conversion price equals their original issuance price per share at December 31, 2024.
Dividends
The Company’s Preferred Stock Series Seed, A, B, C and D pay non-cumulative dividends, at the rate of 8 % per annum, whenever funds are legally available and when, as and if declared by the Company’s Board of Directors (“Board”) prior and in preference to dividends on any other equity of the Company. Dividends, when, as, and if declared, will first be
129
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
paid in full to the holders of Series D Preferred Stock, then Series C Preferred Stock, then Series B Preferred Stock, then Series A Preferred Stock, then Series Seed Preferred Stock, then to holders of common stock. In the event that any dividends are declared to the then-outstanding shares of common stock (other than dividends payable in common stock for which adjustment to the conversion price of the Preferred Stock is made) an additional dividend shall be declared with respect to the then-outstanding shares of Preferred Stock, in an amount equal to the amount of dividends per share that would have been payable on the number of shares of the common stock that the Preferred Stock would have been converted into on an as-converted basis. No
Voting Rights
The holders of Preferred Stock shall be entitled to the number of votes equal to the whole number of shares of the common stock into which such shares of the Preferred Stock are then converted. Additionally, separate vote of all series of preferred stock requires, in addition to any other vote or consent, the vote or consent of at least a majority of each series of preferred stock then-outstanding votes for certain significant actions. Further, as long as at least 20 % of the authorized shares of the Series C Preferred Stock or Series D Preferred Stock remain outstanding, certain significant actions require the vote or consent of at least 60 % of the then-outstanding Series C Preferred Stock or Series D Preferred Stock, respectively.
Liquidation Rights
In the event of a deemed liquidation event of the Company, voluntary or involuntary liquidation, dissolution or winding up of the Company, all holders of Preferred Stock are entitled to full payment under their liquidation preferences out of the assets of the Company legally available for distribution to its stockholders (or the consideration received in such transaction), before any payment shall be made to the holders of the common stock. After payment in full to the Series D liquidation preference, the remaining proceeds shall be pursuantly distributed to the holders of existing Series C, followed by Series B, Series A, and Series Seed Preferred Stock and common stock, in an amount equal to their original issue price plus any declared and unpaid dividends.
All remaining legally available assets of the Company that are not payable to the holders of shares of Preferred Stock shall be distributed among the holders of shares of common stock, pro rata based on the number of shares held by each such holder.
Anti-Dilution and Downround Protections
The conversion price is subject to adjustment for certain dilutive events, including certain types of (i) stock splits and combinations, (ii) common stock dividends and distributions, (iii) reclassification, exchange, and substitutions, (iv) reorganizations, mergers, or consolidations, or (v) sale of shares below conversion price.
Stock Warrants
In 2020, in connection with its issuance of Series B Preferred Stock, the Company issued 1,000,000 of Series B Stock Warrants. The Series B Preferred Stock issuable upon exercise of the Series B Stock Warrants is convertible into shares of common stock in the same manner as each respective underlying series of outstanding Series B Preferred Stock and will be entitled to the same dividend rights as each respective series.
In August 2023, in connection with the closing of the Trinity Loan Agreement, the Company issued a warrant to the Lender to purchase 2,494,457 shares of the Company’s Series D convertible preferred stock (“Trinity Warrant”). The Trinity Warrant has an exercise price of $0.5412 per share, and expires ten years from the issue date of the Trinity Loan Agreement. Upon the advance of the tranche A loan, 831,486 shares vested. The second term tranche was not used and the third term tranche was removed as part of the July 21, 2025 Trinity loan amendment. Immediately prior to the closing of the Company’s IPO on August 1, 2025, the Trinity Warrant converted into warrants to purchase shares of the Company’s common stock. Upon conversion to common stock warrants the Trinity Warrants were subject to final remeasurement and the outstanding fair value reclassified to equity. As of December 31, 2025, the Company had 43,578 common stock warrants outstanding with an exercise price of $10.33 that expire on August 7, 2033.
130
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
For the year ended December 31, 2025, warrant holders net settled 17,827 common stock warrants resulting in 14,714 common shares issued.
The following tables summarize the Company’s outstanding warrants at December 31, 2024:
| Description | Outstanding | Exercise Price per share | Expiration Date | Fair Value | ||||||||||||||||||||||
| Common | $ | April 1, 2031 | $ | |||||||||||||||||||||||
| Series Seed | April 24, 2025 | |||||||||||||||||||||||||
| Series B | January 28, 2030 | |||||||||||||||||||||||||
| Series D | August 7, 2033 | |||||||||||||||||||||||||
| Total | $ | |||||||||||||||||||||||||
10. Stock Compensation
Common Stock
The Company increased the authorized capital from 204,958,600 to 212,366,763 212,366,763 730,000,000 shares of common stock in 2025.
Stock Options
In 2017, the Company adopted a stock compensation plan (the “Plan”) pursuant to which the Company’s Board of Directors may grant stock options or nonvested shares to its full-time employees, directors, and advisors to purchase common stock of the Company. The Plan was amended in 2024 to authorize 1,669,550 shares issuable. The Plan was subsequently amended in 2025 to authorize grants to purchase up to 2,185,560 shares. Stock options can be granted with an exercise price equal to or greater than the stock’s fair value at the date of grant. All awards have 10-year terms and vest based on terms defined in each individual grant agreement, and the expense is recognized on a straight-line basis over the vesting period, which is generally the service period.
In 2025, the Company adopted a stock compensation plan (“2025 Incentive Plan”) pursuant to which the Company’s Board of Directors may grant stock options or nonvested shares to its full-time employees, directors, and advisors to purchase common stock of the Company. The 2025 Incentive Plan authorizes 2,493,739 shares issuable, with a 5 % annual refresh of the aggregate number of shares of common stock outstanding on the final day of the immediately preceding calendar year. Stock options can be granted with an exercise price equal to or greater than the stock’s fair value at the date of grant. All awards have 10-year terms and vest based on terms defined in each individual grant agreement, and the expense is recognized on a straight-line basis over the vesting period, which is generally the service period.
Because the Company’s shares only recently started trading on a public market and shares were rarely traded privately, expected volatility is estimated based on the average historical volatility of similar entities with publicly traded shares. The risk-free rate for the expected term of the option is based on the U.S. Treasury yield curve at the date of grant.
The Company uses a simplified method to determine the expected term for the valuation of employee options. This method effectively assumes that exercise occurs over the period from vesting until expiration, and therefore the expected term is the midpoint between the service period and the contractual term of the award. The simplified method is applicable to options with service conditions.
For the options granted in 2025, which were all issued prior to the completion of the Company’s IPO, the Company utilized a third-party valuation firm to assist in the estimation of the fair value of the underlying shares of the Company utilizing option pricing models (“OPM”). Under the OPM, shares are valued by creating a series of call options with exercise prices based on the liquidation preferences and conversion terms of each equity class. The estimated fair values of the convertible preferred stock and common stock are inferred by analyzing these options. This method is appropriate to use when the range of possible future outcomes is so difficult to predict that estimates would be highly speculative, and dissolution or liquidation is not imminent. The Company accounts for forfeitures as they occur. Shares of the Company’s
131
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
common stock granted under the stock option plan in the form of stock options are counted against the share reserve on a one for one basis.
The following tables summarize the Company’s outstanding stock options:
| Options | Weighted Average Exercise Price | Weighted Average Remaining Contractual Life (in years) | |||||||||||||||
| Outstanding as of December 31, 2023 | $ | ||||||||||||||||
| Granted | $ | — | |||||||||||||||
| Exercised | ( | — | |||||||||||||||
| Forfeited | ( | — | |||||||||||||||
| Options Outstanding December 31, 2024 | $ | ||||||||||||||||
| Granted | $ | — | |||||||||||||||
| Exercised | ( | — | |||||||||||||||
| Forfeited | ( | — | |||||||||||||||
| Options Outstanding December 31, 2025 | $ | ||||||||||||||||
| Options Exercisable, December 31, 2025 | $ | ||||||||||||||||
The aggregate intrinsic value of stock options is calculated as the pre-tax difference between the weighted-average exercise price of the stock options and the valuation of the Company’s common shares of $11.78 and $2.86 as of December 31, 2025 and 2024, respectively. The calculation excludes any stock options with an exercise price higher than the valuation of shares of the Company’s common stock, if any. The stock options were valued under an option pricing model, which considers the estimated volatility of the Company’s common stock at the date of measurement based on selected metrics of applicable volatility calculations from guideline public companies. Stock compensation expense related to the stock options issued totaled $996 in 2025, and $754 in 2024 and is recognized as selling, general, and administrative expenses in the statements of operations and comprehensive loss. The total intrinsic value of options exercised was $3,187 and $15 for the years ended December 31, 2025 and 2024, respectively.
As of December 31, 2025, the Company had unrecognized stock-based compensation of $1,627 that is to be recognized over a weighted average period of 2.52 years.
The fair value of the stock options granted were estimated as of the grant date using an option pricing model with the following weighted average assumptions:
| 2025 | 2024 | ||||||||||
| Weighted average volatility | |||||||||||
| Weighted average risk-free rate | |||||||||||
| Expected dividend yield | |||||||||||
| Expected term (in years) | |||||||||||
11. Loss Per Share
Basic loss per share is computed by dividing the net loss after tax attributable to common stockholders by the weighted average shares outstanding during the period. Diluted loss per share is computed by including potentially dilutive securities outstanding during the period in the calculation of weighted average shares outstanding. The Company did not have any dilutive securities during the periods presented; therefore, diluted loss per share is equal to basic loss per share.
Presented in the table below is a reconciliation of the numerator and denominator for the basic and diluted loss per share calculations for the years ended December 31:
132
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
December 31, | |||||||||||
| 2025 | 2024 | ||||||||||
| Net loss | $ | ( | $ | ( | |||||||
| Basic and diluted weighted average shares outstanding | |||||||||||
| Net loss attributable to common shareholders, basic and diluted | $ | ( | $ | ( | |||||||
The following securities were not included in the computation of diluted shares outstanding because the effect would be anti-dilutive:
December 31, | |||||||||||
| 2025 | 2024 | ||||||||||
| Series Seed | |||||||||||
| Series A | |||||||||||
| Series B | |||||||||||
| Series C | |||||||||||
| Series D | |||||||||||
| Common Options | |||||||||||
| Common Warrants | |||||||||||
| Series Seed Warrants | |||||||||||
| Series B Warrants | |||||||||||
| Series D Warrants | |||||||||||
| Total | |||||||||||
12. Segment Information
The Company reports segment information based on how the Company’s chief operating decision maker (“CODM”), who is the Chief Executive Officer, regularly reviews operating results, allocates resources and makes decisions regarding business operations. The Company’s business structure is comprised of one
All revenue for the years ended December 31, 2025 and 2024 was generated from customers located in the United States. No customers represent 10% or more of the Company’s net revenue for the years ended December 31, 2025 and 2024. The measure of segment assets is reported on the Balance Sheets as total assets.
The table below is a summary of the segment net loss, including significant segment expenses (in thousands):
133
Shoulder Innovations, Inc.
Notes to the Financial Statements
(in thousands, except share, per share data and percentages)
December 31, | ||||||||||||||
| 2025 | 2024 | |||||||||||||
| Total Revenue | $ | $ | ||||||||||||
| Cost of goods Sold | ||||||||||||||
| Gross margin | ||||||||||||||
| Operating expenses: | ||||||||||||||
| General and administrative | ||||||||||||||
| Sales and marketing | ||||||||||||||
| Medical education | ||||||||||||||
| Research and development | ||||||||||||||
| Interest/other expense | ||||||||||||||
| Segment net loss and total net loss | $ | ( | ( | |||||||||||
Depreciation expense for the years ended December 31, 2025 and 2024 totaled $2,908 and $1,896 , respectively. Depreciation expense of $2,824 and $1,842 for instruments is included in sales and marketing and depreciation expense of $84 and $54 for computer equipment, furniture and fixtures, and leasehold improvements is included in general, and administrative expenses in the statements of operations and comprehensive loss for the years ended December 31, 2025 and 2024, respectively.
Amortization expense related to the Company’s software license for the years ended December 31, 2025 and 2024 totaled $300 and $300 , respectively, and is included in general and administrative expense.
13. Commitments and Contingencies
Litigation
From time to time, the Company may be a party to legal proceedings that arise in the ordinary course of business, some of which may be covered by insurance. The Company establishes an accrual for legal proceedings if and when those matters reach a stage where they present loss contingencies that are both probable and reasonably estimable. In such cases, there may be a possible exposure to loss in excess of any amounts accrued. The Company monitors those matters for developments that would affect the likelihood of a loss and the accrued amount, if any, thereof, and adjust the amount as appropriate. If the loss contingency at issue is not both probable and reasonably estimable, the Company does not establish an accrual, but it will continue to monitor the matter for developments that could make the loss contingency both probable and reasonably estimable. If there is at least a reasonable possibility that a material loss will occur, the Company will provide disclosure regarding the contingency. Except as disclosed below, management believes that the Company does not have any pending legal proceedings that, separately or in the aggregate, would have a material adverse effect on the Company’s results of operations, financial condition or cash flows.
On February 28, 2024, the Company filed a complaint against Catalyst Orthoscience Inc. (“Catalyst”) in United States District Court for the District of Delaware claiming patent infringement through Catalyst’s making, using, selling, offering for sale in the United States, and/or importing into the United States, reverse shoulder systems. In response to the Company’s lawsuit, on March 20, 2024, Catalyst filed a counterclaim in United States District Court for the District of Delaware claiming patent infringement by certain of the Company’s products. The Company is seeking an injunction, monetary damages, interest, fees and other costs. The Company believes that it has substantial and meritorious defenses to Catalyst’s claims and intends to vigorously defend its position, including through the trial and appellate stages if necessary. As the Company’s lawsuit and Catalyst’s counterclaim is ongoing, the Company is unable to determine the likelihood of an outcome or estimate a range of reasonably possible losses, if any. Accordingly, the Company has not made an accrual for any possible loss. The outcome of any litigation, however, is inherently uncertain, and an adverse judgment or settlement in the counterclaim proceeding, if any, could have a material and adverse effect the Company’s business, financial position, results of operations or cash flows.
134
Item 9. Changes in and Disagreements With Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Limitations on Effectiveness of Controls and Procedures
In designing and evaluating our disclosure controls and procedures, management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives. In addition, the design of disclosure controls and procedures must reflect the fact that there are resource constraints, and that management is required to apply judgment in evaluating the benefits of possible controls and procedures relative to their costs. Further, because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that misstatements related to error or fraud will not occur or that all control issues and instances of fraud, if any, have been detected.
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our principal executive officer and principal financial officer, evaluated, as of the end of the period covered by this Annual Report, the effectiveness of our disclosure controls and procedures (as defined in Rules 13a‑15(e) and 15d‑15(e) under the Exchange Act). Based on that evaluation, our principal executive officer and principal financial officer concluded that, as of December 31, 2025, our disclosure controls and procedures were effective at the reasonable assurance level.
Changes in Internal Control over Financial Reporting
There were no changes in our internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) during the three months ended December 31, 2025 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Management’s annual report on internal control over financial reporting
This Annual Report does not include a report of management’s assessment regarding internal control over financial reporting or an attestation report of the company’s registered public accounting firm due to a transition period established by rules of the SEC for newly public companies.
In addition, because we are an “emerging growth company” under the JOBS Act, our independent registered public accounting firm will not be required to attest to the effectiveness of our internal control over financial reporting for so long as we are an emerging growth company.
Item 9B. Other Information
Rule 10b5-1 Plans
During the three months ended December 31, 2025, no director or officer of the Company adopted terminated
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections
Not applicable.
135
Part III
Item 10. Directors, Executive Officers and Corporate Governance
The information required by this item will be included in our Proxy Statement for the 2026 Annual Meeting of Stockholders, to be filed with the SEC within 120 days of the year ended December 31, 2025, and is incorporated herein by reference.
Item 11. Executive Compensation
The information required by this item will be included in our Proxy Statement for the 2026 Annual Meeting of Stockholders, to be filed with the SEC within 120 days of the year ended December 31, 2025, and is incorporated herein by reference.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required by this item will be included in our Proxy Statement for the 2026 Annual Meeting of Stockholders, to be filed with the SEC within 120 days of the year ended December 31, 2025, and is incorporated herein by reference.
Item 13. Certain Relationships and Related Transactions, and Director Independence
The information required by this item will be included in our Proxy Statement for the 2026 Annual Meeting of Stockholders, to be filed with the SEC within 120 days of the year ended December 31, 2025, and is incorporated herein by reference.
Item 14. Principal Accountant Fees and Services
The information required by this item will be included in our Proxy Statement for the 2026 Annual Meeting of Stockholders, to be filed with the SEC within 120 days of the year ended December 31, 2025, and is incorporated herein by reference.
136
Part IV
Item 15. Exhibits and Financial Statement Schedules
(a) The following documents are filed as part of this report:
1.Financial Statements
See Index to Financial Statements under Part II, Item 8 of this Annual Report.
2.Financial Statement Schedules
Schedules not listed above have been omitted because they are not required, not applicable, or the required information is otherwise included.
3.Exhibits
The exhibits listed below are filed as part of this Annual Report or are incorporated herein by reference as indicated.
Incorporated by Reference | ||||||||||||||||||||||||||||||||
| Exhibit Number | Exhibit Description | Form | File No. | Exhibit | Filing Date | |||||||||||||||||||||||||||
3.1 | 8-K | 001-42771 | 3.1 | 8/4/2025 | ||||||||||||||||||||||||||||
3.2 | 8-K | 001-42771 | 3.2 | 8/4/2025 | ||||||||||||||||||||||||||||
4.1 | S 1/A | 333-288549 | 4.1 | 7/25/2025 | ||||||||||||||||||||||||||||
4.2* | ||||||||||||||||||||||||||||||||
4.3^ | S 1/A | 333-288549 | 4.2 | 7/25/2025 | ||||||||||||||||||||||||||||
10.1^ | S 1/A | 333-288549 | 10.1 | 7/25/2025 | ||||||||||||||||||||||||||||
10.2† | S 1/A | 333-288549 | 10.2 | 7/25/2025 | ||||||||||||||||||||||||||||
10.3† | S 1/A | 333-288549 | 10.3 | 7/25/2025 | ||||||||||||||||||||||||||||
10.4^† | S-1/A | 333-288549 | 10.4 | 7/25/2025 | ||||||||||||||||||||||||||||
10.5^ | Loan and Security Agreement, dated as of August 7, 2023, by and among the Registrant, the lenders party thereto and Trinity Capital, Inc., as amended by the First Amendment to the Loan and Security Agreement, dated as of April 23, 2024 and the Second Amendment to the Loan and Security Loan Agreement dated as of July 21, 2025. | S-1/A | 333-288549 | 10.5 | 7/25/2025 | |||||||||||||||||||||||||||
10.6 | S-1/A | 333-288549 | 10.6 | 7/25/2025 | ||||||||||||||||||||||||||||
| 10.7† | S-1/A | 333-288549 | 10.7 | 7/25/2025 | ||||||||||||||||||||||||||||
137
10.8^ | S-1/A | 333-288549 | 10.8 | 7/25/2025 | ||||||||||||||||||||||||||||
10.9# | S-8 | 333-289184 | 99.1 | 8/1/2025 | ||||||||||||||||||||||||||||
10.9(a)# | S-8 | 333-289184 | 99.2 | 8/1/2025 | ||||||||||||||||||||||||||||
10.10# | S-1/A | 333-288549 | 10.10 | 7/25/2025 | ||||||||||||||||||||||||||||
10.11(a)# | S-8 | 333-289184 | 99.3 | 8/1/2025 | ||||||||||||||||||||||||||||
10.11(b)# | S-8 | 333-289184 | 99.4 | 8/1/2025 | ||||||||||||||||||||||||||||
10.11(c)# | S-8 | 333-289184 | 99.5 | 8/1/2025 | ||||||||||||||||||||||||||||
10.12# | S-8 | 333-289184 | 99.6 | 8/1/2025 | ||||||||||||||||||||||||||||
10.13# | S-1/A | 333-288549 | 10.13 | 7/25/2025 | ||||||||||||||||||||||||||||
10.14# | S-1/A | 333-288549 | 10.14 | 7/25/2025 | ||||||||||||||||||||||||||||
10.15# | S-1/A | 333-288549 | 10.15 | 7/25/2025 | ||||||||||||||||||||||||||||
10.16# | S-1/A | 333-288549 | 10.16 | 7/25/2025 | ||||||||||||||||||||||||||||
10.17# | S-1/A | 333-288549 | 10.17 | 7/25/2025 | ||||||||||||||||||||||||||||
19.1* | ||||||||||||||||||||||||||||||||
21* | ||||||||||||||||||||||||||||||||
23.1* | ||||||||||||||||||||||||||||||||
24.1* | ||||||||||||||||||||||||||||||||
31.1* | ||||||||||||||||||||||||||||||||
31.2* | ||||||||||||||||||||||||||||||||
32.1+ | ||||||||||||||||||||||||||||||||
32.2+ | ||||||||||||||||||||||||||||||||
97.1* | ||||||||||||||||||||||||||||||||
101.INS* | Inline XBRL Instance Document. | |||||||||||||||||||||||||||||||
101.SCH* | Inline XBRL Taxonomy Extension Schema Document. | |||||||||||||||||||||||||||||||
101.CAL* | Inline XBRL Taxonomy Extension Calculation Linkbase Document. | |||||||||||||||||||||||||||||||
101.DEF* | Inline XBRL Taxonomy Extension Definition Linkbase Document. | |||||||||||||||||||||||||||||||
138
101.LAB* | Inline XBRL Taxonomy Extension Label Linkbase Document. | |||||||||||||||||||||||||||||||
101.PRE* | Inline XBRL Taxonomy Extension Presentation Linkbase Document. | |||||||||||||||||||||||||||||||
104* | Cover Page Interactive Data File (embedded within the Inline XBRL document). | |||||||||||||||||||||||||||||||
__________________
* Filed herewith.
# Indicates management contract or compensatory plan.
^ Pursuant to Item 601(a)(5) of Regulation S-K, the Registrant has omitted schedules (or similar attachments) to this exhibit. The Registrant agrees to furnish supplementally a copy of the omitted schedules (or similar attachments) to the SEC upon request.
† Pursuant to Item 601(b)(10)(iv) of Regulation S-K, the Registrant has redacted portions of this exhibit (indicated by [***]) because the Registrant has determined that such redacted information is not material and is the type that the Registrant treats as private or confidential. The Registrant agrees to furnish supplementally an unredacted copy of this exhibit to the SEC upon request.
+ The certifications attached as Exhibits 32.1 and 32.2 that accompany this Annual Report are deemed furnished and not filed with the SEC and are not to be incorporated by reference into any filing of the Registrant under the Securities Act or the Exchange Act, whether made before or after the date of this Annual Report, irrespective of any general incorporation language contained in such filing.
Item 16. Form 10-K Summary
None.
139
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
SHOULDER INNOVATIONS, INC. | |||||||||||
| Date: | March 10, 2026 | By: | /s/ Robert Ball | ||||||||
Robert Ball | |||||||||||
Chief Executive Officer and Executive Chairman | |||||||||||
(Principal Executive Officer) | |||||||||||
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Robert Ball and Jeffrey Points, and each of them, as his true and lawful attorneys-in-fact, proxies and agents, each with full power of substitution and resubstitution, for him and in his name, place and stead, in any and all capacities, to sign any and all amendments to this report and to file the same, with any exhibits thereto and other documents in connection therewith, with the SEC, granting unto such attorneys-in-fact, proxies and agents full power and authority to do and perform each and every act and thing requisite and necessary to be done in and about the premises, as fully to all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact, proxies and agents, or their or his substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, this Annual Report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
Signature | Title | Date | ||||||||||||
/s/ Robert Ball | Chief Executive Officer and Executive Chairman (Principal Executive Officer) | March 10, 2026 | ||||||||||||
| Robert Ball | ||||||||||||||
/s/ Jeffrey Points | Chief Financial Officer (Principal Financial and Accounting Officer) | March 10, 2026 | ||||||||||||
Jeffrey Points | ||||||||||||||
/s/ Matthew Ahearn | Director | March 10, 2026 | ||||||||||||
| Matthew Ahearn | ||||||||||||||
/s/ Richard J. Buchholz | Director | March 10, 2026 | ||||||||||||
Richard J. Buchholz | ||||||||||||||
/s/ Paul Buckman | Director | March 10, 2026 | ||||||||||||
| Paul Buckman | ||||||||||||||
/s/ Michael Carusi | Director | March 10, 2026 | ||||||||||||
| Michael Carusi | ||||||||||||||
/s/ Andrew Hykes | Director | March 10, 2026 | ||||||||||||
Andrew Hykes | ||||||||||||||
/s/ Kevin Sidow | Director | March 10, 2026 | ||||||||||||
| Kevin Sidow | ||||||||||||||
/s/ Casey Tansey | Director | March 10, 2026 | ||||||||||||
| Casey Tansey | ||||||||||||||
140